
ijͭ��ʯ��Ҫ��Cu2(OH)2CO3����������Fe��Si�Ļ����ʵ�����Դ�ͭ��ʯΪԭ���Ʊ�CuSO4��5H2O ��CaCO3�����ֲ������£�
��ش��������⣺
��1����ҺA������Cu2+�⣬�����ܺ��еĽ���������________(�����ӷ���)����֤�����������õ��Լ���______________��
��2���������ɵ�CO2��ȡ����̼��ơ��Ʊ�ʱ�������Ȼ�����Һ��ͨ�백������ͨ��CO2��
��ʵ����ͨ�����ü����Ȼ�狀��������ƻ����ķ�����ȡ������ijѧϰС��ѡȡ��ͼ��������װ����ȡ���ռ������İ�����
����������������Ӹ������ӿڣ�����Ϊ��ȷ��˳��Ϊa��______��______��______��______�� i��������i����©����������______________��
��ʵ�����л����ù����������ƺ�Ũ��ˮ��ȡ�����������������ʺ���ɸ�ʵ��ļ���װ����_________(����)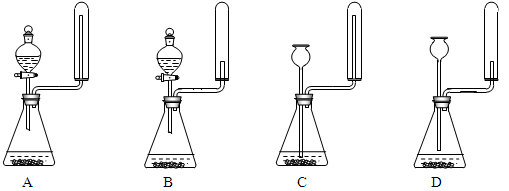
��3���ⶨͭ��ʯ��Cu2(OH)2CO3�����ٷֺ����ķ����ǣ�a����1.25gͭ��ʯ��ȡ��CuSO4��5H2O����ƿ�У���������ˮ��ȫ�ܽ⣻b������Һ�м���100mL0.25mol/L������������ҺʹCu2+��ȫ������c�����ˣ�d����Һ�е�����������Һ��0.5mol/L����ζ����յ㣬����10mL���ᡣ��ͭ��ʯ��Cu2(OH)2CO3��������Ϊ_____________��
��11�֣���1��Fe2+��Fe3+�����Ը��������Һ��KSCN��Һ����4�֣���1�֣�����������Ҳ�÷֣�
��2����a��g��h��e��d�� i ��2�֣�����ֹ������1�֣�����A��2�֣� ��3��88.8%��2�֣�
���������������1��Cu2(OH)2CO3�Լ�Fe��Si�Ļ�������ϡ���ᷴӦ��������ͭ������������������������������ϡ�����Ӧ��������Һ��A�г�����Cu2+�⣬�����ܺ��еĽ���������Fe2+��Fe3+���������Ӿ��л�ԭ�ԣ���ʹ���Ը��������Һ��ɫ����˿��Լ����������ӡ����������ӿ�����KSCN��Һ���顣
��2���ٸ���װ��ͼ��֪��Aװ�����Ʊ������ġ��������ɵİ����к���ˮ������������Ҫ���ѡ�ü�ʯ�Ҹ���������ܶ�С�ڿ����ģ��Ұ�����������ˮ������Ӧ���������ſ������ռ�������Ҫ������İ����������գ������ȷ�IJ���˳����a��g��h��e��d�� i��������������ˮ�������i������©���������Ƿ�ֹ������
���ù����������ƺ�Ũ��ˮ��ȡ���������������Ҫ��Һ©������Ӧ����Ҫ���ȣ��Ұ����������ſ������ռ���������ȷ�Ĵ�ѡA��
��3��������������ʵ�����0.01L��0.5mol/L��0.005mol������ݷ���ʽNaOH��HCl��NaCl��H2O��֪�������ᷴӦ������������0.005mol���������Ƶ����ʵ�����0.1L��0.25mol/L��0.025mol����������ͭ��Ӧ������������0.025mol��0.005mol��0.020mol������ݷ���ʽ2NaOH��CuSO4��Cu(OH)2����Na2SO4��֪������ͭ�����ʵ�����0.020mol��2��0.010mol�����Ը���ԭ���غ��֪��ͭ��ʯ��Cu2(OH)2CO3�����ʵ�����0.010mol��2��0.005mol������Cu2(OH)2CO3��������Ϊ ��100%��88.8%��
��100%��88.8%��
���㣺�������Ӽ��顢�����Ʊ������ʺ����IJⶨ�Լ�ʵ�鷽������������۵�



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
���������Ҫ�ɷֿɱ�ʾΪFeO��Cr2O3��������MgO��Al2O3��Fe2O3�����ʣ��������Ը�����Ϊԭ���Ʊ��ظ���أ�K2Cr2O7��������ͼ��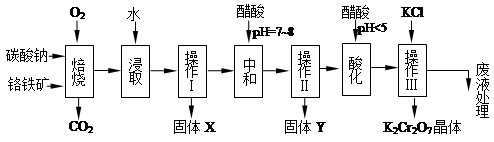
��֪����4FeO��Cr2O3+ 8Na2CO3+ 7O2 8Na2CrO4 + 2 Fe2O3 + 8CO2����
8Na2CrO4 + 2 Fe2O3 + 8CO2����
��Na2CO3 + Al2O3 2NaAlO2 + CO2������ Cr2O72��+ H2O
