
��������(CeO2)��һ����Ҫ��ϡ�������ƽ�������ʾ�����������в��������ķϲ�����ĩ(��SiO2��Fe2O3��CeO2�Լ���������������ϡ�������)��ij�������Դ˷�ĩΪԭ�ϻ����棬���ʵ����������: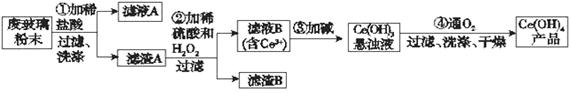
��1��ϴ������A��Ŀ����Ϊ�˳�ȥ____________�������ӷ��ţ�������������Ƿ�ϴ���ķ�����_________________________________________________________��
��2���ڢڲ���Ӧ�����ӷ���ʽ��______________________________________������B����Ҫ�ɷ���___________________��
��3����ȡ�Ƿ���ϡ��Ԫ�صij��÷�������֪������TBP��Ϊ��ȡ���ܽ������Ӵ�ˮ��Һ����ȡ������TBP______________����ܡ����ܡ�����ˮ���ܡ�ʵ���ҽ�����ȡ����ʱ�õ�����Ҫ����������_______________���ձ�������������Ͳ�ȡ�
��4��ȡ���������еõ���Ce(OH)4��Ʒ0.536 g���������ܽ����0.100 0 mol��L��1 FeSO4����Һ�ζ����յ�ʱ���汻��ԭΪCe3����������25.00 mL����Һ���ò�Ʒ��Ce(OH)4����������Ϊ___________�����������λ��Ч���֣���
(1) (��4��) Fe3��(1��)��ȡ���һ��ϴ��Һ(1��)������KSCN��Һ(1��)���������ֺ�ɫ������ϴ��(1��)����֮��δϴ��(���������𰸾���)
(2) (��4��) 2CeO2��H2O2��6H��=2Ce3����O2����4H2O(3��) ����ƽ����2�֡�û�С���1�֣��� SiO2(1��) (3)���ܡ���Һ©��(��2��) (4)97%��0.97( 4��)
���������������1���ϲ�����ĩ�к��еĶ�������Ͷ������趼����������ģ�����ϡ����Ŀ�����ܽ������������ʣ����Եõ���������Ҫ��SiO2��CeO2 ������������ϴ�Ӿ���Ҫϴȥ�����Ŀ����Ե�Fe3�� ���ʣ����Լ���������Ƿ�ϴ����Ӧ��ȡ���һ��ϴ��Һ������KSCN�Լ�����������Ѫ��ɫ������֤���Ѿ�ϴ�Ӹɾ�������δϴ����
��2������A�еĶ��������CeΪ+4�ۣ�������ϡ�����H2O2����CeO2 ���ܽ⣬�ұ���ԭΪ��+3�۵�Ce3+ �����Կ��Կ��������˫��ˮ����ԭ�������������·���������ԭ��Ӧ������ʽΪ��2CeO2��H2O2��6H��=2Ce3����O2����4H2O�����Եõ�������B��SiO2��
��3��������TBP��Ϊ��ȡ���ܽ������Ӵ�ˮ��Һ����ȡ���������Ը���ȡ��������ˮ�������ݣ�����ȡ��Һ�����б���Ҫ�õ���Һ©����
��4��Ce(OH)4��Ʒ���������ܽ��������Ce4�� �������û�ԭ���ζ�����Һ��Ce4�� �����Ce3�� ����Fe2+ ����������Fe3+ �����Ը��ݵ��ӵ����غ���㣬n(Ce(OH)4)=n(FeSO4)="cV=0.100" 0 mol��L��1��25.00 ��10��3 L=0.0025mol������Ce(OH)4����������W=nM/m��Ʒ��100%=0.0025mol��208g/mol / 0.536g=97%����Ҫע�Ᵽ����λ��Ч���֣���
���㣺���⿼����ǻ�ѧ���������⡣



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�ȣ�Sr��Ϊ�������ڢ�A��Ԫ�أ��仯������ˮ�Ȼ��ȣ�SrCl2��6H2O����ʵ������Ҫ�ķ����Լ�����ҵ�ϳ�������ʯ����Ҫ�ɷ�ΪSrSO4��Ϊԭ���Ʊ��������������£�
