
����Ŀ��������Ҫ�ĺϽ�Ԫ�أ������������������ȺϽ���Ҳ�������л�����Ĵ�������ȡ����
��1��д����̬Cr�ļ����Ų�ʽ__________��Cr�й���__________�ֲ�ͬ�ܼ��ĵ��ӡ�
��2��Ni(CO)n��Fe(CO)5ͬ�������ʻ������γ������ʱ��ÿ��CO�ṩһ�Ե��������ԭ���γ���λ�����о����ֽ���ԭ�ӵļ۵��Ӻ�CO�ṩ�ĵ����ܺ͵���18��
��Ni��C��O�ĵ縺���ɴ�С��˳��Ϊ____________________��
�� Ni(CO)n������n=__________��
����֪Ni2+��Fe2+�����Ӱ뾶�ֱ�Ϊ69pm��78pm�������ҽ������������ڵ�NiO��FeO�ҽ�����ȴ�����У�NiO�������Ƚᾧ���Խ�����ԭ��____________________��
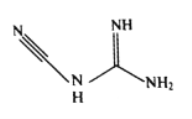
��3���춨�����ܡ�ͭ�ٵȿ���˫�谷����ѧʽC2H4N4����ṹ��ʽ��ͼ��ʾ��˫�谷������̼ԭ�ӵ��ӻ���ʽ��__________�����ӽṹ�м������Ĺ��ۼ���__________��
��4�����ľ���ṹ�������Ͻ�ľ�����ͼ��ʾ��
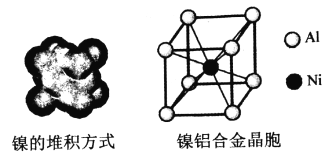
��������Ķѻ���ʽΪ__________��
����֪Al�ĵ�һ���ڶ������ֱܷ�Ϊ��l1=578kJ/mol��l2=1817kJ/mol������l2Զ����l1��ԭ��__________________________________________________��
����֪�������Ͻ��ܶ�Ϊdg/cm3��NA��������٤���������������ĺ˼��Ϊ__________ pm��(�ô���ʽ��ʾ)
���𰸡� [Ar]3d3 6 O>C>Ni 4 Ni2+���Ӱ뾶С��Fe2+��NiO�����Ӽ���ǿ�������ܸ��ߣ���NiO�۵����FeO,�ҽ���ȴ������NiO�Ⱦ��� sp��sp2 C![]() N �����������ܶѻ���ccp��fcc ��ΪAlʧȥһ�����Ӻ��������3s2,s��ȫ����״̬���Ƚ��ȶ����ڶ������Ӻ���ʧȥ������I2Զ����I1
N �����������ܶѻ���ccp��fcc ��ΪAlʧȥһ�����Ӻ��������3s2,s��ȫ����״̬���Ƚ��ȶ����ڶ������Ӻ���ʧȥ������I2Զ����I1 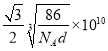
����������1��Cr����24��Ԫ�������ݹ���ԭ����֪Cr�ĺ�������Ų�ʽ��1s22s22p63s23p63d3����Ϊ[Ar]3d3���ɻ�̬��������Ų�ʽ��֪����6�ֲ�ͬ�ܼ���
��2����Ni��C��O�ĵ縺���ɴ�С��˳��ΪO>C>Ni���� Ni(CO)n�����н���ԭ�ӵļ۵��Ӻ�CO�ṩ�ĵ����ܺ͵���18��Ni�ļ۵�����Ϊ10��һ��CO�ṩ2�����ӣ�����nӦ����4����Ni2+���Ӱ뾶С��Fe2+��NiO�����Ӽ���ǿ�������ܸ��ߣ���NiO�۵����FeO,�ҽ���ȴ������NiO�Ⱦ�����
��3�����ݽṹ��ʽͼ��Cԭ�ӵĻ�ѧ�����ӷ�ʽ��ֱ���ͺ�ƽ�����������ֹ��ͣ��ɴ��ƶ��ӻ���ʽΪsp��sp2���֣����ܴ�С˳��Ϊ������>˫��>�������ʷ��ӽṹ�м������Ĺ��ۼ���C![]() N��
N��
��4���ٸ���ͼʾ��֪��������Ķѻ���ʽΪ�������������ܶѻ���ccp��fcc����Al�Ļ�̬�����Ų�ʽΪ1s22s22p63s23p1��ʧȥ3p1�ĵ��ӱȽ��������Ե�һ�����ܽ�С��ʧȥ3p1��3s2Ϊȫ����������ʧȥ���ӣ����Եڶ�������Զ���ڵ�һ�����ܡ��۸��ݾ����ṹ��һ�������к���1��Alһ��Ni������һ������������Ϊ�� ![]() g�������ı߳�Ϊ
g�������ı߳�Ϊ![]() cm���������ľ���Ϊ�����Խ��ߵ�һ�룬���Ծ���Ϊ
cm���������ľ���Ϊ�����Խ��ߵ�һ�룬���Ծ���Ϊ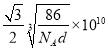 pm��
pm��


 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��������̬����ɵĻ��������ȫȼ�պ����õ�CO2��H2O�����ʵ��������������ʵ����ı仯��ͼ��ʾ.�����йػ�������˵����ȷ���ǣ� ��
