
���ù�������KxFe��C2O4��y��3H2O��FeΪ��3�ۣ���ʵ�����Ʊ��Ͳⶨ����ɵķ���������ʾ��
��.�Ʊ���
��1����K2C2O4��FeCl3�Ʊ��������ϵķ�Ӧ����________������ţ���
�����ӷ�Ӧ���ڷ�������ԭ��Ӧ����������ԭ��Ӧ���ܻ��Ϸ�Ӧ
��2���ᾧʱӦ��������Һ���ںڰ����ȴ����������������������ԭ����_________��
��3������4��ʵ�������____________��
��.��ɲⶨ��
��ȡһ������ʵ�����õľ���������ƿ�У�����������ˮ��ϡH2SO4����C2O42-��ȫת��ΪH2C2O4����0.10 mol��L��1 KMnO4��Һ���еζ�������KMnO4��Һ24.00 mLʱǡ����ȫ��Ӧ������������MnO4-�Ļ�ԭ������Mn2�������ټ��������Ļ�ԭ������Fe3����ȫת��ΪFe2������KMnO4��Һ�����ζ�����Fe2����ȫ����ʱ����ȥKMnO4��Һ4.00 mL��
��4�������������H2C2O4��Fe2�������ӷ���ʽ�ֱ���___________�� ________��
��5������100 mL 0.10 mol��L��1 KMnO4��Һ���������ζ�ʵ��������IJ����������ձ�������������ͷ�ιܡ���Ͳ����ƿ�⣬����________��________��д���ƣ���
��6��ͨ�����㣬�û�����Ļ�ѧʽ��____________��
 ������ϵ�д�
������ϵ�д� �±�Сѧ��Ԫ�Բ���ϵ�д�
�±�Сѧ��Ԫ�Բ���ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��һ��ɫ����Һ����ȷ���Ƿ����������ӣ� Fe2����Mg2����Al3����Ba2���� ��
�� ��Cl����I����
��Cl����I���� ��ȡ����Һ����ʵ�飺
��ȡ����Һ����ʵ�飺
| ʵ�鲽�� | ʵ������ |
| (1)ȡ��������Һ���Ӽ�����ɫʯ����Һ | ��Һ��� |
| (2)ȡ��������Һ���ȣ���CuƬ��ŨH2SO4������ | ����ɫ���������������������ɺ���ɫ |
| (3)ȡ��������Һ����BaCl2��Һ | �а�ɫ���� |
| (4)ȡ(3)���ϲ���Һ����AgNO3��Һ | �а�ɫ�������Ҳ�����ϡHNO3 |
| (5)ȡ��������Һ����NaOH��Һ | �а�ɫ������NaOH����ʱ���������ܽ� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�����̿�MnCO3��������Fe2O3��FeO��HgCO3��2HgO�����ʣ���ҵ�������̿���ȡ�̣������������£�
��ش��������⣺
��1�����Һ1�м����ˮ�����Ҫ �������ܴﵽ����Ҫ��
��2���������õĿ�������Ĥ���뷨�Ʊ��ĸ����������÷�����ԭ���� ��
��3����������Ҫ�ɷ�Ϊ(NH4)2S����Һ2�з�����Ҫ��Ӧ�����ӷ���ʽΪ ��
��4��д�������ĵ缫��Ӧʽ ��˵�����Һѭ����ԭ�� ��
��5��д�����ȷ����̵Ļ�ѧ����ʽ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ij�ִ����к�����ɳ��Ca2����Mg2����Fe3����SO�����ʡ�ijͬѧ��ʵ����������������ִ����Ʊ����εķ�������(���ڳ������Լ��Թ���)��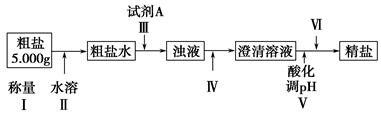
��ش��������⣺
(1)Ϊ������ѡ����������(�ñ����ĸ��д)��________��
A���ձ���B���Թܡ�C����������D����Һ©����E��©����F���ƾ��ơ�
G��������
(2)�������г���Na2CO3��Һ��NaOH��Һ��BaCl2��Һ��Ϊ�����Լ������������Լ���˳��Ϊ��NaOH��Һ��________��________��
(3)�������У��жϼ���BaCl2�ѹ����ķ�����___________________________________
(4)������Ӧѡ�������________����������������������Ⱥ�˳��Ե��������ʵ����������Ӱ����___________________________________
