
��֪Fe2O3��H2��Ӧ�����¶Ȳ�ͬ��������Fe3O4���ɡ�ij��ѧ��ȤС������H2��ԭFe2O3��ʵ���У��ô����������ɵĺ�ɫ��ĩX��Ϊ̽��X����ɣ����ǽ���������ʵ�飺
(1)��ͬѧ��Ϊ��ɫ��ĩX�ܱ��������������X����������ͬѧ��ͬ�����Ľ��ۣ�ԭ����___________________________��
(2)��ͬѧ�Ƚ�������ɫ��ĩX����װ����������ͭ��Һ���ձ��У������岿���ܽ⣬�м�������ɫ�������������ˣ�Ȼ���������м������ᣬ�ٵμӼ���KSCN��Һ����Һ���ֺ�ɫ��ͨ�������������ͬѧ�ó�X�������Fe��Fe3O4��
�ٵμ�KSCN��Һ��Ŀ����_____________________��
��������ĩXֱ�Ӽ��������У��ټ�KSCN��Һ����Һ�����ֺ�ɫ�����ֺ�ɫ��ԭ����(�����ӷ���ʽ��ʾ) _____________��
(3)��ͬѧ��ʵ�鷽����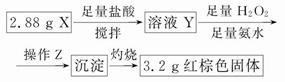
�ٲ���Z��__________________��
��ͨ���������ݣ��ó�2.88 g��ɫ��ĩX�и��ɷֵ����ʵ���Ϊ____________________________________��

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
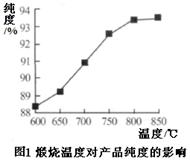
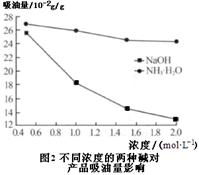
��15�֣�Fe2O3�׳��������죬������������ɫ������ҵ��ú��ʯ����Ҫ�ɷ֣�SiO2 49.5%��Fe2O3 20.6%��Al2O318.9%���Լ�MgO��FeO���������ʣ��Ʊ����о����������Ӧ�á�
��һ���Ʊ���������
1��Ԥ��������ú��ʯ���飬��350���±���2Сʱ��
2�������ܽ⣺��Ԥ�������ú��ʯ������������Ϊ15%������������Һ�У����ˡ�����Һ�м����H2O2��
3������pH����������Һ�м�1mol/LNaOH��Һ������Һ��pH���ٹ��ˣ��õ�������
4����Ʒ����������������ˮϴ����ɡ����ա���ĥ����ɸ�ò�Ʒ��
��֪����������������������ʽ����ʱ��Һ��pH���±�
| ������ | Fe(OH)3 | Al(OH)3 | Fe(OH)2 | Mg(OH)2 |
| ��ʼ���� | 1.3 | 3.3 | 7.5 | 10.3 |
| ��ȫ���� | 2.8 | 5.2 | 9.7 | 12.2 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
Fe2����I�������ֳ����Ļ�ԭ�����ӡ�
��1����FeSO4��Һ�еμ���ˮ����Һ��dz��ɫ��ɻ�ɫ����Ӧ�����ӷ���ʽΪ ����KI��Һ�еμ���ˮ����Һ����ɫ��ɻ�ɫ����Ӧ�����ӷ���ʽ�� ��
��2������FeSO4��Һ��KI��Һ����ˮΪ�Լ���֤I���Ļ�ԭ��ǿ��Fe2�������ʵ�鷽�����������ʵ�鲽�衢Ԥ������ͽ��ۡ�������ѡ�Լ���3 mol��L��1 H2SO4��0.01 mol��L��1 KMnO4��20% KSCN��3%H2O2��������Һ����ɫʯ����Һ��
| ʵ�鲽�� | Ԥ����������� |
| ����1��ȡ2mLFeSO4��Һ��2mLKI��Һ������Թ��У��ٵμ�1��2����ˮ�� | �� |
| ����2��____________________________________ ____________________________________�� | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ijѧϰС����ͨ��ʵ��̽���̷���FeSO4��7H2O���ȷֽ�IJ��
��ʵ��ǰ����
��1�����۷��� С���Ա�������Ϸ�������Ϊ���зֽ���ﲻ���ܵ��� ��
a��Fe2O3��SO3��H2O b��Fe2O3��SO2��SO3��H2O
c��FeO��Fe2O3��SO2��SO3��H2O
��2���������� ��ѹ��SO3�۵�16��8�棬�е�44��6��
��ʵ��̽����
������Ͽ��ܵ���ϲ��룬��ѧϰС���������ͼ��ʾ��ʵ��װ�ã�ͼ�м��ȡ��г������Ⱦ�ʡ�ԣ���
��3��ʵ�����
���������Ӻ��װ��A��B�����ԵIJ���Ϊ ��
��ȡһ�����̷���������A�У�ͨ��N2������װ���ڵĿ������ر�k���þƾ��Ƽ���
˫ͨ�ܡ�
�۹۲쵽A �й��������ɫ��B���Թ��ռ�����ɫҺ�壬C����Һ��ɫ��
�ܴ�A�з�Ӧ��ȫ����ȴ�����º�ȡ������Ӧ��������Թ��У����������ܽ⣬ȡ
�������뼸��KSCN��Һ����Һ���ɫ��
����Bװ�õ��Թ��е��뼸��BaCl2��Һ����Һ����ǡ�
( 4��ʵ��������
����1��B���ռ�����Һ���� ��
����2��C����Һ��ɫ������֪�������� ��
����3���ۺϷ�������ʵ��ۺܿ͢���֪�������һ����Fe2O3��
��ʵ�鷴˼��
��5����ָ����С����Ƶ�ʵ��װ�õ����Բ��㣺 ��
��6���ֽ��Ĺ����п��ܺ�������FeO��ȡ����ʵ����������ܽ�����Һ�������Թ��У�
ѡ��һ���Լ����𣬸��Լ�����ʵ��� ��
a����ˮ��KSCN��Һ b������KMnO4��Һ c�� H2O2 d�� NaOH��Һ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
����ʯ��Ҫ�ɷ�Ϊ����������������в�����Ԫ�غ���Ԫ�أ������ʲ���H2SO4��Ӧ����ij�о���ѧϰС���ij����ʯ������������Ļ�ѧʽ����̽����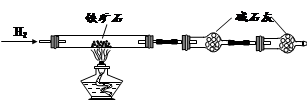
A B C
������ʯ�к������IJⶨ
�� ����ͼ��װ���������װ�õ������ԣ�
�� ��5.0g����ʯ����Ӳ�ʲ������У�װ��B��C�е�ҩƷ��ͼ��ʾ���г�������ʡ�ԣ���
�� ����˵����ܿڴ�����ͨ��H2��____________����ȼA���ƾ���
�� ��ַ�Ӧ�����ƾ��ƣ��ٳ���ͨ����������ȫ��ȴ��
��1��װ��C������Ϊ________________________________________________��
��2�����е�ȼA���ƾ���ǰ�������Ϊ��______________________________ ��
��3����÷�Ӧ��װ��B����1.35g��������ʯ�����İٷֺ���Ϊ____________��
������ʯ�к������IJⶨ
��1�����������������__________________________________________��
��2����������õ��IJ����������ձ�������������ͷ�ιܡ�____________��
��3�������йز���IJ�����˵����ȷ����__________________��
a����Ϊ��ˮΪ��ɫ�����Եζ������в����ָʾ��
b���ζ������п����õ�����Һ��Ϊָʾ��
c���ζ���������ˮϴ�Ӻ����ֱ��װҺ
d����ƿ����Ҫ�ô���Һ��ϴ
e���ζ������У��۾�ע�ӵζ�����Һ��仯
f���ζ���ɫ�仯��30s����Һ���ָ�ԭ������ɫ�ٶ���
��4�����ζ�����������0.5000mol��L?1KI��Һ20.00mL��������ʯ�����İٷֺ���Ϊ____________��
���ɢ���������������ʯ������������Ļ�ѧʽΪ ��
��μ�����ҺA���Ƿ���Fe2+__________����ѡ����ĸ����
A.�ȼ�KSCN��Һ���ټ���ˮ B.��NaOH��Һ C.��K3[Fe(CN)6]
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
����������FeC2O4�������������Լ�����Ӱ���Լ����͵�ز�����������﮵����������������ڸ�������ʱ�����ܹ��ֽ�,��ȤС��Բ��������ķֽ���������ʵ���̽��������֪��CO�����Ȼ���[PdC12]��Һ��Ӧ���ɺ�ɫ���ٷۡ���
��1�������������ֽ�������������ͨ������ʯ��ˮ���Ȼ�����Һ���۲쵽����ʯ��ˮ����ǣ��Ȼ�����Һ���к�ɫ�������ɡ�˵������������� �����ѧʽ��
��2��̽�����������ֽ�õ��ĺ�ɫ�����������Ԫ�صĴ�����ʽ��
���������⡿
���������ֽ��õ��ĺ�ɫ������ʲô��
��������衿
����1�� ������2��FeO������3��FeO��Fe�Ļ���
��ʵ�鷽����
��ѡ�Լ������ᡢ��ˮ��CuSO4��Һ��KSCN��Һ������ˮ��
| ʵ�鲽�� | ʵ������ | ʵ����� |
| ����1�����Թ��м��������������ټ������� ������� | ����Һ��ɫ���Ըı䣬 ���к�ɫ�������ɡ� | ��Fe���ڡ� |
| ����2��������1�еõ�����Һ���ˣ���������ˮ������ϴ����ϴ��Һ����ɫ�� | | |
