
≥―Έ÷–Κ§”–“ΜΕ®ΝΩΒΡΟΨΓΔΧζΒ»‘”÷ Θ§Φ”Ββ―Έ÷–ΒβΒΡΥπ ß÷ς“Σ «”…”Ύ‘”÷ ΓΔΥ°Ζ÷ΓΔΩ’Τχ÷–ΒΡ―θΤχ“‘ΦΑΙβ’’ΓΔ ή»»Εχ“ΐΤπΒΡΓΘ
“―÷ΣΘΚΔΌ―θΜ·–‘ΘΚ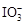 ΘΨFe3ΘΪΘΨI2ΘΜΜΙ‘≠–‘ΘΚ
ΘΨFe3ΘΪΘΨI2ΘΜΜΙ‘≠–‘ΘΚ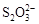 ΘΨIΘ≠ΘΜΔΎKIΘΪI2
ΘΨIΘ≠ΘΜΔΎKIΘΪI2 KI3
KI3
Θ®1Θ©Ρ≥―ßœΑ–ΓΉιΕ‘Φ”Ββ―ΈΫχ––»γœ¬ Β―ιΘΚ»Γ“ΜΕ®ΝΩΡ≥Φ”Ββ―Έ(Ω…ΡήΚ§”–KIO3ΓΔKIΓΔMg2ΘΪΓΔFe2ΘΪΓΔFe3ΘΪ)Θ§”Ο ΝΩ’τΝσΥ°»ήΫβΘ§≤ΔΦ”œΓ―ΈΥαΥαΜ·Θ§ΫΪΥυΒΟ»ή“ΚΖ÷ΈΣ4ΖίΓΘΒΎ“ΜΖί ‘“Κ÷–ΒΈΦ”KSCN»ή“ΚΚσœ‘Κλ…ΪΘΜΒΎΕΰΖί ‘“Κ÷–Φ”ΉψΝΩKIΙΧΧεΘ§»ή“Κœ‘Β≠ΜΤ…ΪΘ§”ΟCCl4ίΆ»ΓΘ§œ¬≤ψ»ή“Κœ‘ΉœΚλ…ΪΘΜΒΎ»ΐΖί ‘“Κ÷–Φ”»κ ΝΩKIO3ΙΧΧεΚσΘ§ΒΈΦ”ΒμΖέ ‘ΦΝΘ§»ή“Κ≤Μ±δ…ΪΓΘ
ΔΌœρΒΎΥΡΖί ‘“Κ÷–Φ”K3Fe(CN)6»ή“ΚΘ§ΗυΨί «ΖώΒΟΒΫΨΏ”–ΧΊ’ςάΕ…ΪΒΡ≥ΝΒμΘ§Ω…Φλ―ι «ΖώΚ§”– Θ®”ΟάκΉ”ΖϊΚ≈±μ ΨΘ©Θ§άΕ…ΪΒΡ≥ΝΒμ «_______Θ®”ΟΜ·―ß Ϋ±μ ΨΘ©ΓΘ
ΔΎΒΎΕΰΖί ‘“Κ÷–Φ”»κΉψΝΩKIΙΧΧεΚσΘ§Ζ¥”ΠΒΡάκΉ”ΖΫ≥Χ ΫΈΣΘΚ______ΓΔ______
Θ®2Θ©KIΉςΈΣΦ”ΒβΦΝΒΡ ≥―Έ‘Ύ±Θ¥φΙΐ≥Χ÷–Θ§”…”ΎΩ’Τχ÷–―θΤχΒΡΉς”ΟΘ§»ί“Ή“ΐΤπΒβΒΡΥπ ß–¥≥ω≥± ΣΜΖΨ≥œ¬KI”κ―θΤχΖ¥”ΠΒΡΜ·―ßΖΫ≥Χ ΫΘΚ_________________ΓΘ
ΫΪI2»ή”ΎKI»ή“ΚΘ§‘ΎΒΆΈ¬ΧθΦΰœ¬Θ§Ω…÷ΤΒΟKI3ΓΛH2OΓΘΗΟΈο÷ ≤Μ ΚœΉςΈΣ ≥―ΈΦ”ΒβΦΝΘ§Τδάμ”… «_________________________________ΓΘ
Θ®3Θ©Ρ≥Ά§―ßΈΣΧΫΨΩΈ¬Ε»ΚΆΖ¥”ΠΈο≈®Ε»Ε‘Ζ¥”Π2IO3Θ≠ΘΪ5SO32Θ≠ΘΪ2HΘΪ=I2ΘΪ5SO42Θ≠ΘΪH2OΒΡΥΌ¬ ΒΡ”ΑœλΘ§…ηΦΤ Β―ι»γœ¬±μΥυ ΨΘΚ