(5)��������________(ѡ��������������ƣ��ñ����ĸ�������Ⱥ�˳����д)��
a�����ˡ�ϴ�� B��������Ũ�� c����ȡ����Һ D����ȴ���ᾧ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��15�֣�п�̸ɵ�������Ĺ���������ڷ�������뻷���н��������Σ�����ԷϾɵ�ؽ�����Դ�������Ե÷dz���Ҫ��ij��ѧ��ȤС����������´����������շϵ���еĸ�����Դ��
��1������п�̸ɵ�صĵ����ΪKOH���ܷ�ӦΪZn+2MnO2+2H2O=2MnOOH+Zn(OH)2���为���ĵ缫��ӦʽΪ_________________________________________________________��
��2���������60����ˮ�ܽ⣬Ŀ���Ǽӿ��ܽ����ʣ�����������¶Ȳ���̫�ߣ���ԭ����___________��
��3������A������Ϊ_____________ ��
��4����������Ҫ�ɷ�Ϊ���̻������̻�����м���һ������ϡ���ᡢϡ���ᣬ�����Ͻ�����������Ϊֹ������Ҫ��ӦΪ2MnO(OH)+MnO2+2H2C2O4+3H2SO4=2MnSO4+4CO2��+6H2O��
�ٵ�1 mol MnO2�μӷ�Ӧʱ������___________mol���ӷ���ת�ơ�
��MnO(OH)��Ũ�����ڼ���������Ҳ�ɷ�����Ӧ����д���䷴Ӧ�Ļ�ѧ����ʽ��________________��
��5��ͭñ�ܽ�ʱ����H2O2��Ŀ����_______________________________���û�ѧ����ʽ��ʾ����ͭñ�ܽ���ȫ�ɲ���_____________������ȥ��Һ�й�����H2O2��
��6��п�̸ɵ�������Ĺ�����KMnO4��Һ���ա��ڲ�ͬpH�£�KMnO4��Һ��Hg�������ʼ���Ҫ��������ͼ��ʾ��
������ͼ��֪��
��pH��Hg�����ʵ�Ӱ�������__________________________________________________.
����ǿ���Ի�����Hg�������ʸߵ�ԭ�������_____________________________________.
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
���Ų�������ʯ�͡���������ˮ�������������ճ�������Ʒ����ҵ�ƹ㣬�ѽ������汻�������ӣ�����Ϊ���ִ��������͡�ս�Խ�����������߹���װ��ˮƽ���ɻ�ȱ����Ҫս�����ʡ���ҵ��Ҫ�Զ�������Ϊԭ��ұ�������ѡ�
��.�������ѿ����������ַ����Ʊ���
����1�����ú���Fe2O3����������Ҫ�ɷ�ΪFeTiO3������TiԪ�ػ��ϼ�Ϊ+4�ۣ���ȡ������Ҫ�������£�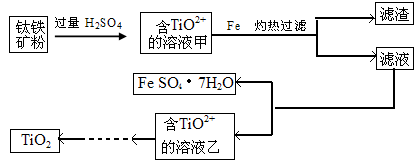
��1������Һ����̷�����IJ��������� ��
��2������Һ�г���TiO2+֮����еĽ����������� ��
��3����֪10kg������������Ԫ�ص���������Ϊ33.6%���ܹ��õ��̷�����22.24kg���Լ������ټ������۵�������
����2��TiCl4ˮ������TiO2��XH2O�����ˡ�ˮϴ��ȥ���е�Cl-���ٺ�ɡ����ճ�ȥˮ�ֵõ�����TiO2���˷����Ʊ��õ��������������ѡ�
��4����TiCl4ˮ������TiO2��XH2O�Ļ�ѧ����ʽΪ ��
�ڼ���TiO2��XH2O��Cl-�Ƿ����ķ����� ��
��.�������ѿ�������ȡ�ѵ���
��5��TiO2��ȡ����Ti���漰���IJ������£�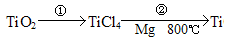
��Ӧ�ڵĻ�ѧ����ʽ�� ���÷�Ӧ�ɹ���Ҫ������������ԭ���� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�������(LiFePO4)����Ϊ������ǰ;������ӵ���������ϡ�ij��ҵ���ø�������Һ�����������,�����˴�������������Һһ����;��������Ҫ��������: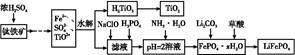
��֪:H2TiO3����������ˮ�����ʡ�
(1)��������Ũ���ᴦ��֮ǰ,��Ҫ����,��Ŀ���� ����
(2)TiO2+ˮ������H2TiO3�����ӷ���ʽΪ ����
(3)����NaClO������Ӧ�����ӷ���ʽΪ ����
(4)��ʵ����,����Һ�й��˳�H2TiO3��,������Һ����,Ӧ��β����� ��
(5)Ϊ�ⶨ�����������ĺ���,ijͬѧȡ��Ũ����ȴ�������Һ(��ʱ�������е�����ȫ��ת��Ϊ����������),��ȡKMnO4��Һ�ζ�Fe2+�ķ���:(������KMnO4���������ʷ�Ӧ)�ڵζ�������,��δ�ñ�Һ��ϴ�ζ���,��ʹ�ⶨ�����������(�ƫ�ߡ�����ƫ�͡�����Ӱ�족),�ζ��յ���������� ���ζ�����ʱ,��ȡa g������,������,��c mol/L KMnO4��Һ�ζ�,����V mL,����Ԫ�ص����������ı���ʽΪ����������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
������أ� ������ǿ�����ԣ�������ԭΪ����أ���80 �����������ֽ⡣ʵ����ģ�ҵ�ϳɹ�����ص��������£�
������ǿ�����ԣ�������ԭΪ����أ���80 �����������ֽ⡣ʵ����ģ�ҵ�ϳɹ�����ص��������£�
��1������狀��������Ƴɵ��Һ���Բ����缫���е�⣬���ɹ��������Һ��д�����ʱ������Ӧ�����ӷ���ʽ_____________________________________
___________________________________��
��2����֪������ʵ��ܽ����������ͼ��ʾ����ʵ�������ᴿ������شֲ�Ʒ��ʵ������������Ϊ����������شֲ�Ʒ��������ˮ�У�________________�����
��3����Ʒ�й�����صĺ������õ��������вⶨ��ʵ�鲽�����£�
����1����ȡ���������Ʒ0.300 0 g�ڵ���ƿ�У�����30 mLˮ�ܽ⡣
����2������Һ�м���4.00 0 g KI���壨�Թ�������ҡ�ȣ��ڰ�������30 min��
����3���ڵ���ƿ�м�������������Һ�ữ���Ե�����Һ��ָʾ������0.100 0 mol��L��1Na2S2O3����Һ�ζ����յ㣬������Na2S2O3����Һ21.00 mL��
����֪��Ӧ��I2��2S2O32-=2I����S4O62-��
��������2��δ������ƿ���ڰ�������30 min�����������в���3����ⶨ�Ľ������________��ѡ�ƫ����ƫС��������Ӱ�족������������3�еζ��յ��������____________________________________________��
�ڸ�����������ɼ��������Ʒ�й�����ص���������Ϊ_______________��
��Ϊȷ��ʵ������ȷ�ԣ�����Ϊ����Ҫ____________________________��
��4����0.40 mol���������0.20 mol�������Ƴ�1 L��Һ����80 �������¼��Ȳ���tʱ������Һ�еμ�������FeCl3��Һ���ⶨ��Һ�и��ɷֵ�Ũ����ͼ��ʾ��H��Ũ��δ��������ͼ������X�Ļ�ѧʽΪ________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ϼʯ�ң���ѧʽΪ KNa3[AlSiO4]4����Ҫ�ɷ�Na2O��K2O��Al2O3��SiO2����̼���ơ�̼��غ��������Ĺ����������£�
��֪��NaHCO3��Һ��pHԼΪ8��9��Na2CO3��Һ��pHԼΪ11��12���ܽ���˹����������Һ�к��ơ��غ����Ŀ��������࣬�ƺ����������������ϼʯ���С��������ʵ��ܽ�ȼ���ͼ��
�Իش��������⣺
��1�����յõ�����M�Ļ�ѧ����ʽ��________________________________��
��2��X������___________����ҺW����Ҫ���е�������____________����д���֣�
��3��������õ�̼���ƾ���IJ���Ϊ���� �� �� ��ϴ�ӡ����
��4��̼�ữ���з�����Ҫ��Ӧ�����ӷ���ʽ��__________________________��
��5��̼�ữ�����pH��8��Ŀ����_______________________________��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com