��֪���� �������ȡ����Һ�г�����Sr2+��Cl���⣬����������Ba2+���ʣ�
�� SrSO4��BaSO4���ܶȻ������ֱ�Ϊ3.3��10��7��1.1��10��10��
�� SrCl2��6H2O��Ħ������Ϊ��267 g/mol��
��1������ʯ����ǰ����ĥ���飬��Ŀ����_________________________________________��
��2�������������±��գ���0.5 mol SrSO4��ֻ��S����ԭ��ת����4 mol���ӡ�д���÷�Ӧ�Ļ�ѧ����ʽ��_____________________________________________��
��3��Ϊ�˵õ��ϴ�����ˮ�Ȼ��Ⱦ��壬����2������е�����������_______________��
��4�����������Ŀ����______________________��Ϊ�����ԭ�ϵ������ʣ���Һ��Sr2+��Ũ��Ӧ������_________ mol/L��ע����ʱ��Һ��Ba2+Ũ��Ϊ1��10��5 mol/L����
��5����Ʒ���ȼ�⣺��ȡ1.000 g��Ʒ�ܽ�������ˮ�У������м��뺬AgNO3 1.100��10��2 mol��AgNO3��Һ����Һ�г�Cl�D�⣬����������Ag+��Ӧ�����ӣ�����Cl�D��ȫ�������ú�Fe3+����Һ��ָʾ������0.2000 mol/L��NH4SCN����Һ�ζ�ʣ���AgNO3��ʹʣ���Ag+ ��AgSCN��ɫ��������ʽ������
�ٵζ���Ӧ�ﵽ�յ��������_________________________________________��
�����ζ�������ȥ����Ũ�ȵ�NH4SCN��Һ20.00 mL�����Ʒ��SrCl2��6H2O�������ٷֺ���Ϊ______________������4λ��Ч���֣���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
���Ͳ�������Fe�۾��г�ǿ�Ĵ��ԣ����������ܶȴż�¼�Ľ����Լ���Ч �����ȡ�
I��ʵ���Ҳ������ԭ���Ʊ�����Fe,���������£�
��1�� ���������ˮͨ��Ҫͨ�� ��
���������ˮͨ��Ҫͨ�� ��
��2����������Fe�Ļ�ѧ����ʽΪ ��
���ڲ�ͬ�¶��£�����Fe����ˮ������Ӧ�Ĺ�����ﲻͬ���¶ȵ���570��ʱ����FeO������570��ʱ���� ����ͬѧ����ͼ��ʾװ�ý�������Fe����ˮ�����ķ�Ӧ����֤������
����ͬѧ����ͼ��ʾװ�ý�������Fe����ˮ�����ķ�Ӧ����֤������
��3��B���ռ����������� (������)��Cװ�õ������� ��
��4����ͬѧΪ̽��ʵ��������Թ��ڵĹ������ʳɷ֣�����������ʵ�飺
��ͬѧ��Ϊ�������·�Ӧ�Ĺ������ΪFeO����ͬѧ��Ϊ�ý��۲���ȷ������������ (�����ӷ���ʽ��ʾ)��
��5����ͬѧ��ȡ5��60gFe�ۣ���Ӧһ��ʱ���ֹͣ���ȡ����Թ��ڵĹ��������ڸ���������ȴ�Ƶ�����Ϊ6��88g��Ȼ����ȴ��Ĺ�������������FeCl3��Һ��ַ�Ӧ������0��08molFeCl3����ͬѧʵ��Ĺ������Ϊ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
±�����Ҫ�ɷ���MgCl2������� Fe3+��Fe2+��Mn2+�����ӡ���±��Ϊԭ�Ͽ��Ƶ���������þ��������������ͼ��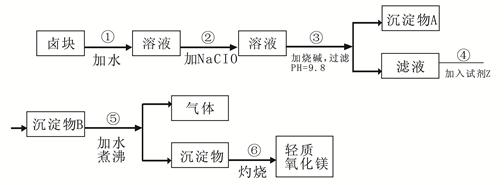