| | 0.01molΓΛLΘ≠1KIO3 Υα–‘»ή“Κ(Κ§ΒμΖέ) ΒΡΧεΜΐ/mL | 0.01molΓΛLΘ≠Na2SO3 »ή“ΚΒΡΧεΜΐ/mL | H2OΒΡ ΧεΜΐ/mL | Β―ι Έ¬Ε» /Γφ | »ή“Κ≥ωœ÷ άΕ…Ϊ ±Υυ –η ±Φδ/s |
| Β―ι1 | 5 | V1 | 35 | 25 | t1 |
| Β―ι2 | 5 | 5 | 40 | 25 | t2 |
| Β―ι3 | 5 | 5 | V2 | 0 | t3 |
Θ®Ι≤14Ζ÷Θ©
Θ®1Θ©ΔΌ Fe2ΘΪ Θ§ Fe 3[Fe(CN)6]2 Θ®Ης1Ζ÷Θ©
ΔΎIO3Θ≠ΘΪ5IΘ≠ΘΪ6HΘΪ=3I2ΘΪ3H2O 2Fe3ΘΪΘΪ2IΘ≠=2Fe2ΘΪΘΪI2 Θ®Ης2Ζ÷Θ©
Θ®2Θ©O2ΘΪ4KIΘΪ2H2O=2I2ΘΪ4KOH Θ®2Ζ÷Θ©
KI3‘Ύ ή»»Θ®Μρ≥± ΣΘ©ΧθΦΰœ¬≤ζ…ζI2ΚΆKIΘ§KI±Μ―θΤχ―θΜ·Θ§I2“Ή…ΐΜΣΓΘΘ®2Ζ÷Θ©
Θ®3Θ©< ΘΜ 40 Θ®Ης2Ζ÷Θ©
ΫβΈω ‘ΧβΖ÷ΈωΘΚ
Θ®1Θ©ΔΌFe2ΘΪ”κΧζ«ηΜ·ΦΊ»ή“Κ”–ΧΊ’ςΖ¥”Πœ÷œσΓΘ
ΔΎ”Π”Ο–≈œΔΘΚΓΑ―θΜ·–‘ΘΚIO3Θ≠ΘΨFe3ΘΪΘΨI2Γ±Θ§ΥΒΟςIO3Θ≠ΚΆFe3ΘΪΨυΡή―θΜ·IΘ≠…ζ≥…I2ΓΘ
Θ®2Θ©KI±Μ≥± ΣΩ’Τχ―θΜ·Εχ“ΐΤπΥπ ßΘ§Υυ“‘ΩΦ¬«IΘ≠±Μ―θΤχ―θΜ·–Έ≥…I2…ΐΜΣ¥”Εχ‘λ≥…Υπ ßΓΘ
ΗυΨί“―÷Σ–≈œΔΘΚΓΑKIΘΪI2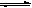 KI3Γ±ΫβΈωΤδ–‘÷ ≤ΜΈ»Ε®–‘Θ§“ΉΖΔ…ζΖ÷ΫβΙΐ≥ΧΓΘ
KI3Γ±ΫβΈωΤδ–‘÷ ≤ΜΈ»Ε®–‘Θ§“ΉΖΔ…ζΖ÷ΫβΙΐ≥ΧΓΘ
Θ®3Θ©”…±μΗώ–≈œΔΩ…÷ΣΘ§»ΐΉι Β―ιΈΣΤΫ––Ε‘’’ Β―ιΘ§“ΣΉώ―≠ΓΑΒΞ“Μ±δΝΩΓ±ΒΡΙφ‘ρΘ§“ρ¥Υ’β»ΐΉι Β―ι»ή“ΚΒΡΉήΧεΜΐΕΦΒ»”Ύ50 mlΘ§“ρ¥ΥV2="40" mlΓΘ Β―ι1ΚΆ Β―ι2±»ΫœΘ§ Β―ι2÷–Υ°ΒΡΧεΜΐΕύΘ§ΥΒΟς»ή÷ ≈®Ε»±» Β―ι1÷–ΒΡ“Σ–ΓΘ§Ζ¥”ΠΥΌ¬ ¬ΐΘ§Υυ”Ο ±Φδ≥ΛΘ§Υυ“‘t1ΘΦt2
ΩΦΒψΕ®ΈΜΘΚΩΦ≤ιΝΥάκΉ”ΒΡΦλ―ιΘ§―θΜ·ΜΙ‘≠Ζ¥”ΠΒΡ ι–¥Θ§ Β―ι…ηΦΤ ΐΨίΖ÷ΈωΒ»œύΙΊ÷Σ ΕΘ§≈ύ―χ―ß…ζΧβΗ…–≈œΔΒΡΕΝ»ΓΚΆΖ÷ΈωΒΡΡήΝΠΓΘ



| ΡξΦΕ | ΗΏ÷–ΩΈ≥Χ | ΡξΦΕ | ≥θ÷–ΩΈ≥Χ |
