
ʵ��������CuSO4��5H2O������480mL0.1mol/L��CuSO4��Һ����ش��������⣺
��1��Ӧ��������ƽ��ȡCuSO4��5H2O����_______g��
��2�����ڳ�����Ʒʱ��ҩƷ������ƽ�����ϣ����������ƽ�����ϣ�1g����ʹ�����룩����ƽƽ��ʱ��ʵ�ʳ�����CuSO4��5H2O������__________g��
��3����ʵ���õ�����Ҫ�����У�������ƽ����Ͳ���ձ�����������________��________��
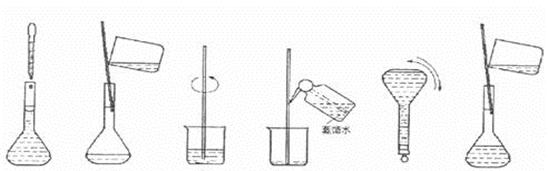
��4�����ƹ������м����ؼ��IJ���Ͳ�������ͼ��ʾ������Щʵ�鲽��A��F��ʵ������Ⱥ������
�� ��
���ж��ݵľ�������� ��
��5�����������ʹ������Һ��Ũ�Ȳ�������Ӱ�죨A.ƫ�� B.ƫ�� C.����,����š�����
���ܽ⾧���õ��ձ��Ͳ�����δϴ�ӣ�____________��
�ڶ���ʱ���ӿ̶��ߣ�____________��
������CuSO4��5H2O������ʧȥ���ֽᾧˮ��____________��
��1��12.5 ��2��11.5 ��3��500ml����ƿ ��ͷ�ι�
��4��CBDFAE ���ж��ݵľ��������:������ƿ�м�������ˮ����Һ����̶���1��2cm��ʱ�����ý�ͷ�ιܼ�����ˮ����Һ����͵���̶������� ��5��B��A��A
���������������1��������Һ�����Ϊ480ml��������ƿ�Ĺ��û��480ml��ֻ��ѡ��500ml����ƿ�������ҪCuSO4�����ʵ���n��cV��0.5L��0.1mol?L-1��0.05mol��CuSO4?5H2O�����ʵ�������CuSO4�����ʵ���������CuSO4?5H2O��������0.05mol��250g/mol��12.5g��
��2����ҩƷ������ŷ���������ƽƽ��ԭ��m(����)��m(����)+�����������ʵ�ʳ�������ͭ���������Ϊ12g-0.5g��11.5g��
��3����Һ����һ�㲽���ǣ�������������ܽ⡢��ȴ����Һ��ϴ�ӡ����ݡ�ҡ�ȡ�װƿ��ǩ��һ������ƽ�������õ�ҩ�ף����壬���ձ����ܽ⣬���ò��������裬��ȴ��ת�Ƶ�500mL����ƿ�У����ò�����������ת����ϣ�����������ˮϴ���ձ���������2��3�β���ϴ��Һȫ��ת�Ƶ�����ƿ�У��ټ���������ˮ������ˮ��Һ�����̶���1��2cmʱ�����ý�ͷ�ιܵμӣ�ʹ��Һ�İ�Һ�����͵��������ƽ������ƿ�����������µߵ�ҡ�ȡ�������Ҫ������Ϊ��������ƽ��ҩ�ס��ձ�����������500mL����ƿ����ͷ�ιܣ���˻���Ҫ������Ϊ��500mL����ƿ��ҩ�ס���ͷ�ιܡ�
��4���ɣ�3������Һ���Ƶ�һ�㲽���֪����ȷ�IJ���˳��Ϊ��CBDFAE�����ж��ݵľ�������ǣ�������ƿ�м�������ˮ����Һ����̶���1��2cm��ʱ�����ý�ͷ�ιܼ�����ˮ����Һ����͵���̶������С�
��5�����ܽ⾧���õ��ձ��Ͳ�����δϴ�ӣ���������ͭմ���ձ����벣�����ϣ���������ƿ������ͭ�����ʵ������٣�������Һ��Ũ��ƫ�ͣ��ʴ�Ϊ��B��
�ڶ���ʱ���ӿ̶��ߣ�Һ���ڿ̶����·�����Һ�����ƫС��������Һ��Ũ��ƫ�ߣ��ʴ�Ϊ��A��
������CuSO4?5H2O������ʧȥ���ֽᾧˮ������ͭ��������������ʵ�ʳ���������ͭ����������ͭ������ƫ��������Һ��Ũ��ƫ����ѡA��
���㣺��������ͭ��Һ�����ơ�����ѡ��������Ȼ���ʵ������Լ���������


 �ŵ������ϵ�д�
�ŵ������ϵ�д� 53������ϵ�д�
53������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�������
��֪һ������������������ȼ�գ����û������100 mL 3.00 mol/L��NaOH��Һ���ܶ�Ϊ1.12 g/mL��ǡ����ȫ���գ������Һ�к���NaClO�����ʵ���Ϊ0.0500 mol��
��1��ԭNaOH��Һ����������Ϊ_______
��2��������Һ��Cl�������ʵ���Ϊ________mol
��3�����������Ͳμӷ�Ӧ�����������ʵ���֮��n(Cl2)��n(H2)��______________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�������
