
��ѧ��ѧ�г����ļ������ʴ������¹�ϵ�����м��Ǻ�ɫ�ǽ������ʣ����������г����Ľ������ʣ�D�Ǻ���ɫ���塣��ͼ�в��ֲ���ͷ�Ӧ��������ȥ��
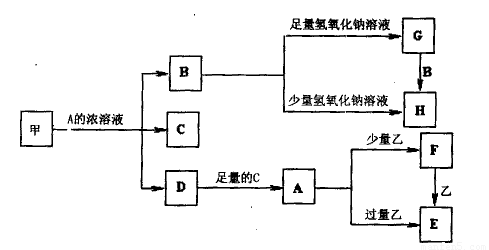
�ش��������⣺
��1��д������A��Ũ��Һ��Ӧ�Ļ�ѧ����ʽ ��
��2�����������Ũ�ȵ�G��Һ��H��Һ��Ϻ�õ�����Һ�е�����Ũ�ȴ�С��ϵΪ ��
��3����ȥG�����к���H���ʲ��õķ����� ��
��4��A��Һ��һ����ʹʪ��ĺ�ɫʯ����ֽ����ɫ�����巴Ӧ������һ���Σ����ε���Һ�����ԣ���ԭ���ǣ������ӷ���ʽ��ʾ�� ��
��5��д����E��Һ�м�������ϡ�����Ӧ�����ӷ���ʽ
��
����ҺF�������ɡ����յ��������ټ���ʱ���ù������ʵĻ�ѧʽΪ ��
��6��ȷ��E��������ʵ��ķ���Ϊ ��
��1��C+4HNO3��Ũ�� CO2��+4NO2��+2H2O��2�֣�
CO2��+4NO2��+2H2O��2�֣�
��2��c��Na+�� >c��HCO3-��>c��CO32-��>c��OH-��> c��H+����2�֣� ��3�����ȣ�2�֣�
��4��NH4++H2O NH3•H2O+H+��2�֣�
NH3•H2O+H+��2�֣�
��5��3Fe2+��NO3��+4H+ ��3 Fe3++NO��+2H2O��2�֣� Fe2O3��2�֣�
��6���ȼ���KSCN��Һ�ޱ仯�ټ�����ˮ��������ȣ���Һ���ɫ�������������𰸣���2�֣�
����������



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�


| ||
| ||
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
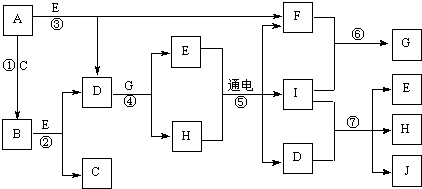
 A-K����ѧ��ѧ�г����ļ������ʣ�����֮���ת����ϵ��ͼ��ʾ����֪������AΪ���嵥�ʣ�BΪ����ɫ��ĩ��G��HΪ��̬���ʣ�I�ڳ�����ΪҺ�壬D��E��F��ˮ��Һ���ʼ��ԣ���C�ı�����Һ��ȡF��E����Ҫ�Ļ����������ش��������⣺
A-K����ѧ��ѧ�г����ļ������ʣ�����֮���ת����ϵ��ͼ��ʾ����֪������AΪ���嵥�ʣ�BΪ����ɫ��ĩ��G��HΪ��̬���ʣ�I�ڳ�����ΪҺ�壬D��E��F��ˮ��Һ���ʼ��ԣ���C�ı�����Һ��ȡF��E����Ҫ�Ļ����������ش��������⣺

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�



�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�



�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�

| ||
| ||
 NH3?H2O+H+
NH3?H2O+H+ NH3?H2O+H+
NH3?H2O+H+�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com