��֪��Fe2+�����������״�����״���Һ�г�ȥ�����Գ���������ΪFe3+������Fe(OH)3������ȥ����Ҫ���Ʒ�����������ʣ�����ݱ�1��2�ṩ�����ϣ���д�հף�
��1 �����������������pH
| ���� | ��ʼ���� | ������ȫ |
| Fe��OH��3 | 2.7 | 3.7 |
| Fe��OH��2 | 7.6 | 9.6 |
| Mn��OH��2 | 8.3 | 9.8 |
| Mg��OH��2 | 9.6 | 11.1 |
| �Լ� | �۸�Ԫ/�֣� |
| ƯҺ����NaClO��25.2%�� | 450 |
| ˫��ˮ����H2O2 ,30%�� | 2400 |
| �ռ��98% NaOH�� | 2100 |
| �����99.5% Na2CO3�� | 600 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�ú���Al2O3��SiO2������FeO��xFe2O3�������Ʊ�Al2(SO4)3��18H2O�������������£�
��.�������м������ϡH2SO4�����ˣ�
��.����Һ�м������KMnO4��Һ��������Һ��pHԼΪ3��
��.���ȣ�����������ɫ���������ã��ϲ���Һ���Ϻ�ɫ��
��.����MnSO4���Ϻ�ɫ��ʧ�����ˣ�
��.Ũ�����ᾧ�����룬�õ���Ʒ��
(1)H2SO4�ܽ�Al2O3�����ӷ���ʽ��___________________________________
_________________________��
(2)��MnO4������Fe2�������ӷ���ʽ����������42��
(3)��֪�������������������pH
| | Al(OH)3 | Fe(OH)2 | Fe(OH)3 |
| ��ʼ����ʱ | 3.4 | 6.3 | 1.5 |
| ��ȫ����ʱ | 4.7 | 8.3 | 2.8 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�������(K2FeO4)�����Ͷ��ˮ�����������������������ȶ����������������£�
��ش���������
��1���ȼҵ��Cl2�Ļ�ѧ��Ӧ����ʽ ��
��2�����ɡ���ӦҺ�����ӷ���ʽ�� ����3��ʹ�ü���KClO��ԭ���� ��
��4���ӡ���ӦҺII���з����K2FeO4�� ������Ʒ��KCl�� (�ѧʽ)���û������� ���������ᴿ������ĸ��ţ���
| A������ | B����Һ | C������ | D���ؽᾧ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�췯��(�ظ����ƣ�Na2Cr2O7��2H2O)����Ҫ�Ļ�������ԭ�ϣ���ӡȾ��ҵ����ƹ�ҵ��Ƥ�﹤ҵ�������������ڻ�ѧ��ҵ����ҩ��ҵ��Ҳ������������Ӧ������ʮ�ֹ㷺��ʵ�����к췯�ƿ���һ�ָ�����(��Ҫ�ɷ֣�FeO��Cr2O3��������������Al2O3)ͨ�����¹�������ȡ��
�ش��������⣺
(1)������з�Ӧ��ѧ����ʽΪ4FeO2��Cr2O3(s)��8Na2CO3(s)��7O2(g)=8Na2CrO4(s)��2Fe2O3(s)��8CO2(g)��
����ʵ���ң������Ӧ��________(��մɡ�������)��������ɡ�
���ڳ����¸÷�Ӧ���ʼ��������д�ʩ����ʹ��Ӧ�����������________(����ĸ)��
A����ԭ�Ϸ��顡B�����Ӵ����������C�������¶�
(2)�������NaOH��������________________________(�û�ѧ��Ӧ����ʽ��ʾ)��
(3)������з�����Ӧ�����ӷ���ʽΪ________����Ӧ��ɵı�־��________��
(4)��������ĸ��ֽⷴӦ�����췯����KCl���尴1?2���ʵ���֮�Ȼ������ˮ���ʵ������ɵõ�K2Cr2O7���壺Na2Cr2O7��2KCl=K2Cr2O7��2NaCl(��֪���¶ȶ��Ȼ��Ƶ��ܽ��Ӱ���С�����ظ���ص��ܽ��Ӱ��ϴ�)
