
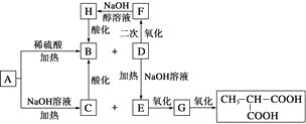
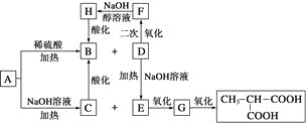
����Ŀ���������أ�KH2PO4����һ�ָ�Ч���Ϸʡ���ҵ��������[��Ҫ�ɷ���Ca3(PO4)2������������Fe2O3��CaF2������]Ϊԭ�ϣ������������ص�������ͼ��

��֪����TBP��D2EHPA��TOA����������ˮ��Һ̬�л������ȡ�ض������н�ǿ��ѡ���ԣ���������ȡ����
����ȡ��TBP��H3PO4��Fe3+�н�ǿ����ȡ���ã�����Ca2+��һ������ȡ���á�
����ȡ��D2EHPA����Fe3+�н�ǿ����ȡ���á�
�ش��������⣺
��1�������ᡱ��������������Ļ�ѧ����ʽΪ______��
��2����������ʱ������Ӧ�Ļ�ѧ����ʽΪ______��
��3����Ŀ��1����______��
��4����Ŀ��2����������л����г�H3PO4�⣬��������ij���������ӡ�ȥ���л����и������ӵķ������ú�H2SO4������ϴ�ӣ���Ӧ�����ӷ���ʽΪ______��
��5������Ӧ��ʱ���������м���KCl���ټ���TOA��TOA��������______��
��6������Ӧ���У�TOA��������Ӱ����Һ��pH��ˮ��Һ��H3PO4��H2PO4-��HPO42-��PO43-�ķֲ�������������Ԫ����ռȫ���������ӵ����ʵ�����������pH�ı仯��ͼ��ʾ��

����Ӧ���У���pH=______����ѡ��2.2������4.5������9.5����12.4����ʱ��ֹͣ����TOA��
���𰸡�Ca3(PO4)2+6HCl��3CaCl2+2H3PO4 SiO2+4HF��SiF4��+2H2O ��ȥ�����к��е�Fe3+ ![]() ˮ���д���KCl+H3PO4
ˮ���д���KCl+H3PO4![]() HCl+KH2PO4������TOA��HClת�Ƶ��л��㣬ƽ��������Ӧ�����ƶ���������KH2PO4���� 4.5
HCl+KH2PO4������TOA��HClת�Ƶ��л��㣬ƽ��������Ӧ�����ƶ���������KH2PO4���� 4.5
��������
������[��Ҫ�ɷ���Ca3(PO4)2������������Fe2O3��CaF2������]Ϊԭ�ϣ������������أ������̿�֪�������ᷢ��Ca3(PO4)2+6HCl��3CaCl2+2H3PO4��Fe2O3+6HCl��2FeCl3+3H2O��CaF2+2HCl��CaCl2+2HF��������Զ������跢��SiO2+4HF=SiF4��+2H2O��D2EHPA����Fe3+�н�ǿ����ȡ���ÿɳ�ȥ�����к��е�Fe3+����Һȡˮ���TBP��ȡH3PO4����Һȡ�л��������������ᣬ����KCl��ϡ��������KH2PO4���ټ����л���-������(TOA)���룬��ˮ��ᾧ�ɵ�KH2PO4��Ʒ���Դ������
��1�������ᡱ��������������Ļ�ѧ����ʽΪCa3(PO4)2+6HCl��3CaCl2+2H3PO4��
��2��HF����������跴Ӧ��������ʱ������Ӧ�Ļ�ѧ����ʽΪSiO2+4HF��SiF4��+2H2O��
��3��������ȡ��D2EHPA����Fe3+�н�ǿ����ȡ���ã���ˡ�Ŀ��1���dz�ȥ�����к��е�Fe3+��
��4����Ŀ��2����������л����г�H3PO4�⣬��������ij���������Ӹ����ӡ�ȥ���л����и������ӵķ������ú�H2SO4������ϴ�ӣ���Ӧ�����ӷ���ʽΪ![]() ��
��
��5������Ӧ��ʱ���������м���KCl���ټ���TOA������ˮ���д���KCl+H3PO4![]() HCl+KH2PO4������TOA��HClת�Ƶ��л��㣬ƽ��������Ӧ�����ƶ���������KH2PO4���ɣ�
HCl+KH2PO4������TOA��HClת�Ƶ��л��㣬ƽ��������Ӧ�����ƶ���������KH2PO4���ɣ�
��6����ͼ��֪��BΪ����![]() ����������pH������
����������pH������![]() ��������pH=4.5ʱ��
��������pH=4.5ʱ��![]() ��࣬pH�����ߣ�������
��࣬pH�����ߣ�������![]() �����ɣ���pH=4.5��ֹͣ����TOA��
�����ɣ���pH=4.5��ֹͣ����TOA��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��A��һ����Ϣ���ϵ����Ӽ�������ͬ�����£�A������ͬ�������������88.25������A�����и�Ԫ�����������ֱ�Ϊw(C)��54.4%��w(H)��7.4%��w(O)��18.1%��w(Cl)��20.1%��A�ڲ�ͬ�����¿ɷ�����ͼ��ʾ��һϵ�б仯��
��ش��������⣺
(1)A�ķ���ʽΪ__________��
(2)D���Ӻ��еĹ�������________________��
(3)����ת����ϵ�Ļ�ѧ����ʽ�У�����ˮ�ⷴӦ����________��(������)��
(4)д����ѧ����ʽ��
��A��ϡ���Ṳ�ȣ�____________________________________________________��
��E������G��__________________________________________________��
��F���������ƴ���Һ���ȣ�__________________________________________��
(5)��B��Ϊͬ���칹�����������״�������ʹ���____�֡�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�����з��ӻ������м����ɴ�С���е��ǣ� ��
��BCl3 ��NH3 ��H2O ��PCl4+ ��HgCl2
A.�ݢܢ٢ڢ�B.�ݢ٢ܢڢ�C.�ܢ٢ڢݢ�D.�ۢڢܢ٢�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ������(H3BO3)��һ��Ƭ��״�ṹ��ɫ���壬�����ֽ⡣���ڵ� H3BO3����ͨ���������(��ͼ��ʾ)���������й�˵������ȷ����

