
����Ŀ��25 ��ʱ����������ĵ��볣�����£�
���ữѧʽ | HNO2 | CH3COOH | HCN | H2CO3 |
���볣�� | 5.1��10-4 | 1.8��10-5 | 6.2��10-10 | K1=4.4��10-7 K2=4.7��10-11 |
��1�������ϱ�������գ�
�����ʵ���Ũ����ͬ�������ᣬ��pH�ɴ�С��˳����___________ ���û�ѧʽ��д����
�ڷֱ�����������ͬpH��HCl��Һ��CH3COOH��Һ�м���������Zn�ۣ���Ӧ�տ�ʼʱ����H2�����ʣ�v(HCl)______v(CH3COOH)���������������������ͬ������Ӧ��ȫ������������������m(H2)����_______m(H2)������
�۽�0.2 mol/L HCN��Һ��0.1 mol/L Na2CO3��Һ�������ϣ�������Ӧ�Ļ�ѧ����ʽΪ___________________________��
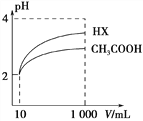
��2�������Ϊ10 mL��pH��Ϊ2�Ĵ�����Һ��һԪ��HX�ֱ��ˮϡ����1000 mL��ϡ��������ҺpH�仯��ͼ��ʾ��ϡ�ͺ�HX��Һ��ˮ�����c(H��)___������Һ��ˮ�����c(H��) �����볣��Ka(HX)___Ka(CH3COOH)�����������������������
��3��CO32- �� NO2- ��CN-��CH3COO-���H+��������ǿ������˳��Ϊ_______________________�������ӷ��ű�ʾ��
���𰸡� HCN > H2CO3 > CH3COOH > HNO2 = < HCN + Na2CO3 = NaCN + NaHCO3 > > CO32- > CN- > CH3COO- > NO2-
����������1�����ݵ���ƽ�ⳣ���Ĵ�С����������ƽ�ⳣ��Խ��������Խǿ�����ԣ�HCN��H2CO3��CH3COOH��HNO2������Խǿ����pHԽС������pH��ϵΪ��HCN��H2CO3��CH3COOH��HNO2��
��pH��ͬ�IJ�ͬ�����У�������Ũ����ͬ����Zn��Ӧ������ͬ�����������Ũ�ȴ���������Ũ�ȣ����������Ũ�ȵ���������Ũ�ȣ����Դ����Ũ�ȴ���HCl��Ũ�ȣ���������������������m��H2��������m��H2��������
���ɵ��볣����֪�����ԣ�H2CO3��HCN��CO3-����0.2 mol/L HCN��Һ��0.1 mol/L Na2CO3��Һ�������ϣ�������Ӧ�Ļ�ѧ����ʽΪHCN+Na2CO3�TNaCN+NaHCO3��
��2����ͼ��֪��ϡ�ͺ�HX�������ɵ�c��H+��С����ˮ�ĵ�����������С������HX��Һ��ˮ���������c��H+����ϡ����ͬ��������ǿ����pH�仯�ϴ�ǿ������볣���ϴ���ͼ��֪ϡ����ͬ�ı�����HX��pH�仯�̶ȴ�������HXǿ������ƽ�ⳣ����
��3����Խ�����������Խ��ˮ�⣬��������ӵ�����Խǿ����CO32- �� NO2- ��CN-��CH3COO-���H+��������ǿ������˳��ΪCO32-��CN-�� CH3COO-��NO2-��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ������������ԭ��Ӧ������˵������ȷ���ǣ�������
A.������Ӧ�ͻ�ԭ��Ӧһ��ͬʱ����
B.һ���е��ʲμӷ�Ӧ
C.��Ӧǰ��һ����Ԫ�صĻ��ϼ۷����仯
D.������һ���Ƿ�Ӧ�����еõ����ӵ�����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��Ӱ�컯ѧ��Ӧ���ʵ����غܶ࣬ij������ȤС����ʵ�鷽������̽����
��1��ȡ�����ʵ���Ũ�ȡ��������H2O2��Һ�ֱ����H2O2�ķֽ�ʵ�飬ʵ�鱨�����±���ʾ������ͽ����ԣ���
��� | �¶�/�� | ���� | ���� | ���� |
1 | 40 | FeCl3��Һ | ||
2 | 20 | FeCl3��Һ | ||
3 | 20 | MnO2 | ||
4 | 20 | �� |
��ʵ��1��2�о�����________________��H2O2�ֽ����ʵ�Ӱ�졣
��ʵ��2��3��Ŀ����_______________��
��2��������֪��Cu2����H2O2�ֽ�Ҳ�д����ã�Ϊ�Ƚ�Fe3����Cu2����H2O2�ֽ�Ĵ�Ч������С���ͬѧ�ֱ��������ͼ�ס�����ʾ��ʵ�顣�ش�������⣺

�ٶ��Է�������ͼ��ͨ���۲�________________�����ԱȽϵó����ۡ���ͬѧ�����CuSO4��Һ��ΪCuCl2��Һ����������������_______________��
�ڶ�����������ͼ����ʾ��ʵ��ʱ���ռ���40 mL����Ϊ��������������Ӱ��ʵ������أ�ʵ������Ҫ������������_____________��
��3�����Ը��������Һ�Ͳ�����Һ�ɷ�����Ӧ��2KMnO4��5H2C2O4��3H2SO4=K2SO4��2MnSO4��8H2O��10CO2����ʵ��ʱ���ֿ�ʼ��Ӧ���ʽ�������Һ��ɫ�����ԣ���һ��ʱ���ͻȻ��ɫ����Ӧ�������Լӿ졣�Դ�չ�����ۣ�
��ijͬѧ��ΪKMnO4��H2C2O4�ķ�Ӧ�� �ȷ�Ӧ������_______________��
�ڴ�Ӱ�컯ѧ��Ӧ���ʵ����ؿ�������Ϊ��������____________________��Ӱ�졣Ҫ֤����IJ��룬ʵ�鷽���� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ������˵����ȷ����
A. ʯ�ͷ�����Ի����ϩ
B. ú����������Һ���������仯��ת��Ϊ�[��ȼ��
C. ����ϩ������Ʒ������ʳƷ��װ
D. ��Ȼ����Һ��������Ҫ�ɷֶ��Ǽ���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��A��B��D��E��G��M����Ԫ��λ��Ԫ�����ڱ�ǰ�����ڣ�ԭ�����������������У�Ԫ��A��һ�ֺ��������ӣ�B�ĵ��ʼ��з��Ӿ�������ԭ�Ӿ��壬������DE2Ϊ����ɫ���壬G��ǰ�������е縺����С��Ԫ�أ�M��ԭ�Ӻ����������G��10��
��ش��������⣺
��1����̬Gԭ�ӵĺ�������Ų�ʽ��________��M��Ԫ�����ڱ��е�λ����_______��Ԫ��B��D��E�ĵ�һ�������ɴ�С��˳��Ϊ______________����Ԫ�ط��ű�ʾ����
��2��Ԫ��A��E��ɵ������ӿռ乹��Ϊ________��������ABD�ĽṹʽΪ______������Bԭ�ӵ��ӻ���ʽΪ________��
<span style="font-size: 15px; font-family: "����";"><span contenteditable="true">��3��</span></span>D������������Ӧ��ˮ�����������DA3�����������ӻ������ң������£����ס�������Һ��pH������5������ˮ�������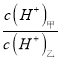 =_________������Һ���������ӵ����ʵ���Ũ���ɴ�С��˳����_______________________________��
=_________������Һ���������ӵ����ʵ���Ũ���ɴ�С��˳����_______________________________��
��4��Ԫ��Xλ�ڵ������ڣ����̬ԭ�ӵ��ڲ���ȫ���������ӣ�������������Ϊ2��Ԫ��Y��̬ԭ�ӵ�3p�������4�����ӡ�
��X��Y���γɻ�������X�Ļ��ϼ۵�������������Y�ﵽ8���ӵ��ȶ��ṹ��û�����Ļ�ѧʽΪ______________��
��E���⻯�H2E�����Ҵ��е��ܽ�ȴ���H2Y����ԭ����_________________��
