
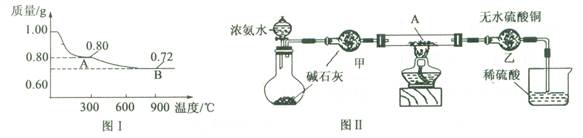
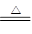 CuO��H2O���ɷ�Ӧǰ�����������ʵ�������Ϊ98��80��1.00��0.80�ɲ²��A����ΪCuO
CuO��H2O���ɷ�Ӧǰ�����������ʵ�������Ϊ98��80��1.00��0.80�ɲ²��A����ΪCuO 2Cu2O+O2���ɿ�������Ӧǰ�����������ʵ�������Ϊ80��72���������⣬�ʿ�֪B����ΪCu2O
2Cu2O+O2���ɿ�������Ӧǰ�����������ʵ�������Ϊ80��72���������⣬�ʿ�֪B����ΪCu2O

 ���Ŀ��ּ�����ҵ�����ҵ����������ϵ�д�
���Ŀ��ּ�����ҵ�����ҵ����������ϵ�д� ����ѵ��ϵ�д�
����ѵ��ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
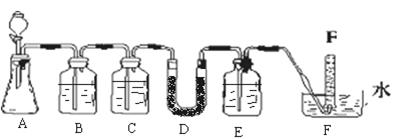
| ���� | �����Լ� | ������Լ���Ŀ�� |
| B | ����NaHCO3��Һ | |
| C | | ��ȥCO2�е�ˮ���� |
| E | | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
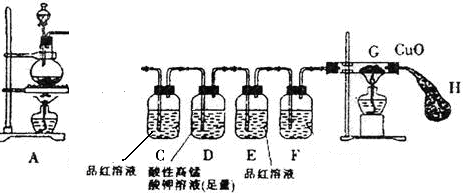
| ʵ������ | ���ۼ����� | |
| �ٽ�������������������ƿ�У��������еμӺ�amolH2SO4��Ũ������Һ | δ���������� | ԭ�� �� |
| �ڵ�ȼA��G���ƾ��� | Ƭ�̺���ƿ����Һ����ɫ�����仯���������ݲ�����C����Һ��ɫ ��E����Һδ�����Ա仯��һ��ʱ���ɫ��CuO����˺�ɫ����ʱEװ����Һ��ɫ��δ����ɫ�仯 | ��ƿ�з��������з�Ӧ �Ļ�ѧ����ʽ�� ˵��������SO2������ȫ��D����Һ���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
| | ʵ�鲽�� | Ԥ������ͽ��� |
| �� | ��A�Թ�ȡ��������ʯ��ˮ���ã���B�Թ�ȡ������Ʒ������B�Թܼ���__________ | ��������ų��ҳ���ʯ��ˮδ�����ǣ������________������ |
| �� | | |
| �� | | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
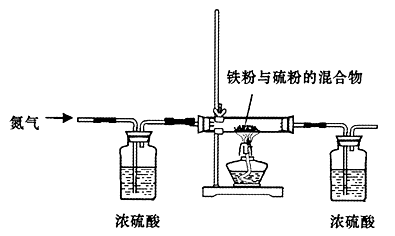
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
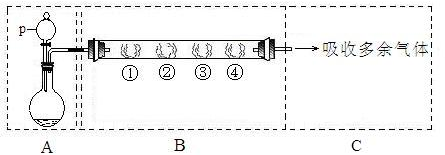
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
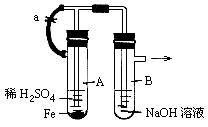
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����

| A��[(b��a)/84]��22.4��1000 mL |
| B��[(b��c)/31]��22.4��1000 mL |
| C��[(c��a)/106]��22.4��1000 mL |
| D��[(c��a)/106]��2��22.4��1000 mL |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
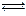 2NH3(g)+CO2(g)
2NH3(g)+CO2(g)| �¶�/�� | 15.0 | 20.0 | 25.0 | 30.0 | 35.0 |
| ƽ����ѹǿ/kPa | 5.7 | 8.3 | 12.0 | 17.1 | 24.0 |
| ƽ��������Ũ��/mol?L-1 | 2.4��10-3 | 3.4��10-3 | 4.8��10-3 | 6.8��10-3 | 9.4��10-3 |
| A��2v(NH2)=v(CO2) | B���ܱ���������ѹǿ���� |
| C���ܱ������л��������ܶȲ��� | D���ܱ������а���������������� |
 NH4HCO2+NH3?H2O
NH4HCO2+NH3?H2O
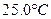 ʱ��0-6min ���������ˮ�ⷴӦ��ƽ������ ______��
ʱ��0-6min ���������ˮ�ⷴӦ��ƽ������ ______���鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com