
����ѧ����ѡ��3�����ʽṹ�����ʡ�(15��)
±��Ԫ�صĵ��ʺͻ�����ܶ࣬���ǿ���������ѧ���ʽṹ�����ʵ����֪ʶȥ��ʶ���������ǡ�
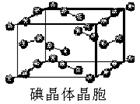
��1��±��Ԫ��λ��Ԫ�����ڱ���_________������ļ۵����Ų�ʽΪ____________________��
��2����һ��Ũ�ȵ���Һ�У���������Զ����ӵ�(HF)2��ʽ���ڵġ�ʹ�������ӵϵ���������________��
��3��������±��ṩ�ĵ�һ�����������жϣ����п������ɽ��ȶ��ĵ��������ӵ�±��ԭ����_________��
| | �� | �� | �� | �� |
| ��һ������ (kJ/mol) | 1681 | 1251 | 1140 | 1008 |
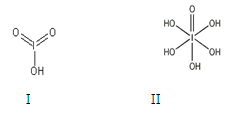
��1��P (1��)4S24P5(1��) ��2�� ���(1��)
��3����(2��) ��4����(2��)
��5��sp3 (2��) Cl2O (��OF2�Ⱥ�����) (2��)
��6��AD(2��)
��7��a3��NA/��(2��)
���������������±��Ԫ��λ�����ڱ���17��(P��)����(����Ԫ��)ԭ�ӵļ۵����Ų�Ϊ4S24P5��
���������Ӽ�ͨ������ϳ�(HF)2��
�����ڵ�ĵ�һ��������±��ԭ������Խ�С�����п������ɽ��ȶ��ĵ��������ӡ�
��HIO3�ķ��ǻ���ԭ������2���࣬���Խ�H5IO6 [���ǻ���ԭ����Ϊ1]ǿ��
������ClO2�� ��������ԭ����Χ���ĶԼ۲���ӣ���������ԭ�ӵ��ӻ��������Ϊsp3�����ݡ��������ڡ�ͬ�廥������ԭ���ȷ��ClO2�� �ĵȵ�����ΪCl2O��OF2��
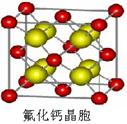
�ʵ⾧�徧���ṹ������������������ÿ�������к���8�� ��6��
��6��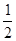 ��4������ӣ�����ԭ����Ϊ8��B����⾧�����ڷ��Ӿ��壬C�����
��4������ӣ�����ԭ����Ϊ8��B����⾧�����ڷ��Ӿ��壬C�����
����CaF2�������Ca2����8�� ��6��
��6��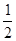 ��4��������F����8�����൱����4��CaF2����ѣ�
��4��������F����8�����൱����4��CaF2����ѣ�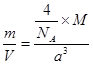 �������M��)a3��NA/����
�������M��)a3��NA/����
���㣺���⿼�����ʽṹ������(Ԫ�صķ������۵����Ų�ʽ���������һ�����ܡ�����������Դ�С����������ؼ������ж�)��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
Ԫ�����ڱ���Ԫ����������ѧϰ���о�������ʵ�����к���Ҫ�����á��±��г��ˢ١������Ԫ�������ڱ��е�λ�á�
| �� ���� | ��A | ��A | ��A | ��A | ��A | ��A | ��A | 0 |
| 2 | | | | �� | | �� | | |
| 3 | �� | �� | �� | | | | �� | �� |
| 4 | �� | | | | | | �� | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��12�֣���ѡ���⡿�������A��B��С�⣬��ѡ������һС�⣬������Ӧ�Ĵ�����������������������AС�����֡�
A��[���ʽṹ������]
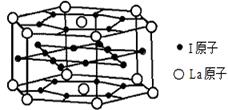
�±�ΪԪ�����ڱ���һ���֣����е���ĸ������Ӧ��Ԫ�ء�
| a | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | b | c | d | | |
| | e | | | | | | | | | | | f | | g | | | |
| | | | | | | | | h | I | | | | | | | | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
(14��)A��B��C��D��E��F���ֶ�����Ԫ�أ���ԭ������������������B��Cͬ���ڣ�D��E��Fͬ���ڣ�A��Dͬ���壬C��Fͬ���壬CԪ�ص�ԭ�������������Ǵ�����������������D����������ԭ�Ӱ뾶��������Ԫ�ء���֪����Ԫ�����γɵij��������ڳ��³�ѹ�������������壬�����ǹ��塣
��ش��������⡣
(1)Ԫ��D�����ڱ��е�λ�� ��
(2)C��D��F����Ԫ���γɵļ����ӵİ뾶�ɴ�С��˳����(�����ӷ��ű�ʾ) ��
(3)��E�Ƿǽ���Ԫ�أ��䵥���ڵ��ӹ�ҵ������ҪӦ�ã���д��������������ǿ����Һ�����ӷ���ʽ�� ��
(4)��E�ǽ���Ԫ�أ��䵥������������Ӧ�����ں��Ӹֹ죬��д����Ӧ�Ļ�ѧ����ʽ�� ��ұ������Eʱ����ʯī���缫��E��(�����������) ���õ����������У�����ʯī��Ҫ���ϲ��䣬��ϵ缫��Ӧ˵����ԭ���� ��
(5)��A��B��C����Ԫ����ԭ�Ӹ�����4��2��3�γɻ�����X��X��������ѧ�������� �������к���X�е�������������ϸ���Ĵ������±���������Ϊ�������ӣ�д�������ӷ���ʽ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��14�֣�A��B��C��D 4��Ԫ�أ�AԪ��������������������������ԭ����������ȣ�B��ԭ�Ӱ뾶����������������С�ģ�B������������Ӧˮ����Ļ�ѧʽΪHBO3��CԪ��ԭ�ӵ������������ȴ������2����C����������D�������Ӿ�����ͬ�ĵ����Ų�����Ԫ�ؿ��γɻ�����D2C��
��1��BԪ�ص�����___ _____��B�����ڱ��е�λ�õ� ______���ڣ���________�壻
��2��A��B�γɵĻ�����ĵ���ʽ____ ____��
��3��C��Ԫ�ط���________��C�����������Ļ�ѧʽ____ ____��
��4�� D������������Ӧ��ˮ����Ļ�ѧʽ____ ____��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
(15��)
A��B��D��E��FΪ������Ԫ�أ��ǽ���Ԫ��A����������������������ͬ��B����������������������������2����B ��D�г��ȼ������������ۻ�����BD2��E����D2��������ͬ�ĵ�������A��F��ȼ�գ���������ˮ�õ�һ��ǿ�ᡣ�ش��������⣺
��1��A�����ڱ��е�λ���� ��д��һ�ֹ�ҵ�Ʊ�����F�����ӷ���ʽ ��
��2��B��D��E��ɵ�һ�����У�E����������Ϊ43%��������Ϊ ����ˮ��Һ��F���ʷ�Ӧ�Ļ�ѧ����ʽΪ ���ڲ����м�������KI����Ӧ�����CC14������ ������ ɫ��
��3������ЩԪ����ɵ����ʣ�����ɺͽṹ��Ϣ���±���
| ���� | ��ɺͽṹ��Ϣ |
| a | ����A�Ķ�Ԫ���ӻ����� |
| b | ���зǼ��Թ��ۼ��Ķ�Ԫ���ӻ������ԭ����֮��Ϊ1:1 |
| c | ��ѧ���ΪBDF2 |
| d | ֻ����һ�������������ҿɵ���ĵ��ʾ��� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
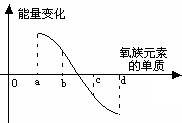
(9��)Ԫ����������ָ������ѧϰԪ�ؼ��仯����֪ʶ����Ҫ���ߡ���֪����Ԫ�أ�����Po���IJ���֪ʶ���±���ʾ��
| Ԫ�� | 8O | 16S | 34Se | 52Te |
| �����۵㣨�棩 | -218.4 | 113 | | 450 |
| ���ʷе㣨�棩 | -183 | 444.6 | 685 | 1390 |
| Ԫ����Ҫ���ϼ� | -2 | -2,+4,+6 | -2,+4,+6 | |
| ԭ�Ӱ뾶 | ������ | |||
| ������H2��Ӧ��� | ��ȼʱ���� | ���Ȼ��� | �����ѻ��� | ����ֱ�ӻ��� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��16�֣��ȵ���ԭ���Ļ����۵��ǣ�ԭ������ͬ�Ҽ۵���������ȵķ��ӻ����Ӿ�����ͬ�Ļ�ѧ�����ͺͿռ乹�ͣ�����Ϊ�ȵ����塣�ȵ�����Ľṹ���ƣ���������������磺N2��CO��C22����CN��Ϊ�ȵ����塣
��1����֪CaC2Ϊ���ӻ������CaC2�ĵ���ʽΪ ��
��2���۱�ϩ���׳�������ë���ɱ�ϩ�����CH2=CH��CN���ۺϷ�Ӧ���ɣ���CH2=CH��CN��Cԭ�ӵ��ӻ���ʽΪ �������ЦҼ��ͦм���֮��Ϊ ��
��3��CO������ɽ���ԭ��M�γ������M(CO)n ��������������ԭ�Ӽ۵���������λ���ṩ��������֮��Ϊ18����MΪFe����n= ��
��4��CO��N2�Ľṹ���ƣ������к��й����������ɱ�ʾΪC��O ���±������ߵļ������ݣ���λ��kJ��mol��1��
| | C��O | C=O | C��O |
| CO | 357.7 | 798.9 | 1071.9 |
| | N��N | N=N | N��N |
| N2 | 154.8 | 418.4 | 941.7 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
(12��)����ѧ�������ʽṹ�����ʡ�
��1�����ɽ���Ԫ�������γɶ��������磺[Fe(H2NCONH2)6](NO3)3[�����������غ���(��)��Fe(CO)x�ȡ�
�ٻ�̬Fe3+��M������Ų�ʽΪ ��
������(H2NCONH2)������Cԭ�ӵ��ӻ���ʽ�� ��
�������Fe(CO)x������ԭ�Ӽ۵������������ṩ������֮��Ϊ18����x= ��
Fe(CO)x�����³�Һ̬���۵�Ϊ-20.5�棬�е�Ϊ103�棬�����ڷǼ����ܼ����ݴ˿��ж�Fe(CO)x�������� (�������)��
��2������˵����ȷ���� (����ĸ���)��
| A����һ�����ܴ�С��S>P>Si |
| B���縺��˳��C<N<O<F |
| C����Ϊ������CaO��KCl�ߣ�����KCl���۵��CaO�۵�� |
| D�����Ӿ����У����ۼ�����Խ�÷��Ӿ�����۷е�Խ�� |
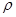 g��cm-3,�����ӵ�����ΪNA�����߳�a= cm��(�ú�
g��cm-3,�����ӵ�����ΪNA�����߳�a= cm��(�ú�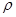 ��NA�ļ���ʽ��ʾ)
��NA�ļ���ʽ��ʾ)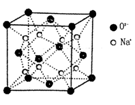
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com