2NaAlO2 + CO2������ Cr2O72��+ H2O 2CrO42�� + 2H+
2CrO42�� + 2H+
��������ش��������⣺
��1������X����Ҫ����_________����д��ѧʽ����Ҫ����ữ��������Һ��pH�Ƿ����4.5��Ӧ��ʹ��__________����д�������Լ����ƣ���
��2���ữ�����ô��������ҺpH<5����Ŀ����_________________________________��
��3���������жಽ��ɣ����K2Cr2O7����IJ��������ǣ�����KCl���塢����Ũ���� �����ˡ�_______�����
��4���±���������ʵ��ܽ�����ݣ�����������Ӧ�Ļ�ѧ����ʽ�ǣ�Na2Cr2O7+2KCl ��K2Cr2O7��+2NaCl���÷�Ӧ����Һ���ܷ�����������_______________��
| ���� | �ܽ��/(g/100gˮ) | ||
| 0��C | 40��C | 80��C | |
| KCl | 28 | 40.1 | 51.3 |
| NaCl | 35.7 | 36.4 | 38 |
| K2Cr2O7 | 4.7 | 26.3 | 73 |
| Na2Cr2O7 | 163 | 215 | 376 |

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��þ����һ�ֹ�ҵ���ϣ���Ҫ�ɷ���MgO(ռ40%)������CaO��MnO��Fe2O3��FeO��Al2O3��SiO2�����ʣ��Դ�Ϊԭ����ȡ������þ��������ӡȾ����ֽ��ҽҩ�ȹ�ҵ������þ������ȡMgSO4��7H2O�Ĺ����������£�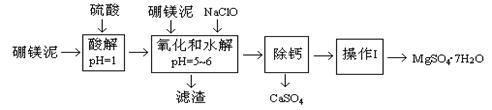
��֪��NaClO��Mn2+��Ӧ����MnO2������
| ������ | Fe(OH)3 | Al(OH)3 | Fe(OH)2 |
| ��ʼ����pH | 2.3 | 4.0 | 7.6 |
| ��ȫ����pH | 4.1 | 5.2 | 9.6 |
| �¶ȣ��棩 | 40 | 50 | 60 | 70 |
| MgSO4 | 30.9 | 33.4 | 35.6 | 36.9 |
| CaSO4 | 0.210 | 0.207 | 0.201 | 0.193 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
���ˮ������Ӽ���CoCl2-6H2O���������Ժ��ܷ��ϣ�������Fe��Al��������ȡCoCl2��6H2O��һ���¹�����������ͼ��
��֪�����������ᷴӦ�Ļ�ѧ����ʽΪ��Co+2HCl CoCl2+H2��
��CoCl2��6H2O�ܵ�86oC��������ˮ�����ѵȣ��������ȶ�����������110oC��120OCʱ��ʧȥ�ᾧˮ����ж�����ˮ�Ȼ��ܡ�
�۲���������������������ʽ����ʱ��Һ��pH���±���
��ش��������⣺
��1�����������е��Լ�a����Ϊ ���������� ��
��2������̼���Ƶ���pH��b��b�ķ�Χ�� ��
| ������ | Fe��OH��3 | Fe��OH��2 | Co��OH��2 | Al��OH��3 |
| ��ʼ������pH�� | 2.3 | 7.5 | 7.6 | 3.4 |
| ��ȫ������pH�� | 4.1 | 9.7 | 9.2 | 5.2 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��пʪ��ұ�������У����������ͭ��������Ҫ����Zn��Cd��Fe��Cu�ȣ���ֱ�Ӷ�������ɻ�����Ⱦ��Ҳ����Դ�˷ѣ�����Ϊ���մ���ͭ�����Ʊ�����п����Ĺ�ҵ���̣�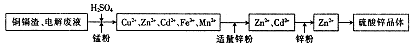
��֪������п����������ˮ�������ھƾ���
��1�����������Ŀ�� ��
��2��������ܳ�ͭ�������̷���Ϊ�˽�������ӵı�������ݵ��ӣ���ͭ�ܳ����̷۵���Ҫ�ɷ�ΪMnO2����ط���ʽΪ2Fe3++Cu=2Fe2++Cu2+�� ��
��3��������п��Һ�������п���壬��Ҫ�IJ�������Ϊ ��
��4���Ƶõ�����п������Ҫϴ�ӣ�ѡ����Լ�Ϊ ��ԭ���� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��ʴ���
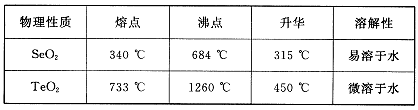
��14�֣���34Se����52Te���ǵ�VIA��Ԫ�أ����Ƿֲ��ڵؿ��е�ϡ��Ԫ�ء���ҵ�������ķ��ϣ���Ҫ�ɷ������ڡ�̼��ͭ�����Ͻ𣩻��վ��������������£�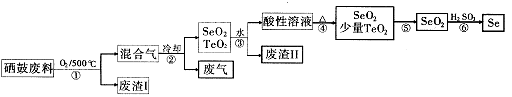
