
��18�֣���ҵβ���е�������ͨ�����ð������շ�����ԭ����NH3��NOx�ڴ��������·�Ӧ�����������ʡ�ijУ�С��ͬѧ��������װ�úͲ���ģ�ҵ�ϵ������� �Ĵ������̡�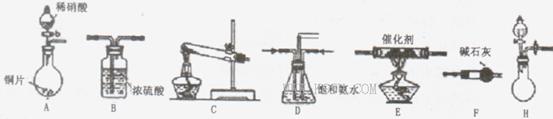
I��̽����ȡNH3�ķ���
��1��Bװ�õ����ƣ�______________
��2��������װ���У�H�ܿ��١������ȡ��װ������Ҫ���ӵķ�Ӧ�Լ�Ϊ_________ ��
��3��Ϊ̽�����õ�ʵ��Ч�����С��ͬѧ��������Cװ������ȡ�������ڿ���ʵ��������ͬ������£�����±���ʵ�����ݡ�
�����������ݣ�����Ϊ���ַ�����ȡ������Ч�����________������ţ����Ӹ÷���ѡ���ԭ�Ϸ�������Ч���õĿ���ԭ����________��__________��
| �Լ������� | �����Լ� | NH3�����mL�� | |
| a | 6.0gCa(OH)2���� | 5.4gNH4Cl | 1344 |
| b | 5.4g(NH4)2SO4 | 1364 | |
| c | 6.0gNaOH���� | 5.4gNH4Cl | 1568 |
| d | 5.4g(NH4)2SO4 | 1559 | |
| e | 6.0gCaO���� | 5.4gNH4Cl | 1753 |
| f | 5.4g(NH4)2SO4 | 1792 | |

��18�֣���1��ϴ��ƿ(1��)
��2����ʯ����Ũ��ˮ�����ռ���Ũ��ˮ������ѧʽ���֣�(2��)
��3��f (2��)����NH4��2��SO4�����Ȳ��ֽ⣬CaO������ˮ���ã������𰸾����֣�(2��)
��1��F E B(2��)
��2��3Cu+2NO3-+8H+=2NO��+4H2O +3Cu2+(2��)
��3��ʹ�����Ͼ��ȣ����������ٶȣ���ֹ�����ܽ⣨���������(3��)
��4��b(2��)
��5��δ��Ӧ��NO������������Ⱦ����(2��)
���������������1��װ��B��������ϴ��ƿ��
��2���ܿ��١������ȡ���������Լ�ΪŨ��ˮ����ʯ�һ�Ũ��ˮ���������ƶ����ԣ�����H����Ҫ���ӵķ�Ӧ�Լ�Ϊ��ʯ����Ũ��ˮ�����ռ���Ũ��ˮ����
��3�����ݱ������ݣ���ͬ�����������Ӧ�����İ���������ͬ��f���������İ�����࣬�Ҽ���ʱ����鱗��ֽ⣬��ʯ�һ�������ˮ���ã����Ȼ�����ȷֽ�����������Ȼ��⣬�����ֽ�ϳ��Ȼ�泥��������ƻ�������������η�Ӧ������ˮ�϶࣬����f������ȡ������Ч����ã�
��1��װ��A���ɵ�NO��װ��C���ɵ�NH3ͨ��װ��Dʹ�����Ͼ��ȡ����������ٶȣ�װ��F�������壬��ͨ��װ��E��Ӧ���з�Ӧ����Ӧ�������ͨ��װ��B�������ռ����壬�ʴ�Ϊ��FEB��
��2��ϡ�������ǿ�����ԣ���ͭ����������ԭ��Ӧ��������ͭ��NO�ȣ���Ӧ�����ӷ���ʽΪ 3Cu+8H++2NO3-=3Cu2++2NO��+4H2O��
��3��Dװ��ʹ�����Ͼ��ȡ����������ٶȣ����⣬���������ڱ��Ͱ�ˮ���ɷ�ֹ������
��4����������������ͭ��Һ��ˮ�Լ������У��ɲ�������������Ϊ���Է��ӣ������ڷǼ����ܼ�������CCl4���汥�Ͱ�ˮ����ѡb��
��5��ȱ��β������װ�ã�������NO����Ⱦ������
���㣺���鵪��������Ի�����Ӱ�죬��������ȡ���������ӷ���ʽ����д��װ�����õ��жϣ��Լ���ѡ ��


 ���ݼ���ϵ�д�
���ݼ���ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
A��G��X��Y������ѧ��ѧ�������ʣ�����B��D��E��F��G�ڳ�����Ϊ������BΪ����ɫ��C�Ǻ�ɫ�Ľ������ʡ�����֮��������ת����ϵ�����з�Ӧ�ۢܢߵIJ����е�ˮ����ȥ���������
��1��B�Ļ�ѧʽ________��
��2����Ӧ�����������뻹ԭ�������ʵ���֮��Ϊ ��
��3��д����Ӧ�ߵ����ӷ���ʽ_____________________��
��4��д�����X��Һ��������Ӧʽ______________________�������һ��ʱ���ת����0��2mol���ӣ����ʱ�������������ڱ�״���µ����Ϊ ��
��5��д����Ӧ�ܵĻ�ѧ����ʽ______________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
���仯��������Ȼ���ձ���ڡ�����ת����
��ɽ�緢�������ʽ���ʱ�������ж�����H2S��SO2�Ƚ����������һЩ��Ȼת����������������
��1��H2S��SO2������ײʱ����Ⱦ�ή�͡���Ӧ����ʽΪ_________________________��
��2��H2S�ڳ����£��ᱻ��������������Ӧ����ʽΪ_________________________��
��3��SO2��Ʈ�����£��ᱻ��������ΪSO3����ˮ�������ᣬ������ʯ����ת��Ϊ�ȶ���ʯ�����(CaSO4��2H2O)��SO2�������Ļ�ѧ����ʽΪ_____________________________��
�ڻ�����ʩ�ϣ��������Ǵ�����ʦ����Ȼ������ʾ��
����Ϊ�����ŷŶ�������ȣ��ᳬ����Ȼ���������������������ػ�����Ⱦ����ʯ��-ʯ�෨���͡��ռ���ǹ�ҵ�ϳ��õĹ�ҵ��������
��4����ʯ��-ʯ�෨�����ն������������Ϊ��
��SO2������ʯ����ܻ�ѧ����ʽΪ________________________________________��
��5�����ռ��������������Ũ��Һ���պ�������������������ŵ����������Ƽ���ǿ�����տ졢Ч�ʸߡ���ѧ����ʽΪ______________________________________�����ռ�ļ۸�Ϲ�ʯ��0��36Ԫ/kg���ռ�2��90Ԫ/kg����
��6��������ʯ��-ʯ�෨�� �͡��ռ����������������������������ٸ�Ч�ͳɱ�����Ⱦ�ĺ�Ч���������Եõ�ʯ���Ʒ��������������ͼ�Ģ١��ܴ���д���ʵĻ�ѧʽ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
���Ǵ���������������ҪԪ��֮һ���Ӵ�ͳ���ϵ���Ϣ���ϵķ�չ�����д�����һ����һ���漣��
��1������Ԫ�����ڱ��е�λ����_______��
��2����ҵ�����ֹ�ķ�Ӧ�У�SiO2+2C Si���֣�+2CO����SiO2+3C
Si���֣�+2CO����SiO2+3C SiC+2CO��������Ʒ�е��ʹ���̼��������ʵ���֮��Ϊ1��1����μӷ�Ӧ��C��SiO2������֮��Ϊ ��
SiC+2CO��������Ʒ�е��ʹ���̼��������ʵ���֮��Ϊ1��1����μӷ�Ӧ��C��SiO2������֮��Ϊ ��
��3����ҵ�Ͽ���ͨ������ͼ��ʾ��������ȡ���裺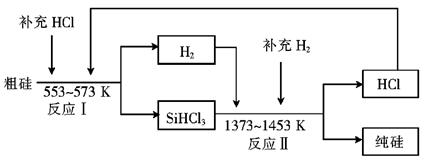
������ӦIΪ Si���֣�+3HCl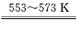 SiHCl3+H2��
SiHCl3+H2��
��ӦII�Ļ�ѧ����ʽΪ ��
�������Ʊ����̱����ϸ������ˮ������SiHCl3��ˮ���ҷ�Ӧ����H2SiO3��HCl���� һ�����ʣ�д���÷�Ӧ�Ļ�ѧ����ʽ�� ��
�ۼ���ÿһ�ִ��Ʊ�1mol���裬�����������й�Ԫ��û����ʧ����ӦI��HCl��������Ϊ90%����ӦII��H2��������Ϊ93.75%�����ڵڶ��ִε������У�����Ͷ��HCl ��H2�����ʵ���֮���� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��ʴ���
��12�֣����������������繤ҵ����ˮ����ķ������ʺϴӵ�Ũ�Ⱥ�����Һ����ȡ�塣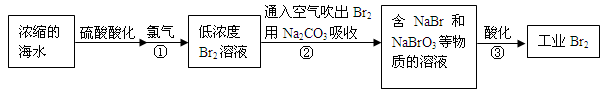
��1��NaBr�ĵ���ʽ�� ��
��2����Ӧ���ǽ�Br��ת��ΪBr2����Ӧ�ٵ����ӷ���ʽ�� ��
��3��ͨ���������Br2������Na2CO3���յ�Ŀ���� ��
��4����Ӧ�ڵĻ�ѧ����ʽ�� ��
��5����Ӧ����ÿ����3 mol Br2��ת�Ƶ��ӵ����ʵ����� mol��
��6��Ϊ�˳�ȥ��ҵBr2������Cl2������ҵBr2�� ������ĸ����
a��ͨ��HBrb������NaBr��Һ c������Na2CO3��Һ d������Na2SO3��Һ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��ʴ���
ͼ��-?�ֱ�����йط�Ӧ�е�һ�����ʣ����������¿հס�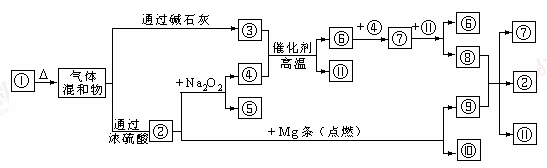
��1���١��ܵĻ�ѧʽ�ֱ���______��______��
��2���õ���ʽ��ʾ�ڵ��γɹ���Ϊ ��
��3������ᷴӦ�Ļ�ѧ����ʽ��______������۵Ĵ��ڵķ����� ��
��4��MgҲ��������ϡ��Һ��Ӧ���ɢޣ�д����Ӧ�����ӷ���ʽ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�������
��ͭƬ�м���115mLijŨ�ȵ����ᣬ�ڼ��������·�Ӧ����ͭƬȫ���ܽ������Һϡ�͵�500mL���ټ�������п�ۣ�ʹ֮��ַ�Ӧ���ռ���2��24L����״�������塣���˲������壬��������������������7��5g��
��1����μӷ�Ӧ��п�۵����ʵ���
��2��ԭ��������ʵ���Ũ�ȡ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��15�֣����������μ����ֽ��Ҳ���ϸ��ӡ���Mg(NO3)2Ϊ�о������ĸ�ѧϰС����ͨ��ʵ��̽�����ȷֽ�IJ���ֱ��������4�ֲ��룺
���飺Mg(NO2)2��NO2��O2���� ���飺MgO��NO2��O2��
���飺MgO��NO2��N2�������� ���飺Mg3N2��O2
��1��ʵ��ǰ��С���Ա�������϶� ��IJ���һ����������������_____________��
�������ϵ�֪��2NO2+2NaOH=NaNO3+NaNO2+H2O
�������С����룬�������ͼ��ʾ��ʵ��װ�ã�ͼ�м��ȡ��г������Ⱦ�ʡ�ԣ�����̽����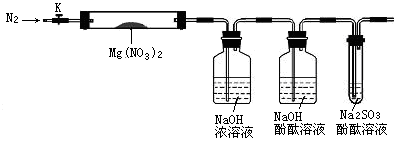
A B C D
��2��ʵ�����
�ټ��������װ�������Եķ��� ��
�ڳ�ȡӲ���Թ�A������Ϊ18.0g���Թ�A��Mg(NO3)2���干21.8 g����A�У�����ǰͨ��N2������װ���ڵĿ�������Ŀ���� ���ر�K���þƾ��Ƽ���ʱ����ȷ�������� Ȼ��̶��ڹ��й��岿λ�¼��ȡ�
�۹۲쵽A���к���ɫ�������ɣ�C�������ݡ�
�ܴ���Ʒ��ȫ�ֽ⣬Aװ����ȴ�����¡����������Ӳ���Թ�A��ʣ������������Ϊ19.0g��
��ȡ����ʣ��������Թ��У���������ˮ��δ����������
��3��ʵ������������
��֤��һ����O2���ɵ������� �������صĻ�ѧԭ��Ϊ���û�ѧ����ʽ��ʾ�� ��
����ʵ�������ʣ�����������������ɳ���ȷ�ϲ���_______���������ȷ�ġ�
��һλͬѧ��Ϊ����װ�ò���ȷ�Ϸֽ��������O2��������������Ϊ������ ���������Ľ�װ�ã�Ӧ�� ��
��4�������Ϸ�����Mg(NO3)2�ֽ�Ļ�ѧ����ʽΪ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
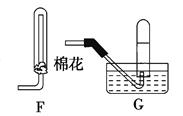
��18�֣�ʵ���ҳ�������װ���Ʊ����ռ������������壬��̽�������ʡ�
��1��װ��A�еķ�Һ©����ʢװ��Һ��ͨ���� ��Բ����ƿ��Ԥ�ȼ������ͭƬ�������ļ۸��ͭ�ļ۸�ͣ��˴���ͭƬ������Ƭ��ԭ���� ��
��2�������D��װ�ĸ��������ˮ�Ȼ��ƣ��������� ��
��3������ʱ������ƣ�Ũ������ϡ��װ��E������������β�������չ����з�����Ӧ�Ļ�ѧ����ʽ���£�2NO2��2NaOH��NaNO3��NaNO2��H2O ��NO2��NO��2NaOH��2NaNO2��H2O
��NO��NO2����������ɿɱ�ʾ��NOx���û������ͨ��NaOH��Һ����ȫ����ʱ��x��ֵ����Ϊ ������ĸ��
| A��1.1 | B��1.2 | C��1.5 | D��1.8 |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com