
����Ŀ����2mol��L��1A�����1mol��L��1B�����ϣ�����һ�������·�����Ӧ��2A(g)��B(g)![]() 2C(g)������2s����C��Ũ��Ϊ0.6mol��L��1���������м���˵����
2C(g)������2s����C��Ũ��Ϊ0.6mol��L��1���������м���˵����
��������B��ʾ�ķ�Ӧ��ƽ������Ϊ0.6mol��L��1��s��1
��������A��ʾ�ķ�Ӧ��ƽ������Ϊ0.3mol��L��1��s��1
��2sʱ����A��ת����Ϊ70%
��2sʱ����B��Ũ��Ϊ0.7mol��L��1
������ȷ���ǣ� ��
A.�� ��B.�� ��C.�� ��D.�� ��

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����Ԫ�����þ��ֲ���Ҷ����������������Ľ�������ʮ����Ҫ�����ã��仯����Ҳ���Ź㷺��Ӧ�á�
(1)��̬Bԭ�ӵļ۵����Ų�ͼΪ_____________________�����һ�����ܱ�Be__________��������������С������
(2)����B���γ������ӣ���[B(OH)4]����[BH4]���ȡ�������[BH4]��������ԭ�ӵ��ӻ���ʽΪ________��д�����以Ϊ�ȵ������һ�������Ӻ�һ�ַ��ӵĻ�ѧʽ��_________________��
(3)���Ȼ���ķе�Ϊ12.5�棬���Ȼ�þ�ķе����1200�棬ԭ����____________________________________��
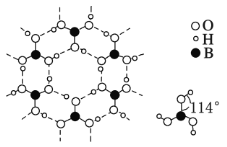
(4)���ᾧ����Ƭ״�ṹ����ͼ��ʾ��������һ��Ľṹ��ÿһ���ڴ��ڵ���������______________�����ᾧ������ˮ���ܽ�Ⱥ�С��������ˮ�нϴ�ԭ���� _____________________________________________��
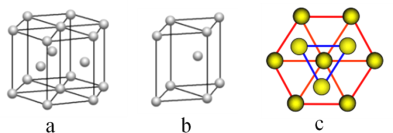
(5)þ���ʾ�����ԭ�ӵĶѻ�ģ����ͼ�����Ķѻ�ģ������Ϊ__________________�����ڵ��ĸ�þԭ�ӵ��������߹��ɵ��������弸����������2a cm3��þ���ʵ��ܶ�Ϊ�� g��cm��3����֪����٤������ΪNA����þ��Ħ����������ʽΪ________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�����������̼����������ۺ������ǵ�ǰ����Ҫ����֮һ��
��֪����.NO(g)+CO2(g)![]() NO2(g)+CO(g) ��H1=+234kJ��mol-1
NO2(g)+CO(g) ��H1=+234kJ��mol-1
��.2NO(g)+2CO(g)![]() N2(g)+2CO2(g) ��H2=-745kJ��mol-1
N2(g)+2CO2(g) ��H2=-745kJ��mol-1
(1)NO2(g)��CO(g)��Ӧ����������������Ȼ�ѧ����ʽΪ______________��
(2)��Ӧ��������Ӧ�Ļ��E_____��H1(�>������<����=��)��
(3)һ��ѹǿ�£��ܱ������з�����Ӧ���ͷ�Ӧ�����ﵽƽ����������������䣬�����¶ȣ�CO(g)���������________(����������١�����ȷ����)��ԭ��Ϊ_____________________��
(4)����ʼ�¶�Ϊt�桢�ݻ�Ϊ10L�ĺ��ݾ��ȵ��ܱ������г���2molNO(g)��1molCO2(g)��������Ӧ����5minʱ�ﵽƽ�⡣��
��������ʵ��˵���÷�Ӧ�ﵽƽ��״̬����________(��ѡ����ĸ)
A.��������¶Ȳ��� B.��������ѹǿ����
C. NO2��CO��Ũ��֮��Ϊ1: 1 D.��������ƽ����Է�����������
��t��ʱ������һ�ݻ�Ϊ10 L�ĺ��º��ݵ��ܱ������г���2 mol NO(g)��1 mol CO2(g)��������Ӧi���ﵽƽ���ʱ��______5 min(�>������<����=��)��
(5)��ij�ܱ������г���10 mol CO(g)��20 mol NO(g)��������Ӧii��CO��ƽ��ת����(��)���¶�(T)��ѹǿ(p)�Ĺ�ϵ��ͼ��ʾ��
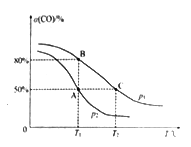
��A��B��C �����ƽ�ⳣ��KA��KB��KC�Ĵ�С��ϵΪ________��p1��p2�Ĵ�С��ϵΪ__________��
��A��ʱ.������������Ϊ10 L����T1��ʱ���÷�Ӧƽ�ⳣ��K��ֵΪ____________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��������ͼװ�ÿ�����֤�ǽ����Եı仯���ɡ�

(1)����A������Ϊ________�������D��������________________��
(2)��֪�ڳ����£�������غ�Ũ���ᷴӦ������������ʵ����������ҩƷNa2S��Һ��KMnO4��Ũ���ᡢMnO2����ѡ�����ҩƷ���ʵ����֤�ȵķǽ����Դ�����ģ�װ��A��B��C����װҩƷ�ֱ�Ϊ________��________��________��װ��C�е�ʵ������Ϊ_________________�����ӷ���ʽΪ______��
(3)��Ҫ֤���ǽ����ԣ�S>C>Si����A�м�________��B�м�Na2CO3��C�м�________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ���ۺϼ�ʽ�Ȼ���[![]() ��mΪ�ۺ϶ȣ�Ϊ���߷������������㷺Ӧ����ˮ������������ʯ��������Ƥ��
��mΪ�ۺ϶ȣ�Ϊ���߷������������㷺Ӧ����ˮ������������ʯ��������Ƥ��![]() Ϊԭ�ϣ������������¾�������ˮ�⡢�ۺϡ��컯�Ȳ��裬���Ƶþۺϼ�ʽ�Ȼ�����
Ϊԭ�ϣ������������¾�������ˮ�⡢�ۺϡ��컯�Ȳ��裬���Ƶþۺϼ�ʽ�Ȼ�����
(1)������ϴ��Һ����Ҫ����![]() ��
��![]() ��ͨ��������Ƶ�
��ͨ��������Ƶ�![]() ����Ӧ�Ļ�ѧ����ʽ________��������
����Ӧ�Ļ�ѧ����ʽ________��������![]() Ҳ���Խ�
Ҳ���Խ�![]() ������
������![]() �������ڵ�ȱ����________________��
�������ڵ�ȱ����________________��
(2)�������������У��������Ũ�Ȼ�Ͷ���������˻ή��![]() ��ת���ʣ������ܲ�����Ӱ��Ϊ________________��
��ת���ʣ������ܲ�����Ӱ��Ϊ________________��
(3)��ȷ��ȡ�ۺϼ�ʽ�Ȼ�����Ʒ1.5000g������250mL����ƿ�У���������ϡ���ᣬ���ȣ�Ѹ�ټ����Թ�����![]() ��Һ��
��Һ��![]() ��
��![]() ������Ϊ
������Ϊ![]() ������ַ�Ӧ�����
������ַ�Ӧ�����![]() ��
��![]() ������ȥ���ټ���������
������ȥ���ټ���������![]() ��ɵĻ��ἰ4~5��ָʾ������
��ɵĻ��ἰ4~5��ָʾ������![]() ��Һ�ζ����յ㣨�ζ�������
��Һ�ζ����յ㣨�ζ�������![]() ��
��![]() ��Ӧ����
��Ӧ����![]() ��
��![]() ��������
��������![]() ��Һ
��Һ![]() ��
��
������ȡ�������ľۺϼ�ʽ�Ȼ�����Ʒ����ˮ�У����500mL��Һ��ȡ25mL��Һ����ƿ�У��ø���أ�![]() ����Һ��ָʾ������
����Һ��ָʾ������![]() ����Һ�ζ����յ㣬����
����Һ�ζ����յ㣬����![]() ��Һ8.10mL��
��Һ8.10mL��
�������Ʒ����Ԫ�غ���Ԫ�ص���������֮��![]() Ϊ________��д��������̣����������λ��Ч���֣���
Ϊ________��д��������̣����������λ��Ч���֣���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����1��д����������ת���Ļ�ѧ����ʽ��

��_____________________________________________��
��_____________________________________________��
��_____________________________________________��
��2��д����������ת���Ļ�ѧ����ʽ��

��_____________________________________________��
��_____________________________________________��
��_____________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����NAΪ�����ӵ�������ֵ������˵������ȷ���ǣ� ��
A.32g S8��S��ĿΪNA
B.71g����ͨ��������NaOH��Һ��ת�Ƶ�����ĿΪNA
C.100mLŨ��Ϊ0.1mol/L��������Һ�У�SO42-��ĿΪ0.02NA
D.11.2L Cl2����ˮ����Һ��Cl-��ClO-��HClO������֮��һ��ΪNA
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����֪��N2(g)��3H2(g) ![]() 2 NH3(g) ��H����92.4 kJ��mol��1��һ�������£������ݻ���ͬ�Һ��ݵ��ܱ����������ң��� �����ͨ��1 mol N2��3 mol H2���ﵽƽ��ʱ�ų�����Q1 kJ���� ������ͨ��0.5 mol N2��1.5 mol H2���ﵽƽ��ʱ�ų�����Q2 kJ�������й�ϵʽ��ȷ����
2 NH3(g) ��H����92.4 kJ��mol��1��һ�������£������ݻ���ͬ�Һ��ݵ��ܱ����������ң��� �����ͨ��1 mol N2��3 mol H2���ﵽƽ��ʱ�ų�����Q1 kJ���� ������ͨ��0.5 mol N2��1.5 mol H2���ﵽƽ��ʱ�ų�����Q2 kJ�������й�ϵʽ��ȷ����
A. Q1��2Q2��92.4B. 92.4��Q1��2Q2
C. 92.4��Ql��2Q2D. Q1��2Q2��92.4
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ������ͼ��ʾװ�ý�������ʵ�飺��������Һ������У�Ԥ���������ʵ���������
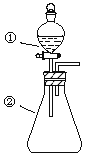
ѡ�� | �������� | �������� | Ԥ����е����� |
A | ϡ���� | ̼�������������ƵĻ����Һ | ������������ |
B | Ũ���� | ��ɰֽ��ĥ�������� | ��������ɫ���� |
C | ������ˮ | ���۵⻯����Һ | ��Һ����ɫ |
D | Ũ���� | MnO2 | ��������ɫ���� |
A.AB.BC.CD.D
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com