
����Ŀ�����мס��ҡ����dz������л��������Ҫ��ش��������⣺
�ף�CH3CH=CH2 �ң�![]() ����HOCH2CH2CH2COOH
����HOCH2CH2CH2COOH
(1)��������_______��
(2)������������Ȼ�̼��Һ��Ӧ����Ӧ�Ļ�ѧ����ʽΪ______��
(3)��Ҳ�ܷ����ۺϷ�Ӧ������Ľṹ��ʽΪ_____________��
(4)����Ũ�����ϼ��ȣ�������Ԫ�����ʵĻ�ѧ��Ӧ����ʽ��____________��
(5)д���ҵ�ͬ���칹���У����������Һ˴Ź���������2��壬�������Ϊ3��2�Ľṹ��ʽΪ__________��
���𰸡���ϩ CH3CH= CH2 +Br2![]() CH3CHBrCH2Br
CH3CHBrCH2Br 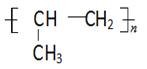 HOCH2CH2CH2COOH
HOCH2CH2CH2COOH![]()
 +H2O
+H2O ![]()
��������
(1)�ɼĽṹ��ʽ����ϳ������ʵ�������ʽ������
(2)����̼̼˫���������巢���ӳɷ�Ӧ��
(3)����̼̼˫�����ɷ����Ӿ۷�Ӧ���ɸ߾��
(4)�������ǻ����Ȼ����ɷ���������������Ӧ���ɻ�״����
(5)��Ϊ�ұ�(![]() )���ҵ�ͬ���칹���У����б�����Ϊ���ױ�����Ϻ˴Ź����������жϡ�
)���ҵ�ͬ���칹���У����б�����Ϊ���ױ�����Ϻ˴Ź����������жϡ�
(1)�ɽṹ��ʽ��֪��Ϊ��ϩ���ʴ�Ϊ����ϩ��
(2)��(CH3CH=CH2)�к���̼̼˫���������巢���ӳɷ�Ӧ����Ӧ�ķ���ʽΪCH3CH=CH2+Br2��CH3CHBrCH2Br���ʴ�Ϊ��CH3CH=CH2+Br2��CH3CHBrCH2Br��
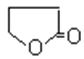
(3)��(CH3CH=CH2)�к���̼̼˫�����ɷ����Ӿ۷�Ӧ���ɸ߾������Ϊ![]() ���ʴ�Ϊ��
���ʴ�Ϊ��![]() ��
��
(4)��(HOCH2CH2CH2COOH)�к����ǻ����Ȼ����ɷ���������������Ӧ���ɻ�״����������Ԫ���ķ�ӦΪHOCH2CH2CH2COOH![]()
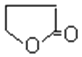 +H2O���ʴ�Ϊ��HOCH2CH2CH2COOH
+H2O���ʴ�Ϊ��HOCH2CH2CH2COOH![]()
 +H2O��
+H2O��
(5)��Ϊ�ұ�(![]() )���ҵ�ͬ���칹���У����б�������ͬ���칹��Ӧ��Ϊ���ױ������ڡ��䡢��3�֣����к˴Ź���������2��壬�������Ϊ3��2�Ľṹ��ʽΪ
)���ҵ�ͬ���칹���У����б�������ͬ���칹��Ӧ��Ϊ���ױ������ڡ��䡢��3�֣����к˴Ź���������2��壬�������Ϊ3��2�Ľṹ��ʽΪ![]() ���ʴ�Ϊ��
���ʴ�Ϊ�� ![]() ��
��


 ÿ��10���ӿ�����������������ϵ�д�
ÿ��10���ӿ�����������������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�������������ĿǰΪֹ������ǿ���������ô����������������������Ҫԭ������ϡ�������ϡ��ߴ�����������ȡ��ش��������⣺
��1����(Nd)Ϊ60��Ԫ�أ������ڱ���λ�ڵ�_________���ڡ���̬��ԭ����ռ������ܼ��ĵ��ӣ�����������ͼ��״Ϊ_______�������ӵĵ����Ų�ʽ__________________
��2��ʵ����AlCl3��ʵ�ʴ�����ʽΪAl2Cl6������ӵ����ģ������ͼ��ʾ��
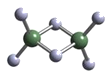
�� ������A1ԭ�Ӳ�ȡ____�ӻ���Al2Cl6����____����(���������������Ǽ�����)��
��AlCl3�����NaOH��Һ��Ӧ����Na[Al(OH)4]��[A1(OH)4]���д��ڵĻ�ѧ����_________(��ѡ����ĸ)��
A.���Ӽ� B.���ۼ� C.��λ�� D.������ E.���
��3����ҵ�Ͽ��õ�����ڵ�FeO��Fe2O3ұ���ߴ�������̬Fe2+�ļ۵����Ų�ͼ(�������ʽ)Ϊ______________�������ʽṹ֪ʶ˵��Fe2+���ױ�������Fe3+��ԭ��________________________��
��4������������ľ����ṹ��ͼ1��ʾ���þ�������������������֮��Ϊ_____��
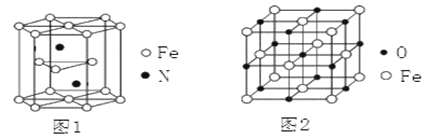
��5��������������ľ�����ͼ2��ʾ����֪����������������ܶ�Ϊ��g��cm��3��NA���������ӵ�������ֵ���ڸþ����У���Fe2+�����ҵȾ����Fe2+��ĿΪ_________��Fe2+��O2������̺˼��Ϊ_____________pm��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����ͼ�ǽ���þ��±�ص���(X2)��Ӧ�������仯ʾ��ͼ������˵����ȷ����
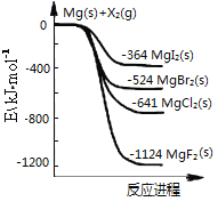
A. ±�ص���(X2)��ˮ��Ӧ��������������
B. MgF2�ĵ���ʽΪ��![]()
C. ���ȶ��ԣ�MgI2��MgBr2��MgCl2��MgF2
D. ��ͼ��֪���¶���MgI2(s)��Cl2(g)��Ӧ���Ȼ�ѧ����ʽΪ��MgI2(s)��Cl2(g)=MgCl2(s)��I2(g) ��H����277 kJ��mol��1
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��һ���¶��£���һ��������ܱ������н��п��淴ӦN2(g)+3H2(g)![]() 2NH3(g)������˵���У���˵���÷�Ӧ�Ѿ��ﵽ��ѧƽ��״̬����( )
2NH3(g)������˵���У���˵���÷�Ӧ�Ѿ��ﵽ��ѧƽ��״̬����( )
A.������N2��H2��NH3�������ʹ���
B.������N2��H2�ķ�����֮��Ϊ1:3
C.������N2��H2��NH3��Ũ�����
D.NH3���������ʺͷֽ��������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����������أ�K2S2O8���ڿ����빤ҵ������Ҫ��;��
��1��S2O82-�ĽṹʽΪ[![]() ]2-������SԪ�صĻ��ϼ�Ϊ_____����Ag+���£�S2O82-��ʹ��Mn2+����Һ����Ϻ�ɫ������������_____�������ӷ��ţ���
]2-������SԪ�صĻ��ϼ�Ϊ_____����Ag+���£�S2O82-��ʹ��Mn2+����Һ����Ϻ�ɫ������������_____�������ӷ��ţ���
��2��ij������ʪ��K2S2O8���������Ͱ����������ۺϴ���ȼú��¯�������������������Ч�ʣ�����Һ��������Ϊ����ֲ���̻��ķ��ϡ�
����������У�������ҺpH=6ʱ��n(SO32-)�sn(HSO3-)=____��[��֪��25��ʱ��Ka1(H2SO3)��1.5��10-2��Ka2(H2SO3)��1.0��10-7]
���������������η���������Ӧ����1��K2S2O8��NO������HNO2����2��K2S2O8��������HNO2����2����Ӧ�Ļ�ѧ����ʽΪ________________________________��һ�������£�NOȥ�������¶ȱ仯�Ĺ�ϵ��ͼ��ʾ��80��ʱ����NO��ʼŨ��Ϊ450 mg��m-3��t min�ﵽ���ȥ���ʣ�NOȥ����ƽ����Ӧ���ʣ�v(NO) =_______mol.L-1��min-1���д���ʽ����

��3����������ؿ�ͨ��������ת�����ᴿ�������Ƶã����װ������ͼ��ʾ��

�ٵ��ʱ�����缫���ӵ�Դ��______����
�ڳ����£����Һ�к�������Ҫ������ʽ��pH�Ĺ�ϵ����ͼ��ʾ����֪������Ķ������볣��1.02��10-2���������ŵ��������Ҫ��HSO4-���������������Һ��pH��ΧΪ________�������ĵ缫��ӦʽΪ________��

��������Ʒ�м�������أ�ʹ��ת��Ϊ��������شֲ�Ʒ���ᴿ�ֲ�Ʒ�ķ�����____________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��Ϊ��֤±�ص��������Ե����ǿ����ijС������ͼ��ʾװ�ý���ʵ��(�г���������ȥ���������Ѽ���)��
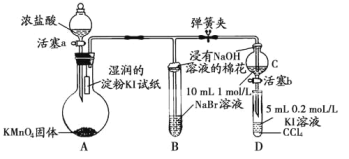
ʵ����̣�
��.���ɼУ�����a���μ�Ũ���ᡣ
��.��B��C�е���Һ����Ϊ��ɫʱ���н����ɼС�
��.��B����Һ�ɻ�ɫ��Ϊ����ɫʱ���رջ���a��
��.����
(1)A�в�������ɫ���壬�����ʽ��_____��
(2)��֤������������ǿ�ڵ��ʵ��������____��
(3)B����Һ������Ӧ�����ӷ���ʽ��______��
(4)Ϊ��֤���������ǿ�ڵ⣬���̢��IJ�����������_____��
(5)���̢�ʵ���Ŀ����______��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��������������ػ�������;�dz��㷺���ش��������⣺
(1)��̬Nԭ�ӵĺ�������Ų�ʽΪ___��Crλ��Ԫ�����ڱ���������___�塣
(2) Cr��Kλ��ͬһ������������������ͬ������Ԫ��ԭ�ӵ�һ�����ܵĴ�С��ϵΪ___��Crcl3���۵�(83��)��CrF3���۵�(1100��)�͵ö࣬������Ϊ___��
(3) Cr��һ�������ṹ��ͼ��ʾ��

��������C1O4���Ŀռ乹��Ϊ___�Ρ�
���������У��������ӵ���λ��Ϊ___��N������ԭ���γɵĻ�ѧ����Ϊ___����
������H2 NCH2 CH2 NH2(�Ҷ���)��̼ԭ�ӵ��ӻ���ʽ��______ ������������Ԫ�ص縺�ԴӴ�С��˳��Ϊ___
(4)���������۵�Ϊ1770�棬����һ�־���ľ����ṹ��ͼ��ʾ�����ܶ�Ϊ5. 9 g��cm ��3���������ľ����߳�Ϊ___(�г�����ʽ)nm.

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��![]() Ϊ�����ӵ�������ֵ������˵����ȷ����
Ϊ�����ӵ�������ֵ������˵����ȷ����
A. 0.1 mol ��![]() ������
������![]() ������
������
B. pH=1��H3PO4��Һ�У�����![]() ��
��![]()
C. 2.24L����״��������O2����ȫȼ�գ��õ�![]() ��CO2����
��CO2����
D. �ܱ�������1 mol PCl3��1 mol Cl2��Ӧ�Ʊ� PCl5��g��������![]() ��P-Cl��
��P-Cl��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��ʵ��С��Ӹ��� NaBr �Ĺ�ҵ��ˮ����ȡBr2 �Ĺ�����Ҫ��������������ȡ����Һ������Ȳ��衣
��֪��������ʱ����ֵķе����Խ����Ч��Խ�ã��ڿ����õ���������Ϣ��װ�����£�
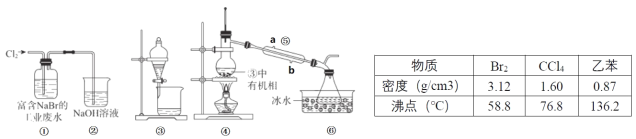
����˵���������
A.�������������ն��� Cl2����ֹ������Ⱦ
B.����������ȡʱ��ѡ���ұ��� CCl4 ������
C.�ұ��� CCl4 ��ȡ Br2 ʱ������ͬ�����Ƿֲ㣬�ϲ���ɫ���²��ɫ
D.������������Ϊ�����ܣ����� b Ϊ��ˮ�ڣ�a Ϊ��ˮ��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com