| ΗΏ“Μ | ΗΏ“ΜΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ | ≥θ“Μ | ≥θ“ΜΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ |
| ΗΏΕΰ | ΗΏΕΰΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ | ≥θΕΰ | ≥θΕΰΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ |
| ΗΏ»ΐ | ΗΏ»ΐΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ | ≥θ»ΐ | ≥θ»ΐΟβΖ―ΩΈ≥ΧΆΤΦωΘΓ |
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ Χβ–ΆΘΚΧνΩ’Χβ
–¥≥ωœ¬Ν–Ζ¥”ΠΒΡΜ·―ßΖΫ≥Χ ΫΘ§≤Δ÷Η≥ω―θΜ·ΦΝΚΆΜΙ‘≠ΦΝΓΘ
(1)ο°‘ΎΩ’Τχ÷–»Φ…’
(2)ΦΊ”κΥ°Ζ¥”Π
(3)ΟΨΧθ‘ΎΕΰ―θΜ·ΧΦΤχΧε÷–»Φ…’
(4)‘χ«ύΒΟΧζ‘ρΜ·ΈΣΆ≠ΓΣΓΣ ΣΖ®ΝΕΆ≠
(5)¥”≥ύΧζΩσ÷–ΧαΝΕΧζΓΣΓΣΙΛ“Β“±Χζ
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ Χβ–ΆΘΚ Β―ιΧβ
Ρ≥ΩΈΆβ–ΓΉι―ß…ζΕ‘Cl2”κFeBr2»ή“ΚΖ¥”ΠΫχ–– Β―ιΧΫΨΩΓΘ
| ≤Ό Ής | œ÷ œσ |
| »Γ0Θ°1 mol/LΒΡFeBr2»ή“Κ10 mLΘ§≤β»ή“ΚΒΡpH | pH ‘÷Ϋ±δΚλΘ®‘ΦΈΣ4Θ© |
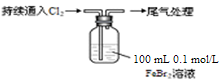 | ΔΓΘ°ΩΣ ΦΆ®»κ100 mLΘ®’έΚœ±ξΉΦΉ¥ΩωΘ©Θ§»ή“Κ”…«≥¬Χ…Ϊ÷πΫΞ±δΜΤΘΜ ΔΔΘ°…‘ΚσΘ§ΦΧ–χΆ®»κCl2Θ§»ή“ΚΜΤ…ΪΦ”…νΘ§±δΈΣΉΊΜΤ…ΪΘΜ iiiΘ°…‘ΚσΘ§»ή“Κ”…ΉΊΜΤ…Ϊ±δ«≥Θ§Ήν÷’±δΈΣΜΤ…Ϊ |
| ‘Ιή | ≤ΌΉς | œ÷œσ | Ϋα¬έ |
| a | »Γ2 mL FeCl2»ή“ΚΘ§ΒΈΦ”…ΌΝΩ¬»Υ°ΓΔ2ΒΈKSCN»ή“ΚΘ§’ώΒ¥ | | Cl2ΒΡ―θΜ·–‘«Ω”ΎFe3+ |
| b | »Γ2 mL FeCl3»ή“ΚΘ§ΒΈΦ”…ΌΝΩδεΜ·ΡΤ»ή“ΚΚΆ…ΌΝΩCCl4Θ§’ώΒ¥ | CCl4≤ψΟΜ”–Οςœ‘±δΜ· | |
