
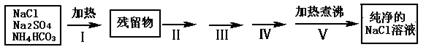
| �������� | ʵ������ | ʵ��Ŀ�� |
| ����II�����������ܽ�õ���Һ�� | | |
| ����III�� |  | |
| ����IV�����ˣ�����Һ�� | | |
| ����V������Һ������� |  | |
| �������� | ʵ������ | ʵ��Ŀ�� |
| ����II������Һ�еμӹ�����BaCl2��Һ | ���ɰ�ɫ���� | ��ȥSO42������ |
| ����III��������Һ�У�����˺�����Һ�У��μӹ�����Na2CO3��Һ |  | ��ȥ������Ba2+���� |
| ����IV���μ����������� | ������������ | ��ȥ������CO32������ |
  | | �����ܽ�����Һ�е�CO2��HCl���壨2�֣� |
 NH3��+H2O��+CO2����ǿ�����������Ӧ�����Ρ�������ˮ����ȥ�����ʵ�ͬʱ�������������ʣ��������Լ��������������ࣻ��2����ȥ̼����狀��Ƚ����ð�ɫ�����ܽ⣬������Һ�м������BaCl2��Һ��ʹ��������ȫ��Ӧ�����ɰ�ɫ�����ᱵ������NaCl����ȥ��Һ�е���������ӣ���������ı��������µ����ʣ�Ϊ�˳�ȥ�����ӣ�Ӧ������Һ�����������Һ�м������̼������Һ��ʹ�Ȼ�����ȫ��̼���Ʒ�Ӧ�����ɰ�ɫ��̼�ᱵ�������Ȼ��ƣ���ȥ��Һ�еı����ӣ����������̼����������µ����ʣ����ˣ���������ᱵ��̼�ᱵ����֮��Ϊ�˳�ȥ��Һ�е�̼������ӣ�Ӧ�����м����������ᣬ̼���������ᷴӦ�������Ȼ��ơ�������̼�����ˮ����ȥ��Һ�е�̼������ӣ�����������������µ����ʣ������ˮ�ķе�ϵͣ��Ȼ��Ƶķе�ߣ�����Һ��У����Գ�ȥ��Һ�е��Ȼ��⡢������̼��ˮ�������ɵô������Ȼ��ƹ��壻��3�������⣬36.0gNaCl����100gˮ�У���������20��ı���NaCl��Һ��������Һ���������������������ܼ�����֮�ͣ���36.0gNaCl����100gˮ�γ�136.0g����NaCl��Һ��������Һ���������Һ��������Һ�ܶȵı�ֵ����NaCl��Һ�����Ϊ
NH3��+H2O��+CO2����ǿ�����������Ӧ�����Ρ�������ˮ����ȥ�����ʵ�ͬʱ�������������ʣ��������Լ��������������ࣻ��2����ȥ̼����狀��Ƚ����ð�ɫ�����ܽ⣬������Һ�м������BaCl2��Һ��ʹ��������ȫ��Ӧ�����ɰ�ɫ�����ᱵ������NaCl����ȥ��Һ�е���������ӣ���������ı��������µ����ʣ�Ϊ�˳�ȥ�����ӣ�Ӧ������Һ�����������Һ�м������̼������Һ��ʹ�Ȼ�����ȫ��̼���Ʒ�Ӧ�����ɰ�ɫ��̼�ᱵ�������Ȼ��ƣ���ȥ��Һ�еı����ӣ����������̼����������µ����ʣ����ˣ���������ᱵ��̼�ᱵ����֮��Ϊ�˳�ȥ��Һ�е�̼������ӣ�Ӧ�����м����������ᣬ̼���������ᷴӦ�������Ȼ��ơ�������̼�����ˮ����ȥ��Һ�е�̼������ӣ�����������������µ����ʣ������ˮ�ķе�ϵͣ��Ȼ��Ƶķе�ߣ�����Һ��У����Գ�ȥ��Һ�е��Ȼ��⡢������̼��ˮ�������ɵô������Ȼ��ƹ��壻��3�������⣬36.0gNaCl����100gˮ�У���������20��ı���NaCl��Һ��������Һ���������������������ܼ�����֮�ͣ���36.0gNaCl����100gˮ�γ�136.0g����NaCl��Һ��������Һ���������Һ��������Һ�ܶȵı�ֵ����NaCl��Һ�����Ϊ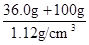 =
=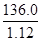 cm3=