��X���Ȼ����백ˮ��Ӧ���γ������[X(NH3)4]Cl2��1mol��������к��Ц� ������ĿΪ___________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ������Ȳ��������ȩ��ɵĻ������ⶨ����̼Ԫ����������Ϊ36%������������Ԫ�ص���������Ϊ
A. 44% B. 54. 22% C. 39.12% D. ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�������£���1 mol Na2CO3��10H2O(s)����ˮ��ʹ��Һ�¶Ƚ��ͣ���ЧӦΪ��H1����1 mol Na2 CO3(s)����ˮ��ʹ��Һ�¶����ߣ���ЧӦΪ��H2��Na2CO3��10H2O���ȷֽ�Ļ�ѧ����ʽΪNa2CO3��10H2O (s) ![]() Na2CO3(s)+10H2O(1)����ЧӦΪ��H3���������ж���ȷ���ǣ� ��
Na2CO3(s)+10H2O(1)����ЧӦΪ��H3���������ж���ȷ���ǣ� ��
A����H2 <��H3 B����H1>��H3 C����Hl+��H3=��H2 D����H1+��H2>��H3
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��������Ԫ��A��B��C��D��E��ԭ����������������֪��
��C�ļ���̬�⻯����ʪ��ĺ�ɫʯ����ֽ����ɫ��
��A��D�����γ�ԭ�Ӹ����ȷֱ�Ϊ2��1��1��1������Һ̬������X��Y��A��Eͬ���壬C��B��D���ڣ�
��A��B��C��D����Ԫ�����һ�ֻ�����F�������ԭ����֮��Ϊ5��1��1��3��
��ش��������⣺
(1)Cԭ�������ڱ���λ��Ϊ_____________��D��ԭ�ӽṹʾ��ͼΪ_____________��д��������EDA�ĵ���ʽ__________��
(2)C������������Ӧ��ˮ����������̬�⻯�ﷴӦ�������Σ�д���仯ѧʽ___________��д��Y�Ľṹʽ_____________��
(3)F��Һ������EDA��ϡ��Һ��ϣ������ӷ���ʽΪ_______________________��
(4)������C�ļ���̬�⻯�������а��̼�C2���ɣ�д����Ӧ����ʽ______________��
(5)��CH4������������β���е����������Ⱦ��
��֪��CH4(g)��2NO2(g)![]() N2(g)��CO2(g)��2H2O(l) ��H����955 kJ��mol��1
N2(g)��CO2(g)��2H2O(l) ��H����955 kJ��mol��1
2NO2(g)![]() N2O4(g)����H����56.9 kJ��mol��1
N2O4(g)����H����56.9 kJ��mol��1
д��CH4����ԭN2O4(g)����N2��H2O(l)���Ȼ�ѧ����ʽ��____________________ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����1������п������ϡ������ɵ�ԭ���װ�ã�����������2mol����ͨ��ʱ��пƬ�ܽ���______________g ����Ƭ������___________�� H2����״���£���
����ܷ�Ӧ�Ļ�ѧ����ʽΪ______________��
��2����������������ɴ���ʹ����һ������װ�ã��乹������ͼ��ʾ��

A��B�����缫���ɶ��̼����ɡ�
�õ�ص�������ӦʽΪ��____________________�����õ�ع���ʱ������1mol H2O����·��ת�Ƶ��ӵ����ʵ���Ϊ_______________��
��3�����������װ����ͨ���H2�ij�CH4���壬Ҳ�������һ��ԭ���װ�ã���õ�صĸ�����ӦʽΪ��____________��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com