��֪��
��1��Se���⻯��ĵ���ʽ��____��
��2���������ͨ�˵�����ʹ���ķ��Ϸ��ڣ�Ŀ���� ��
��3����������Ҫ�ɷ���____ ������II����Ҫ�ɷ��� ��
��4�����������Ҫ��Ӧ�Ļ�ѧ����ʽ�� �������Ӧ�Ļ�ѧ����ʽ�� ��
��5�����ݱ������ݣ�����������˵ķ��뷽���� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�������
���������Ҫ�ɷֿɱ�ʾΪFeO��Cr2O3��������MgO��Al2O3��Fe2O3�����ʣ��������Ը�����Ϊԭ���Ʊ��ظ���أ�K2Cr2O7��������ͼ��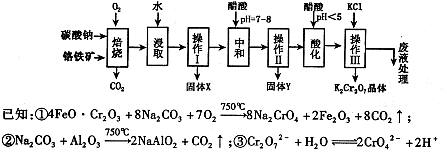
��������ش��������⣺
��1������X����Ҫ���� ����д��ѧʽ����Ҫ����ữ��������Һ��pH�Ƿ����4��5��Ӧ��ʹ�� ����д�������Լ����ƣ���
��2���ữ�����ô��������ҺpH <5����Ŀ���� ��
��3���������жಽ��ɣ����K2Cr2O7����IJ��������ǣ�����KCl���塢����Ũ����
�����ˡ� �����
��4���±���������ʵ��ܽ�����ݣ�����III������Ӧ�Ļ�ѧ����ʽ��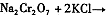
 ���÷�Ӧ����Һ���ܷ����������ǣ� ��
���÷�Ӧ����Һ���ܷ����������ǣ� ��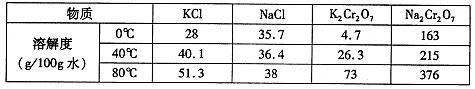
��5������ƷY��Ҫ��������������������þ���������ܻ����P���������ʣ���ȷ����Y���������������ķ����dz�ȡng��Ʒ��������� ����д�Լ������ܽ⡢���ˡ��ټ������ ����д�Լ������������ա���ȴ���������ø������mg��������Ʒ��������������������Ϊ ���ú�m��n�Ĵ���ʽ��ʾ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��12�֣�����ͭ��������������Ӧ�ù㷺��ij�������ú��������ķ�ͭ��Ϊԭ�������������������£�
��1��д������ʱͭ��ϡ���ᡢϡ���ᷴӦ��������ͭ�Ļ�ѧ����ʽ�� ��
��2��ȡ��������Ϊ��ȷ��Fe3+�Ƿ��������ļ��鷽���� ��
��3������c�� ��
��4������a���Ա�ѭ�����ã��û�ѧ����ʽ��ʾ����a��ѭ�����õ�ԭ��Ϊ��
2NO+O2 =2NO2�� ��
��5��һ���¶��£�����ͭ���ȷֽ�����CuO��SO2���塢SO3�����O2���壬д������ͭ���ȷֽ�Ļ�ѧ����ʽ�� ��
ijͬѧ���������ͼ��ʾ��ʵ��װ�÷ֱ�ⶨ���ɵ�SO2���塢SO3�����������O2����������������в�����֮������˵�����ɣ� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
������ⷨ���Ѱ۲����ķ�Һ[���д���FeSO4��H2SO4������Fe2(SO4)3TiOSO4]���������������Ͳ�Ѫ�������������������������£�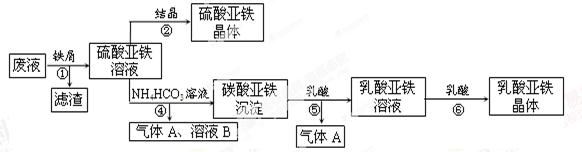
��ش�
��1��������з��������������______������ޱ������һ������նȣ�ԭ����_________��
��2����Һ�е�TiOSO4�ڲ������ˮ��������������Ҫ�ɷ�ΪTiO2?xH2O���Ļ�ѧ����ʽΪ__________������ܵ����ӷ���ʽΪ_____________________��
��3����ƽ���ƶ�ԭ�����Ͳ�����м������ܵõ�����������ԭ��_______��
��4����ƽ���Ը��������Һ������������Һ��Ӧ�����ӷ���ʽ��_____Fe2++_____MnO4��+_____H+=_____Fe3++_____Mn2++_____
ȡ��������þ�����Ʒag,����ϡ�������100.00 mL��Һ��ȡ��20. 00 mL��Һ,��KMnO4��Һ�ζ���������KMnO4����Ӧ����������0.1000 mol��L��1KMnO4��Һ20.00mL�������þ�����FeSO4?7H2O����������Ϊ���Ժ�a��ʽ�ӱ�ʾ��_____________________��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com