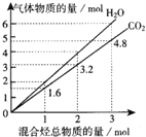
A. �û������һ��������ϩ
B. �û������һ�����������Ȳ
C. �ڳ����£��û������������������ϣ���ȫȼ�պ�ָ�������ȼ��ǰ�����������
D. �����������CH4��C2H4��ɣ����������Ϊ2��3
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�������ѱ���Ϊ��21���ͽ���������ҵ����������(��Ҫ�ɷ�FeTiO3)�Ʊ�TiCl4����Ҫ������������:

�ش���������:
��1����֪�����ӦFeTiO3(s)+2H2SO4(aq)=FeSO4(aq)+TiOSO4(aq)+2H2O����FeTiO3����Ԫ�صĻ��ϼ�Ϊ_____������TiOSO4��Һ���Ʊ�TiO2��nH2O���壬�䷴Ӧ�Ļ�ѧ����ʽΪ__________��Ϊ����������������ʣ��ɲ�ȡ�İ취�������ʯ�⣬�����Բ�ȡ�İ취��__________��_______(д�����ַ���)��
��2������A��Ŀ����____(����ĸ)��
A.��ֹFe2+������ B.��Fe3+��Ӧ C.��Fe2+ˮ��
��ҺII������II�ɵø���ƷFeSO4��7H2O�������II����Ũ������ȴ��_____��_____��
��3�������£�����ҺII��Fe2+��Ũ��Ϊ0.49mol/L��Ϊ��ֹ����Fe(OH)2��������Һ��pH������_____��(��֪Ksp[Fe(OH)2]=4.9��10-17)
��4���������յ��м����Ļ�ѧ����ʽΪH2TiO3(s) ![]() TiO2(s)+H2O(g),�������������У������뽹̿������ʱ����TiCl4�Ļ�ѧ����ʽΪ__________________________��
TiO2(s)+H2O(g),�������������У������뽹̿������ʱ����TiCl4�Ļ�ѧ����ʽΪ__________________________��
��5��������TiO2ͨ���������ַ����Ʊ�������:
����һ: ͨ�����·�Ӧ�Ʊ�������
��TiO2 (s)+2Cl2(g) ![]() TiCl4(g)+O2(g) �� H=+151kJ/mol
TiCl4(g)+O2(g) �� H=+151kJ/mol
��TiCl4+2Mg![]() 2MgCl2+Ti
2MgCl2+Ti
������:��TiO2��������ʯī������������CaOΪ���Һ��̼�������۳أ����TiO2�Ƶ��ѣ�������һ�����ɵ�������___________���������ɵ�������______________��
����ɫ��ѧ�Ƕȣ�����Ϊ�������ַ��������ָ��ã���˵������___________________________
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����ʽ̼��ͭ��ʾΪ��xCuCO3��yCu(OH) 2��zH2O,�ⶨ��ʽ̼��ͭ��ɵķ����ж��֡�
����������ԭ������ش��������⣺
(1)xCuCO3��yCu(OH) 2��zH2O��������Ӧ�Ļ�ѧ����ʽ______________________________
(2)���������������Ӷ��ɣ������������������˳���ǣ����������ӿ���ĸ��ţ���
��a������_______����_______������_______����_______������_______����_______������_______����_______������_______����_______������l��

(3)23.9gij��ʽ̼��ͭ��Ʒ����ַ�Ӧ��õ�12.7g���������4.4g������̼��7.2gˮ������Ʒ�Ľᾧˮ����Ϊ__________ g����ѧʽΪ____________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����(Se)�ǵ�34��Ԫ�أ��������ڲ��ɻ�ȱ����Ԫ�أ������γ�H2Se��SeO2��H2SeO3��H2SeO4��CuSe�ȶ��ֻ������ش��������⣺
��1������Ԫ�����ڱ��е�λ��________________________��
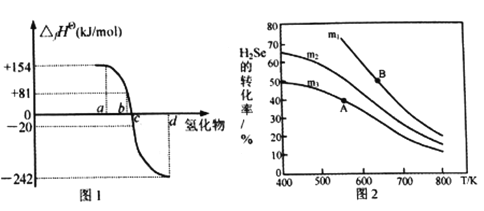
��2����101kPa��һ���¶�(һ����298K)�£����ȶ����ʷ�����Ӧ����1mol������ķ�Ӧ�Ƚиû�����ı�������(��fH��)��ͼ1Ϊ����Ԫ���⻯��a��b��c��d����̬ʱ�����������ݡ�
��ͼ1���⻯��d�ĵ���ʽΪ__________________________��
����298Kʱ��������ֽⷴӦ���Ȼ�ѧ��Ӧ����ʽΪ__________________________��
����ͼ�����ݼ��㣬2H2Se(g)+O2(g) ![]() 2Se(s)+2H2O(g) ��H=_____________KJ/mol
2Se(s)+2H2O(g) ��H=_____________KJ/mol
��3���ں��ݷ�Ӧ���У���H2Se(g)��O2(g)����ͬ����[n(H2Se)/n(O2)=m]Ͷ�뷴Ӧ������÷�Ӧ2H2Se(g)+O2(g) ![]() 2Se(s)+2H2O(g)��H2Se��ƽ��ת�������¶ȱ仯��ͼ2��ʾ����A��B����ƽ�ⳣ����С��ϵΪKA________KB(����<������>������=��)��ͼ��m1��m2��m3�ɴ�С��˳��Ϊ ____________��������____________________________________��
2Se(s)+2H2O(g)��H2Se��ƽ��ת�������¶ȱ仯��ͼ2��ʾ����A��B����ƽ�ⳣ����С��ϵΪKA________KB(����<������>������=��)��ͼ��m1��m2��m3�ɴ�С��˳��Ϊ ____________��������____________________________________��
��4���������ܶȻ���Ksp(CuSe)=7.9x10-49��Ksp(CuS)=1.3��10-36����ӦCuS(s)+Se2-(aq) ![]() CuSe(s)+S2-(aq)�Ļ�ѧƽ�ⳣ��KΪ____________(����ÿ�ѧ��������ʾ��������2λС��)������Һ��c(S2-)=100c(Se2-)ʱ����Ӧ��v(��)_____v����)(����<������>������=��) ��
CuSe(s)+S2-(aq)�Ļ�ѧƽ�ⳣ��KΪ____________(����ÿ�ѧ��������ʾ��������2λС��)������Һ��c(S2-)=100c(Se2-)ʱ����Ӧ��v(��)_____v����)(����<������>������=��) ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����������������ǣ�������
A. �����۵��ɸߵ��ͣ�MgO��H2O��NH3
B. �۵��ɸߵ��ͣ����ʯ����������������
C. Ӳ���ɴ�С�����ʯ��̼���裾�����
D. �������ɴ�С��NaF��NaCl��NaBr��NaI
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����֪�Ȼ�ѧ����ʽ��H��(aq)��OH��(aq)===H2O(l)����H1����57.3 mol��L��1��![]() H2SO4(Ũ)��NaOH(aq)===
H2SO4(Ũ)��NaOH(aq)===![]() Na2SO4(aq)��H2O(l)����H2��m������˵������ȷ����
Na2SO4(aq)��H2O(l)����H2��m������˵������ȷ����
A. �����Ȼ�ѧ����ʽ�еļ�������ʾ��������
B. ��H1>��H2
C. ��H2ӦΪ![]() H2SO4(Ũ)����ˮ�������仯���к���֮����
H2SO4(Ũ)����ˮ�������仯���к���֮����
D. |��H1|��|��H2|
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ������ʵ�������Ҫ�ò����������в�����������ͬ���ǣ�������
�ٹ��� ������ ��������ƿת��Һ�壮
A.�ٺ͢�
B.�ٺ͢�
C.�ں͢�
D.�٢ڢ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����������ǵ�ǰ����Դ�����о���һ�����Ų��ϡ���һ�������£���Cu-Znԭ�������Դ������ͼ��ʾװ�ÿ�ʵ���л���Ĵ��⣬�����й�˵����ȷ���ǣ� ��
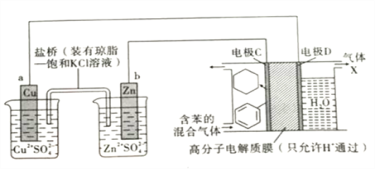
A����Cu-Znԭ���װ�ù���ʱ�������ڵ�Cl-��ͭ�缫һ���ƶ�
B���缫CΪ�������ڵ缫D�Ϸ�����ԭ��Ӧ
C������X����Ҫ�ɷ���H2
D��H+���߷���Ĥ�����ҽ������ң��ڵ缫C�Ϸ�����ԭ��Ӧ
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com