| ����3��ȡ����2�õ��������������Թ��У��μӹ������ᣬ���ú�ȡ�ϲ���Һ�� �� | �� | ��FeO���ڡ� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ijУ�о���ѧϰС���ͬѧ���������»�ѧʵ�飺�����������ڿ�����ȼ�գ�Ȼ�������ù�������м���һ��������ˮ���˹����з�Ӧ�ų��������ȣ����ҷų��г�ζ�����塣
��1����ͬѧ������������ѧϰ��˼�룬Ca��Mg��ͬһ����Ԫ�أ���ѧ���ʾ���һ���������ԡ�
��д��Ca�ڿ�����ȼ�շ�����Ӧ�Ļ�ѧ����ʽ�� ��
��2����ͬѧ�����Ca�����ʱ�Na���ã��ڿ�����ȼ�ջ�Ӧ��CaO2���ɣ���д��ȼ�պ���������ˮ��Ӧ�ų�����Ļ�ѧ����ʽ�� ��
��ͬѧ�����ʵ��ķ���̽���ų���ζ����ijɷݣ�
���������ϡ�1��CaO2��ˮ��Ӧ����H2O2��H2O2���ܻ�ֽ����һ������O3
2������������õij����ⶨ��������ԭ��Ϊǿ������������O3����⻯�أ�KI��ˮ��Һ��Ӧ��������⣨I2����������ԭΪ��������ӦʽΪ��O3+2KI+H2O��O2+I2+2KOH ��������衿����1���ó�ζ����ֻ��NH3��
����2���ó�ζ����ֻ�� ��
����3���ó�ζ���庬�� ��
����Ʒ�������ʵ��̽����
��3�����ڼ���l����Сͬѧ���������ʵ�鷽����������ʵ�顣���ڴ���ϰ��±���ʽ�����ص�ʵ��������衢Ԥ�������ۣ�������ѡ����
����ѡʵ���Լ�������ɫʯ����ֽ����ɫʯ����ֽ��pH��ֽ������һKI��Һ������ˮ�����ʵ�鷽��֤����ļ��裬�����±���ʽд��ʵ��������衢Ԥ������ͽ��ۡ�
| ʵ����� | Ԥ������ͽ��� |
| ȡ������Ӧ��������Թ��У� �� | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
I������ռ������Ҫ�Ļ���ԭ�ϡ�
��1��������ͼ��ʾװ�ÿɼ��֤��������̼���ռ���Һ�����˷�Ӧ����A��B���ӣ���ֹˮ�У�����ͷ�ι��е�Һ�強����ƿ����ʱ��ʵ��������___________________��
�������������䣬��A��C���ӣ��ɹ۲쵽��������__________________________��
��2����NaOH��Һ��ͨ��һ����CO2���ᾧ��õ���ɫ���壬�ð�ɫ�������ɿ����ǣ�
A��NaOH��Na2CO3��B����������������C������������������D����������������
��3�����ʵ��ȷ����2���а�ɫ�����д���A���е������ӣ�
| ʵ����� | ʵ������ | ���� |
| ��ȡ������ɫ�������Թ��У�������ˮ�ܽ⣬�ټ�����BaCl2��Һ | | |
| �� | | |

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ijУ����С��Ϊ�ⶨij̼���ƺ�̼�����ƻ������̼���Ƶ������������ס�������ͬ
ѧ�ֱ�������������ʵ�顣
��������ͬѧ�ó�����������������ͼ��ʾ��ʵ�����̽���ʵ�飺[��֪Ba(HCO3)2������ˮ]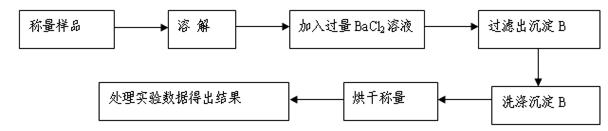
��1��ʵ��ʱ�����˲����У����˲�������©���⣬��Ҫ�õ��IJ�������Ϊ ��
��2��ϴ�ӳ���B�IJ����� ��
��3����ʵ���в����Ʒ����Ϊm g����������Ϊn g����̼���Ƶ���������Ϊ____________��
����������ͬѧ����Ҫʵ������ͼ���£�
������ͼ��ʾװ�ý���ʵ�飺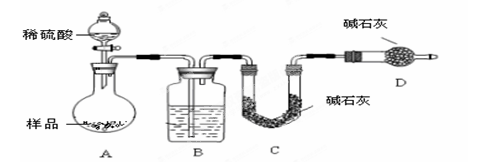
��4����ʵ����װ��Bʢ�ŵ�������_____________________����Һ©���� ����ܡ����ܡ������������ϡ�������ʵ�顣
��5����C��װ��ʯ�������վ���������塣
����Ʒ��̼���Ƶ���������Խ����ʵ�������վ���������ĸ�����ڳ����������ǰ���������
____________________�����Խ����ԽС�����仯����
��Dװ�õ�������_________________________��
��6���е�ͬѧ��ΪΪ�˼���ʵ�����ڷ�Ӧǰ��Ҫͨ��N2������ͼ������Ӧ��ͨ��N2��Ŀ����____
__________________________________________________________________________________��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com