cm3=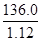 mL�����ʵ����ʵ�������������Ħ�������ı�ֵ����136.0g����NaCl��Һ��n(NaCl)=
mL�����ʵ����ʵ�������������Ħ�������ı�ֵ����136.0g����NaCl��Һ��n(NaCl)=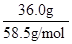 =
=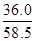 mol������c=n/V����20��ı���NaCl��Һ�����ʵ���Ũ��Ϊ
mol������c=n/V����20��ı���NaCl��Һ�����ʵ���Ũ��Ϊ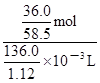 =
=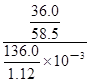 mol/L��5.07mol/L��
mol/L��5.07mol/L��


| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A�����к͵ζ���ʵ���У�����ƿ����ƿ������ˮϴ����ʹ�ã��ζ��ܺ���Һ��������ˮϴ������������ϴ��ʹ�� |
| B����ȥ����CO2�л��е�����SO2���ɽ������������ͨ��ʢ������KMnO4��Һ��Ũ�����ϴ��ƿ |
| C��������þ��Һ�м����������ƿ��Եõ�������þ���� |
| D����Mg(OH)2����ת���������У�������ϡ���ᣬ�������ɵ���ˮMgCl2���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�������
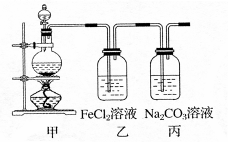
| ʵ�鲽�� | Ԥ������ͽ��� | |
| �� | ��A�Թ�ȡ��������ʯ��ˮ���ã���B�Թ�ȡ������Ʒ������B�Թ� | ��������ų��ҳ���ʯ��ˮδ�����ǣ������һ������ |
| �� | | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A����������ϡ���ᷴӦ����ȡ��������������ˮ���ռ� |
| B�����������и�ʮˮ̼���Ƽ�����ˮ |
| C������ʽ�ζ�����ȡ6.55mL��KMnO4��Һ |
| D���÷�Һ©������������������������Һ������Ӧ��������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A������ˮ�Ҵ���ȡ��ˮ�еĵⵥ�� |
| B������ϡHNO3��BaCl2��Һ������ɫ��������Һ��һ����SO42�� |
| C������FeCl3��Һһ���Լ���CCl4��������Һ��NaI��Һ��NaCl��Һ |
| D��������������0.1mol/L NaCl��Һ������ᴿ��ʵ���������������ͬ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| | ʵ��Ŀ�� | ʵ�鷽�� |
| A | �о������Թ�������ֽ����ʵ�Ӱ�� | �ֱ�����֧�Թ��м�����������Ũ�ȵĹ���������Һ����������һֻ�Թ��м�������MnO2 |
| B | ֤��Mg(OH)2��������ת��ΪFe(OH)3���� | ��2mol/LNaOH��Һ���ȼ���3��1mol/L MgCl2��Һ���ټ���3��1mol/L FeCl3��Һ |
| C | ���Լ�����������Һ | ��Na2CO3��Һ��HCl��Һ��μ� |
| D | �ⶨ�������������ĺ��� | ȡa g����������ϡ�����ַ�Ӧ�����ݳ�������ͨ����ʯ�Һ������ΪbL����״���£� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
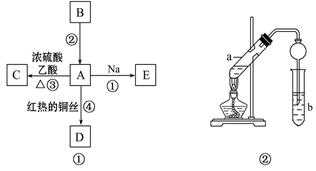
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�ʵ����
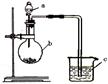
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com