A.�����ᾧ������ԭ�Ӿ���B.H3BO3���ӵ��ȶ���������й�
C.1 mol H3BO3�����к���3 mol���D.��������ԭ�������Ϊ8�����ȶ��ṹ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��A��һ����Ϣ���ϵ����Ӽ�������ͬ�����£�A������ͬ�������������88.25������A�����и�Ԫ�����������ֱ�Ϊw(C)��54.4%��w(H)��7.4%��w(O)��18.1%��w(Cl)��20.1%��A�ڲ�ͬ�����¿ɷ�����ͼ��ʾ��һϵ�б仯��
��ش��������⣺
(1)A�ķ���ʽΪ__________��
(2)D���Ӻ��еĹ�������________________��
(3)����ת����ϵ�Ļ�ѧ����ʽ�У�����ˮ�ⷴӦ����________��(������)��
(4)д����ѧ����ʽ��
��A��ϡ���Ṳ�ȣ�____________________________________________________��
��E������G��__________________________________________________��
��F���������ƴ���Һ���ȣ�__________________________________________��
(5)��B��Ϊͬ���칹�����������״�������ʹ���____�֡�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��ij�л���A(C4H6O5)�㷺����������ˮ���ڣ�����ƻ�������ѡ����ϡ�ɽ���Ϊ�࣬��һ�ֳ��õ�ʳƷ���Ӽ����û���������������ʣ�
(i)��25��ʱ������ƽ�ⳣ��K��3.9��10-4��K2��5.5��10-6
(ii)A+RCOOH(��ROH)![]()
![]() ����ζ�IJ���
����ζ�IJ���
(iii)1molA![]() ��������1.5mol����
��������1.5mol����
(iv)�˴Ź�������˵��A��������5�ֲ�ͬ��ѧ��������ԭ����A��صķ�Ӧ��ͼ���£�

��1�����ջ�����A�����ʣ���A�Ľṹ���������ж���___��
a��ȷ����̼̼˫�� b���������Ȼ� c��ȷ�����ǻ� d���У�COOR������
��2��д��A��F�Ľṹ��ʽ��A��__��F��__��
��3��д��A��B��B��E�ķ�Ӧ���ͣ�A��B___��B��E__��
��4��д�����·�Ӧ�ķ�Ӧ������E��F�ڢٲ���Ӧ__��
��5���ڴ��������£�B���Ҷ����ɷ������۷�Ӧ�����ɵĸ߷��ӻ������������첣���֡�д���÷�Ӧ�Ļ�ѧ����ʽ��__��
��6��д����A������ͬ�����ŵ�A��ͬ���칹��Ľṹ��ʽ��___��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����������;�㷺�Ļ���ԭ�ϣ�������ˮ������ˮ�����������ʹ����ȡ�
��1����ҵ������ͭ�ķ����ܶࡣ
�� ����һ����Ũ�����ͭ��ȡ����ͭ���÷�Ӧ�Ļ�ѧ����ʽ��________���˷������ȱ����__________��
������������ϡ���ᡢͭ����������ȡ����ͭ����������Ҫ��������ͼ��ʾ��

ϡ���ᡢͭ����������Ӧ�Ļ�ѧ����ʽ��_________________��������Һ��ͨ���ȿ����ķ�Ӧ�����ӷ���ʽ��___________________������Һ�õ���ˮ����ͭ��ʵ�������______________��
��2���������������������Ṥҵβ���еĶ�������ͬʱ�Ƶ�����李���Ҫ�Ĺ�����������ͼ��ʾ��

���������з�����Ӧ�Ļ�ѧ����ʽ��_______________________��
�� �����ݱ���������������Һ��pH��5.5~6.0֮�䣬����Ч�ʽϸߡ�������һ��������β��ʱ��������Һ��pH�ķ�����________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��373Kʱ��ij 1L�ܱ������м���2mol NH3�������¿��淴Ӧ��2NH3��g��![]() N2��g��+3H2��g������������H2�����ʵ����仯����ͼ��ʾ��
N2��g��+3H2��g������������H2�����ʵ����仯����ͼ��ʾ��
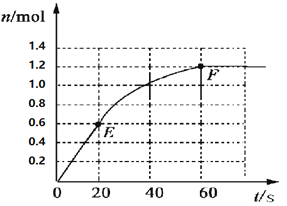
��1��ǰ20 s��NH3��g����ƽ����Ӧ����Ϊ___________��
��2��373Kʱ�÷�Ӧ��ƽ�ⳣ����ֵΪ______________��
��3�����ڴ�ƽ����ϵ���ټ���1mol��NH3����ԭƽ��Ƚϣ���ƽ��ʱNH3��ת����______�������С������ͬ����NH3��ƽ��Ũ��_________��
��4����ԭƽ��������473K�����´�ƽ��ʱ�������������䣩��H2��ƽ��Ũ��ΪNH3��2�����÷�Ӧ������ӦΪ_________������ȷ�Ӧ�������ȷ�Ӧ������Ϊ����ƽ����ϵ��H2�����ʵ��������д�ʩ��ȷ���ǣ�����������ͬ��______��
a�������¶� b��������������� c��������ʵĴ��� d���ٳ���N2
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��ij������ȤС���H2O2�ķֽ�������������ʵ��̽����
(1)�±��Ǹ�С���о�Ӱ���������(H2O2)�ֽ����ʵ�����ʱ�ɼ���һ�����ݣ���10mL H2O2��ȡ150mLO2�����ʱ��(��)
| 30% H2O2 | 15% H2O2 | 10% H2O2 | 5% H2O2 |
������������ | ��������Ӧ | ��������Ӧ | ��������Ӧ | ��������Ӧ |
���������� | 360 | 480 | 540 | 720 |
MnO2���������� | 10 | 25 | 60 | 120 |
�ٸ��о�С������Ʒ���ʱ��������Ũ�ȡ�___________��____________�����ضԹ�������ֽ����ʵ�Ӱ�졣
�ڴ�����Ӱ���������ֽ����ʵ���������ѡһ����˵�������ضԷֽ������к�Ӱ�죿_______________________��
(2)��������ͬ���ۼ�״̬��ͬ��MnO2�ֱ���뵽5mL 5%(�ܶ�Ϊ1.0g/cm3)��˫��ˮ�У����ô����ǵ�ľ�����ԡ��ⶨ������£�
ʵ�� ��� | ������MnO2�� | ���� ��� | �۲��� | ��Ӧ��� �����ʱ�� |
A | ��ĩ״ | ��ϲ��� | ���ҷ�Ӧ�������ǵ�ľ����ȼ | 5span>���� |
B | ��״ | ��Ӧ���������Ǻ�����ľ��δ��ȼ | 30���� |
�� д��H2O2�����ֽ�Ļ�ѧ��Ӧ����ʽ________________�����ʵ��A��H2O2��5�����ڵ�ƽ����Ӧ����________________�����������С�������λ���֣�
�� ʵ����˵���������õĴ�С��____________________�йء�
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com