
ij�о�С��������9ͼװ��̽��ͭ��Ũ���ᷴӦ�IJ��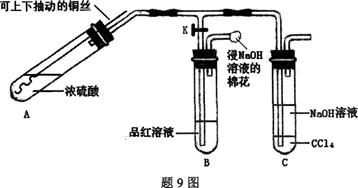
ʵ�鲽�����������£�
�ټ��װ�������ԣ���A�м���5 mLŨ���ᣬ��K����ͭ˿����Ũ�����У���A�����ȣ��۲쵽B��Ʒ����Һ��ɫ���ٹر�K��
��ʵ��۲쵽A��Һ����ͭ˿��ڣ��������ݣ�Һ���Ϸ���������������һ��ʱ�����Һ���а�ɫ������֣�����ͭ˿��ֹͣ���ȡ��ش��������⣺
��1����Һ�а�ɫ������ (�ѧʽ)��C��CCl4�������� ��
��2��ȡ��ͭ˿��������ˮϴ�������뵽ϡ�����У�ͭ˿��������Һ������д��A�п��ܷ�����Ӧ�Ļ�ѧ����ʽ�� �� ��
��3�������C�е��ϲ�Һ�壬�μӹ���ϡ�����BaCl2��Һ���а�ɫ����������
�ٷ���IJ�������Ϊ�� ��
�ڰ�ɫ����Ϊ�� ��
�۲�����ɫ������ԭ������� ��
��1��CuSO4����������
��2��Cu+H2SO4(Ũ) CuO+SO2��+H2O��Cu+2H2SO4(Ũ)
CuO+SO2��+H2O��Cu+2H2SO4(Ũ) CuSO4+SO2��+2H2O��
CuSO4+SO2��+2H2O��
��3���ٷ�Һ���� BaSO4����װ���е�O2�����˷�Ӧ
���������������1��Ũ�����ڼ���ʱ������ӦCu+2H2SO4(Ũ) CuSO4+SO2��+2H2O��������Ũ������ˮ���٣����Եõ�����ˮ����ͭCuSO4����ɫΪ��ɫ��SO2��Ư���ԣ���ʹijЩ��ɫ������Ʒ���Ϊ��ɫ�����װ��B��Ʒ����Һ��Ϊ��ɫ��SO2���������������NaOH������Ӧ��SO2��2NaOH=Na2SO3��H2O�����µ������ڵ�����ѹǿ��С��������������ķ���������SO2��CCl4�в������ܽ⡣��˿��Է�ֹ��������ķ�������2��A�п��ܷ�����Ӧ�Ļ�ѧ����ʽΪCu+H2SO4(Ũ)
CuSO4+SO2��+2H2O��������Ũ������ˮ���٣����Եõ�����ˮ����ͭCuSO4����ɫΪ��ɫ��SO2��Ư���ԣ���ʹijЩ��ɫ������Ʒ���Ϊ��ɫ�����װ��B��Ʒ����Һ��Ϊ��ɫ��SO2���������������NaOH������Ӧ��SO2��2NaOH=Na2SO3��H2O�����µ������ڵ�����ѹǿ��С��������������ķ���������SO2��CCl4�в������ܽ⡣��˿��Է�ֹ��������ķ�������2��A�п��ܷ�����Ӧ�Ļ�ѧ����ʽΪCu+H2SO4(Ũ) CuO+SO2��+H2O��Cu+ 2H2SO4(Ũ)
CuO+SO2��+H2O��Cu+ 2H2SO4(Ũ) CuSO4+SO2��+2H2O����3�������C�е��ϲ�Һ�壬�μӹ���ϡ�����BaCl2��Һ���а�ɫ�����������ٷ���IJ�������Ϊ����Һ����Na2SO3��BaCl2=BaSO3��+2NaCl��2BaSO3 +O2=2BaSO4. BaSO4�Ȳ�����ˮҲ�������ᡣ��˰�ɫ����ΪBaSO4����BaSO3�л�ԭ�ԣ�O2�������ԣ�����Һ�з���������ԭ��Ӧ������ɫ�Ȳ�����ˮҲ�����������BaSO4��
CuSO4+SO2��+2H2O����3�������C�е��ϲ�Һ�壬�μӹ���ϡ�����BaCl2��Һ���а�ɫ�����������ٷ���IJ�������Ϊ����Һ����Na2SO3��BaCl2=BaSO3��+2NaCl��2BaSO3 +O2=2BaSO4. BaSO4�Ȳ�����ˮҲ�������ᡣ��˰�ɫ����ΪBaSO4����BaSO3�л�ԭ�ԣ�O2�������ԣ�����Һ�з���������ԭ��Ӧ������ɫ�Ȳ�����ˮҲ�����������BaSO4��
���㣺����ͭ��Ũ���ᷴӦ��ԭ��������ijɷֵIJⶨ��֪ʶ��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
a��b��c��d��e�Ǻ���һ����ͬԪ�ص��������ʣ��ɷ�����ͼ��ʾ��ת�������У� a�ǵ��ʣ�c��d�������e������������Ӧ��ˮ���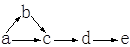
��1�����a�ǵ���ɫ���塢b�dz�������ζ�����塣��a�Ļ�ѧʽΪ ��b��e�����Ʒֱ�Ϊ ��c��d�Ļ�ѧ����ʽΪ ��
��2�����a�ǿ����к����������塢bΪ��̬�⻯�d�Ǻ���ɫ���塣��c�Ļ�ѧʽΪ ��a����b�Ļ�ѧ����ʽΪ ��d����e�����ӷ���ʽΪ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��֪ij����X�ܷ�������ת����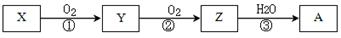
�����й�����ת����ϵ�����ʼ��䷴Ӧ�������������
| A����XΪN2��NH3��������������AΪ���� |
| B����XΪS ��H2S����ȫȼ�գ�����AΪ���� |
| C����XΪ�ǽ������ʻ�ǽ����⻯���A��һ���������ͭ��Ӧ����Y |
| D����Ӧ�ٺ͢�һ��Ϊ������ԭ��Ӧ����Ӧ��һ��Ϊ��������ԭ��Ӧ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��γ�ȥ���и���ĩ״������е�����(������Ϊ����)����ѡ�������ṩ���Լ��Ͳ�������������ڱ��ڡ�
��ѡ�Լ���A���B�ռ���Һ��C������Dˮ��ECO2��F���������Լ�
��ѡ�������ټ��ȣ��ڼ������ڣ��۹��ˣ��ܽᾧ
| �����ʵ����� | �����Լ� | ��Ҫ���� |
| (1)SiO2(NaHCO3) | | |
| (2)SiO2(CaCO3) | | |
| (3)SiO2(Si) | | |
| (4)NaCl(SiO2) | | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�������ȣ�ClO2������������ˮ�������Դ���Ϊԭ������ClO2�Ĺ�����Ҫ�������ٴ��ξ��ƣ��ڵ������NaCl��Һ����ClO2����ȡ��������������ͼ��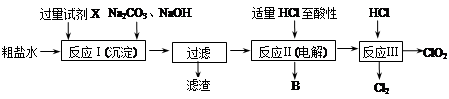
�� ��ʳ��ˮ�к���Ca2+��Mg2+��SO42-�����ʡ����Ӳ���ʱ��������ˮ���ȼ���������Լ�X��X���� ��(�ѧʽ)�����������ٲ������ټ��������Na2CO3��NaOH����ַ�Ӧ����һ����ȥ������ⷢ����Һ���Ժ���һ������SO42-����ԭ������ ������֪��Ksp(BaSO4)��1.1��10��10��Ksp(BaCO3)��5.1��10��9��
�����������У���ʳ��ˮ���ض������µ��õ��������������ᷴӦ����ClO2�����ʱ���ɵ�����B���� ����Ӧ��Ļ�ѧ����ʽΪ�� ����
�� ClO2�ܲ��ȶ������������ƣ�������ˮ���յõ�ClO2��Һ��Ϊ�ⶨ������Һ��ClO2�ĺ���������������ʵ�飺
����1��ȷ��ȡClO2��Һ10.00 mL��ϡ�ͳ�100 mL������
����2����ȡV1mL�������뵽��ƿ�У�����������pH��2.0������������KI���壬ҡ�ȣ��ڰ�������30���ӡ�(��֪��ClO2��I����H+��I2��Cl����H2O δ��ƽ)
����3���Ե�����Һ��ָʾ������c mol��L��1Na2S2O3��Һ�ζ����յ㣬����Na2S2O3��ҺV2 mL������֪��I2��2S2O32-��2I����S4O62-��
�� ȷ��ȡ10.00 mL ClO2��Һ�IJ����������� ����
�� �ζ������У�������ƽ�вⶨ���ε�ԭ������ ����
�� ������������ɼ����ԭClO2��Һ�����ʵ���Ũ��Ϊ�� mol��L��1���ú���ĸ�Ĵ���ʽ��ʾ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�ڻ�ѧ����Χ��Ũ����Ļ�ѧ���ʽ���������ʵ��̽���������������Ƿ����ձ��У����뼸��ˮ��������ȣ�Ȼ�����Ũ���ᣬ���ɺ�ɫ���ʡ�
��ش��������⣺
(1)���ɵĺ�ɫ����(����)�� (�ѧʽ)��
(2)���ֺ�ɫ���ʼ�����Ũ�������û�����������壬����һ�������д̼�����ζ���Ǵ�������Ҫ��Ⱦ��֮һ�����ִ̼�����ζ������ɷ��� (�ѧʽ)����Ӧ�Ļ�ѧ����ʽΪ ��
(3)��������Ũ���ᷴӦ��ʵ������˵��Ũ������� (�����)��
| A������ | B����ˮ�� | C����ˮ�� | D��ǿ������ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��AԪ�ص�һ�ֵ�����һ����Ҫ�İ뵼����ϣ���AԪ�ص�һ�ֻ�����C��������������ܵ��ִ�ͨѶ���ϡ������ά��C���ռӦ���ɺ�AԪ�صĻ�����D��
(1)��Ԫ�����ڱ��У�Aλ�� �壬��Aͬ�嵫���ԭ��������AС��Ԫ��B��ԭ�ӽṹʾ��ͼΪ ��A��B��ԭ�ӵĵ��Ӳ�ṹ�ϵ���ͬ���� ��
(2)����C������ѧ��Ӧ������ ����Ӧ�Ļ�ѧ����ʽ�� ��
(3)��C�봿���ϸ�������ʱҲ������ѧ��Ӧ����D��ͬʱ������B�����������E����ȫ����E��ȫ����D��������ˮ�л�Ϻ��ַ�����ѧ��Ӧ���ɺ�A�Ļ�����F��
��д������D��F�Ļ�ѧ��Ӧ����ʽ�� ��
��Ҫ����������ۻ������������п�ѡ�õ��� ��
A����ͨ�������� B��ʯӢ��������
C����������� D�������
(4)100 g C��ʯ��ʯ�Ļ�����ַ�Ӧ�����ɵ������ڱ�״���µ����Ϊ11.2 L,100 g�������ʯ��ʯ������������ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ͼ��A��J�ֱ������ط�Ӧ��һ�����ʡ���֪A�ֽ�õ������ʵ�����B��C��D��ͼ���в���������δ�����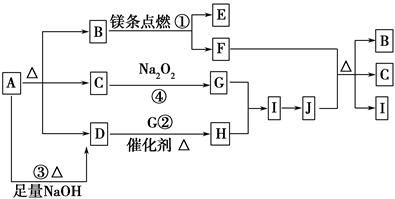
��֪��2Mg��CO2 2MgO��C������д���¿հף�
2MgO��C������д���¿հף�
(1)A�Ļ�ѧʽ________��
(2)д����Ӧ�ڵĻ�ѧ����ʽ��________________________________________��
(3)д����Ӧ�۵����ӷ���ʽ��________________________________________��
(4)J��F��Ӧ�Ļ�ѧ����ʽ��_________________________________________��
(5)�ڷ�Ӧ���У������ɱ�״����2.24 L Gʱ��ת�Ƶ�����Ϊ________ mol��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�������ƣ�NaClO2����һ��ǿ������Ư�����㷺���ڷ�֯��ӡȾ��ʳƷ��ҵ�����ڼ��Ի������ȶ����ڡ�ijͬѧ�������Ϻ��������NaClO2����Ҫ�������¡�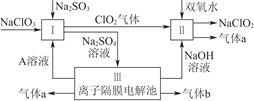
��1��˫��ˮ�ĽṹʽΪ____________�����з�����Ӧ�Ļ�ԭ����__________���ѧʽ����
��2�����з�Ӧ�����ӷ���ʽ��_____________________________________________________________________________________________________________________________________��
��3��A�Ļ�ѧʽ��________��װ�â���A��________����������
��4��ClO2��һ�ָ�Чˮ�������������������ƺ�ϡ����Ϊԭ���Ʊ���
��д���÷�Ӧ�Ļ�ѧ����ʽ��________________________________________________________________________________________________________________________________________________��
���о�����������Ӧ��ʼʱ����Ũ�Ƚϴ��������������Cl2�������ӷ���ʽ���Ͳ���Cl2��ԭ��________________________________________________________________________
________________________________________________________________________��
��5��NaClO2���ʿɷֽ�ΪNaClO3��NaCl��ȡ������NaClO2������һ�ݸ����ʵ�����ʹ֮���ʣ���һ���ϸ棬�������Һ�����ֱ�������FeSO4��Һ��Ӧʱ������Fe2�������ʵ���________�����ͬ��������ͬ�������жϡ�����
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com