���ⶨ�����е�SO2��������������ʵ�飺ȡ100 L�ÿ���(��״��)��ͨ��ʢ��100 mL��������(H2O2)ˮ��Һ������ƿ��ʹ�����е�SO2��ȫ���գ��������ᡣ�ڷ�Ӧ���ˮ��Һ�м�������BaCl2��Һ�����ɰ�ɫ���������ⶨ������Ϊ11.65g�������������ɷֶԲⶨ�����Ӱ�죩��
(1)д��SO2��H2O2��Ӧ�����ӷ���ʽ��
(2)д��H2SO4��BaCl2��Ӧ�����ӷ���ʽ��
(3)��100 L������SO2�����������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ʵ����Ҫ��98%���ܶ�Ϊ1��84g��cm-3������������3��68mol/L��������Һ480mL��
��1����ȷ��ȡ98%������ mL��
��2��Ҫ����3��68mol/L��������Һ480mL�������õ����������ձ�������������Ͳ
��3��������3��68mol/L��������Һ��������������ȷ�����������д����������ʹ�����Ƶ�������ҺŨ��ƫ�͵��� ��
| A����ϡ�͵�������Һת��������ƿ��δϴ���ձ��Ͳ����� |
| B�����ձ��ڵ�ϡ����������ƿ��ת��ʱ�����������ʹ����ϡ���ὦ��ƿ�� |
| C���ý�ͷ�ι�������ƿ�м�ˮʱ��Һ���������ƿ�̶ȣ���ʱ�����õιܽ�ƿ��Һ��������ʹ��Һ��Һ����̶����� |
| D���ý�ͷ�ι�������ƿ�м�ˮʱ�����ӹ۲���Һ��Һ��������ƿ�̶����� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ijһ��θҩ�е������Ϊ̼���,��������������������IJⶨ����:
��������0.1 mol��L-1��HCl��Һ��0.1 mol��L-1��NaOH��Һ
��ȡһ��(ҩƬ������ͬ)0.2 g�Ĵ�θҩƬ,ĥ������20.0 mL����ˮ
���Է�̪Ϊָʾ��,��0.1 mol��L-1��NaOH��Һ�ζ�,��ȥV mL��ζ��յ�
�ܼ���25 mL 0.1 mol��L-1��HCl��Һ
(1)д��ʵ����̵IJ���(д���˳��)������������������������
(2)��ͼ��ʾ����������0.1 mol��L-1��HCl��Һ��0.1 mol��L-1��NaOH��Һ�϶�����Ҫ��������(�����)������,����������Һ����Ҫ�IJ���������������(����������)�� 
(3)����������ҺӦѡ�õ�����ƿ�����(����ĸ)������������
| A��50 mL��50 mL |
| B��100 mL��100 mL |
| C��100 mL��150 mL |
| D��250 mL��250 mL |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��ͼ�������Լ�ƿ��ǩ�ϵ����ݡ�
| �����ѧ��(CP)(500mL) Ʒ�������� ��ѧʽ��H2SO4 ��Է���������98 �ܶȣ�1.84g��cm-3 ����������98% |
|
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
�¹�������ҵ��־��Ӧ�û�ѧ���Ͽ������½��ܣ�ij�е������о�С�����������һ�֡�ˮƿ�����ø���ϩ(C60)��������������ƿ�塱��һ������������ƿ�ǡ���ǡ�ÿɽ�һ��ˮ���ӹ������档����˵����ȷ���� ( )
| A��ˮ��˫��ˮ��ˮ�������Ǵ����� |
| B��ʯī��C60����Ϊͬλ�� |
| C��������ǿ�����ǿ����� |
| D��һ��������ʯīת��ΪC60�ǻ�ѧ�仯 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
���з���������ȷ����
��Һ�ȡ���ˮ���ɱ����⻯����Ϊ������
��CaCl2��NaOH��HCl��IBr��������
��������ˮ�����ռ�����Ϊǿ�����
��C60��C70�����ʯ��ʯī��Ϊ̼��ͬ��������
�ݵ�ơ����ۡ�ˮ�������ײ��Ͼ�Ϊ����
| A���٢ۢܡ����� | B���ڢۡ��� | C���ڢܡ��� | D���ڢۢܢ� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
���෨�ڻ�ѧѧ�Ƶķ�չ������Ҫ���á����з������������
| A�����ݷ�ɢϵ���ȶ��Դ�С�����Ϊ��Һ���������Һ |
| B�����ݷ�Ӧ����ЧӦ����ѧ��Ӧ��Ϊ���ȷ�Ӧ�����ȷ�Ӧ |
| C������������к��е�Hԭ�Ӹ��������ΪһԪ�ᡢ��Ԫ��Ͷ�Ԫ�� |
| D������Ԫ��ԭ�������������Ķ��ٽ�Ԫ�ط�Ϊ����Ԫ�غͷǽ���Ԫ�� |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com