
A��B��C��D��Ϊ��ѧ��ѧ�г��������ʣ�����֮���ת����ϵ����ͼ�����ֲ�������ȥ����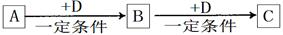
�Իش�
��1����D�Ǿ��������Եĵ��ʣ������ڶ����ڵ��������Ԫ��AΪ ����Ԫ�ط��ţ���
��2����D�ǽ������ʣ�D�ڳ�ʪ�Ŀ���������������ʴ��C��Һ�ڱ���ʱӦ�����������D��ֹ����ʣ�������D��C��Һ�ڿ����б��ʵ����ӷ���ʽΪ ����D���Ȼ����ˮ��Һ���ɲ����ղ����� ��
��3����A��B��C��Ϊ��������Ҿ����ؿ��к�����ߵĽ���Ԫ��E������Һ��A��C��Ӧ����B����д��Bת��ΪC�����п��ܵ����ӷ���ʽ ��
��4�����ڣ�1�����Ƴ���A������ڣ�3����E���ʵĻ����11.9gͶ��һ������ˮ�г�ַ�Ӧ��A��E��û��ʣ�࣬���ռ�����״���µ�����vL����������Һ����μ���Ũ��Ϊ2mol?L-1��H2SO4��Һ����100mLʱ��ɫ�����ﵽ���������v= ��
��1��Na
��2��4Fe2����O2+4H+��4Fe3��+2H2O�� Fe2 O3
��3��Al(OH)3��3H����Al3����3H2O��Al(OH)3��OH����AlO2����2H2O
��4��7.84L
���������������1�������������Եĵ���ΪO2��Cl2��AΪ�����ڽ���Ԫ�أ�����ת����ϵ����A��Na��B��Na2O��C��Na2O2��D��O2��
��2��D���ڳ�ʪ�Ŀ���������������ʴ�Ľ������ʣ�����Ԫ�أ���C�к���Fe2+����FeCl3��ˮ��Һ���ɲ����գ�����Fe3+��ˮ���HCl�Ļӷ������յõ��IJ�����Fe2O3��
��3���ؿ��к�����ߵĽ���Ԫ����������A��B��C�ת����ϵ��֪B��Al(OH)3��A��C�зֱ���Al3+��AlO2��������ȷ�Ͼ��庬���ĸ����ӣ���Bת��ΪC��������Al(OH)3ת��ΪAl3������Al(OH)3ת��ΪAlO2����
��4������100 mL 2mol?L-1��H2SO4��Һʱ���������������ʱ��Һ�н���Na2SO4����ԭ���غ��֪��n(Na)=2n(Na2SO4)=2n(H2SO4)="2" ��0.1 L��2mol?L-1="0.4" mol����ԭ����������Ƶ�����Ϊ0.4 mol��23 mol·L��1=9.2g����������Ϊ11.9g��9.2g=2.7g���ʷ�Ӧ����H2�����Ϊ(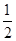 ��0.4mol+
��0.4mol+ ��
�� )��22.4L/mol="7.84" L��
)��22.4L/mol="7.84" L��
���㣺�ƵĻ�ѧ���ʣ���ѧ����ʽ���йؼ��㣻���Ļ�ѧ����


 �����Ծ���ĩ���100��ϵ�д�
�����Ծ���ĩ���100��ϵ�д� ˫��ͬ������ѵ��ϵ�д�
˫��ͬ������ѵ��ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��10�֣�A��B��C��X��Ϊ�����Ĵ��������֮��������ת����ϵ������Ʒ����ȥ����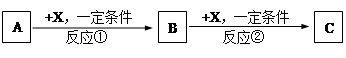
�Իش�
��1����X��ǿ�����Ե��ʣ���A�������� ��
a. S b. N2 c. Na d. Mg e. Al
��2����X�ǽ������ʣ���C��ˮ��Һ�е���AgNO3��Һ������������ϡHNO3�İ�ɫ�����������C��Һ�н���Ԫ�ؼ�̬�IJ��������ǣ� ����B�Ļ�ѧʽΪ ��
��3����A��B��CΪ��ij����Ԫ�ص��������XΪǿ����ʣ�A��Һ��C��Һ��Ӧ����B��BΪ���Ի������X�Ļ�ѧʽ����Ϊ��д����ͬ�����ʣ� �� ��
��Ӧ�ٵ����ӷ���ʽΪ��дһ���� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�����ڵ�����Ԫ��A��B��C��D��E,ԭ��������������A��B��C����Ԫ�ص��Ӳ���֮����5��A��B��Ԫ��ԭ������������֮�͵���CԪ��ԭ������������;BԪ��ԭ��������Ӳ��ϵĵ����������ĵ��Ӳ�����2��,A��D�����γ�ԭ�Ӹ����ȷֱ�Ϊ1��1��2��1������Һ̬������;E�������ھ���ˮ�ʡ�
��ش�:
(1)д��D��Ԫ�����ڱ��е�λ���� ,
E��ԭ�ӽṹʾ��ͼ��������������������
���п�����֤C��D��Ԫ��ԭ�ӵõ�������ǿ����ʵ����ʵ����������(��д���)��
A.�Ƚ�������Ԫ�ص���̬�⻯��ķе�
B.�Ƚ�ֻ��������Ԫ�����γɵĻ������еĻ��ϼ�
C.�Ƚ�������Ԫ�ص���̬�⻯����ȶ���
D.�Ƚ�������Ԫ�صĵ������������ϵ�����
(2)��A��B����Ԫ����ɵ���Ļ�����,д�������ʽ����������
(3)����A��B��C��D����Ԫ����ɵļס������ֻ�����,���ȿ��������ᷴӦ�ֿ�����NaOH��Һ��Ӧ,��Ϊ����,�仯ѧʽΪ��������,��Ϊ��Ȼ�߷��ӻ������ˮ�����,����ͬ����������Է���������С��,��ṹ��ʽΪ������������������������
(4)��̬��������ҽѧ������Ҫ����;,������Fe3O4�Ǵ������е���Ҫ����,���Ʊ����̿ɼ�ʾ����:
�ٽ�������CA3ͨ������ʵ�����FeSO4��Fe2(SO4)3�Ļ����Һ��,�������ּ�,д���÷�Ӧ���̵��ܵ����ӷ���ʽ�� ��
��������Ӧ���ɵ����ּ��������,�õ�Fe3O4��
(5)��֪�±�����:
| ���� | Fe(OH)2 | Fe(OH)3 |
| Ksp/25 �� | 2.0��10-16 | 4.0��10-36 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ij��ʯ��������FeCO3��SiO2��Al2O3�е�һ�ֻ�����ɣ�ij�о���ѧϰС����������ɷ֣�ʵ���¼����ͼ��ʾ��
��1���ÿ�ʯ�к��� (�ѧʽ)����д��ʵ����̢��з�Ӧ�����ӷ���ʽ ��
��2�����������������״̬�µ�⣬���Եõ�ij�������ʡ����������ռ�������33��6L(������ɱ�״��)ʱ�������ϵõ��ý��� g��
��3����д���ý�����������I��Ӧ�Ļ�ѧ����ʽ �����÷�Ӧ�IJ�����뵽�����ռ���Һ�У���д��������Ӧ�����ӷ���ʽ ��
��4������һ�������ÿ�ʯ�л���������SrCO3(����������ⲻ��)����(Sr)Ϊ�������ڢ�A��Ԫ�ء�����������ȷ���� (�����)��
���Ȼ���(SrCl2)��Һ������
��SrSO4������ˮ
�۹�ҵ�Ͽ����õ��SrCl2��ˮ��Һ��ȡ������(Sr)
�ܸߴ���ˮ�Ȼ��Ⱦ���(SrCl2��6H2O)������HCl��Χ�м��Ȳ��ܵõ�SrCl2
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
����Ԫ��A��B��M��ɵ��������ʷ�����Ӧ:��+��=��+��,���м���A��M���,����B��M���,��ֻ����M��
��1������Ϊ����ɫ���壬�Һͱ���Ϊ�����µ���ɫ��ζ���塣���ҵĵ���ʽΪ ;���ɱ�״����5.6L��ת�Ƶĵ�����Ϊ ;�����¶���ҺpH 7,�����ӷ���ʽ���� ��
��2������Ϊ��ʹƷ����ɫ����ɫ���壬��Ϊ������ɫ������������ס�����ԭ�Ӹ����Ⱦ�Ϊ1:2(M����+1��),ԭ������B����A�����A�����ڱ���λ��Ϊ �ڶ���ˮ��Ӧ�Ļ�ѧ����ʽΪ ��Ӧ����Һ�еμ�������ɫʯ����Һ������Ϊ
����ȷ��д�������ɱ��Ļ�ѧ����ʽ
����MCl2����Һ��ͨ�붡,�ɹ۲쵽��ɫ��MCl����,д���÷�Ӧ�����ӷ���ʽ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
X��Y��Z��W��Ԫ�����ڱ�ǰ�����ڵij���Ԫ�أ��������Ϣ���±�:
| X | X��һ�ֺ��ص�������Ϊ56��������Ϊ30 |
| Y | ��ˮ��Ԫ�غ�����ߵĽ���Ԫ�� |
| Z | ���³�ѹ�£�Z�����ǵ���ɫ���壬���ڻ�ɽ�ڸ������� |
| W | �۵��ӵ��Ų�ʽΪ3s23p3 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
A��B��C��D��ԭ��������������Ķ���������Ԫ�أ�A��C��Ԫ�����ڱ��е����λ����ͼ��AԪ��������������ϵĵ�����֮��Ϊ3��BΪ�ؿ��к������Ľ���Ԫ�ء�
| A | |
| | C |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
X��Y��Z��Q��MΪ�����Ķ�����Ԫ�أ���ԭ���������������й���Ϣ���±�:
| X | ��ֲ����������ȱ�ٵ�Ԫ�أ��ǵ����ʵ���Ҫ�ɷ� |
| Y | �ؿ��к����ӵ�һλ |
| Z | ����������ԭ�Ӱ뾶��� |
| Q | �����д���ʹ����Ͻ���Ʒ����ҵ�Ͽ��õ����������ķ����Ʊ� |
| M | ��ˮ�д���������Ԫ��֮һ������������ϼ��븺�۵Ĵ�����Ϊ6 |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com