����ʵ�鲽��Ϊ���ܽ⣻��________����________������ȴ���ᾧ���ٹ��˵�K2Cr2O7���塣���Т�Ӧ��________(����¡����¡�)�����½��С�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
������һ���ḻ����Դ���⣬ͨ����ˮ���ۺ����ÿɻ���������ʹ�����ʹ�á�
��1�� ��ˮ���εĿ������ã�
��.��ˮ����Ŀǰ�����Ϊ�������������ѡ��Զ�뽭���뺣�ڣ�������꣬��ϫ��������ƽ̹�տ��ĺ�̲�����������Ϊ��ˮ�ء������غ�_______�ء�
II.Ŀǰ��ҵ�ϲ��ñȽ��Ƚ������ӽ���Ĥ���۷������ȼҵ�������ڵ����������ӽ���Ĥֻ����������ͨ������ֹ�����Ӻ�����ͨ������˵���ȼ������������ӽ���Ĥ������____________________________________________����дһ�㼴�ɣ�
��2�����������ǽ�������չ������һ�ֽϺõĺ�ˮ������������ԭ������ͼ��ʾ����ش��������⣺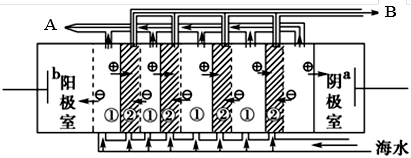
��.��ˮ����ֱ��ͨ�뵽��װ���У�������_____________________________________________��
��. B���ų�����________(���ˮ����Ũˮ��)��
��3���ÿ�±����Na+��K+��Mg2+��Cl-��Br-�����ӣ�����ȡ�壬�������������£�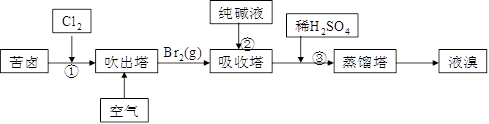
��.���������е���Һ��BrO3�������������з�Ӧ�����ӷ���ʽΪ�ߣߣߣߣߣߣߣߣߣߣߣߣ�
_________________________________________��
��.ͨ�����Ȼ��ѻ�ú�Br2����Һ��Ϊ�λ��辭�����������ա��ữ�����»�ú�Br2����Һ��_____________________________________________________________________��
��.����������ͨ��ˮ�������ȣ������¶���900C���ҽ��������ԭ����___________________________________________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
������������;�㷺����ҵ�ϳ��ú�����Al2O3�ķ�����FeO��V2O5�����۷���ȡV2O5����Ҫ�������£�
��֪���ٱ���ʱ�ɷ�����Ӧ��V2O5 + Al2O3+2Na2CO3 2NaVO3 +2NaAlO2 +2CO2
2NaVO3 +2NaAlO2 +2CO2
�ڳ��������ʵ��ܽ�ȣ�NaVO3��21��2 g /100gˮ��HVO3��0��008 g /100gˮ
��1����������B������Ҫ�ɷ��� ����д��ѧʽ��
��2�������У���ֱ����H2SO4���ݡ�����A����ȡHVO3��ԭ���� ��
��3���������١����� ��ϴ�ӡ������ϴ�ӣ����Ʒ�п��ܺ��еĽ����������� �� ������װ�ã����ּг�����ʡȥ��������ʵ���ҽ��С������ڡ����� ��������ţ�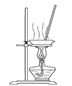

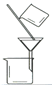
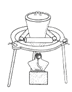
A B C D
��4��NaVO3����ԭ�͵�����������V2O5����NaOH��Һ����ȡ����Ӧ�����ӷ���ʽΪ ��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com