| c | | CCl4≤ψ”…Έό…Ϊ±δΈΣ≥»ΜΤ…Ϊ | Cl2ΒΡ―θΜ·–‘«Ω”ΎBr2 |
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ Χβ–ΆΘΚ Β―ιΧβ
KMnO4»ή“Κ≥Θ”ΟΉς―θΜ·ΜΙ‘≠Ζ¥”ΠΒΈΕ®ΒΡ±ξΉΦ“ΚΘ§”…”ΎKMnO4ΒΡ«Ω―θΜ·–‘Θ§ΥϋΒΡ»ή“ΚΚή»ί“Ή±ΜΩ’Τχ÷–ΜρΥ°÷–ΒΡΡ≥–©…ΌΝΩΜΙ‘≠–‘Έο÷ ΜΙ‘≠Θ§…ζ≥…Ρ―»ή–‘Έο÷ MnO(OH)2Θ§“ρ¥Υ≈δ÷ΤKMnO4±ξΉΦ»ή“ΚΒΡ≤ΌΉς»γœ¬Υυ ΨΘΚΔΌ≥Τ»Γ…‘Εύ”ΎΥυ–ηΝΩΒΡKMnO4ΙΧΧε»ή”ΎΥ°÷–Θ§ΫΪ»ή“ΚΦ”»»≤Δ±Θ≥÷ΈΔΖ–1 hΘΜΔΎ”ΟΈΔΩΉ≤ΘΝ߬©ΕΖΙΐ¬Υ≥ΐ»ΞΡ―»ήΒΡMnO(OH)2ΘΜΔέΙΐ¬ΥΒΟΒΫΒΡKMnO4»ή“Κ÷ϋ¥φ”ΎΉΊ…Ϊ ‘ΦΝΤΩ≤ΔΖ≈‘ΎΑΒ¥ΠΘΜΔήάϊ”Ο―θΜ·ΜΙ‘≠ΒΈΕ®ΖΫΖ®Θ§”ΟΜυΉΦ ‘ΦΝ(¥ΩΕ»ΗΏΓΔœύΕ‘Ζ÷Ή”÷ ΝΩΫœ¥σΓΔΈ»Ε®–‘ΫœΚΟΒΡΈο÷ )»ή“Κ±ξΕ®Τδ≈®Ε»Θ§KMnO4‘ΎΒΈΕ®÷–±ΜΜΙ‘≠≥…Mn2+ΓΘ«κΜΊ¥πœ¬Ν–Έ ΧβΘΚ
Θ®1Θ© ΉΦ»ΖΝΩ»Γ“ΜΕ®ΧεΜΐΒΡKMnO4»ή“Κ–η“Σ Ι”ΟΒΡ“«Τς «____________ΓΘ
Θ®2Θ© ‘Ύœ¬Ν–Έο÷ ÷–Θ§”Ο”Ύ±ξΕ®KMnO4»ή“ΚΒΡΜυΉΦ ‘ΦΝΉνΚΟ―Γ”Ο________(Χν–ρΚ≈)ΓΘ
| AΘ°÷ ΝΩΖ÷ ΐΈΣ30%ΒΡΥΪ―θΥ° | BΘ°FeSO4 | CΘ°ΡΠΕϊ―Έ | DΘ°Na2SO3 |
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ Χβ–ΆΘΚ Β―ιΧβ
ΈΣ―ι÷Λ―θΜ·–‘ΘΚCl2ΘΨFe3+ΘΨSO2Θ§Ρ≥–ΓΉι”Οœ¬ΆΦΥυ ΨΉΑ÷ΟΫχ–– Β―ιΘ®Φ–≥÷“«ΤςΚΆA÷–ΒΡΦ”»»ΉΑ÷Ο“―¬‘Θ§ΤχΟή–‘“―Ψ≠Φλ―ιΆξ±œΘ©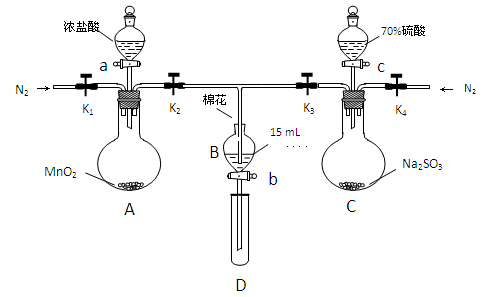
Β―ιΙΐ≥ΧΘΚ
ΔώΘ°¥ρΩΣΒ·Μ…Φ–K1-K4Θ§Ά®»κ“ΜΕΈ ±ΦδN2Θ§‘ΌΫΪT–ΆΒΦΙή≤ε»κB÷–Θ§ΦΧ–χΆ®»κN2Θ§ΒΞΚσΙΊ±’K1ΓΔK2ΓΔK3 .
ΔρΘ°¥ρΩΣΜν»ϊaΘ§ΒΈΦ”“ΜΕ®ΝΩΒΡ≈®―ΈΥαΘ§ΗχAΦ”»».
ΔσΘ°Β±B÷–ΒΡ»ή“Κ±δΜΤ ±Θ§ΆΘ÷ΙΦ”»»Θ§Φ–ΫτΒ·Μ…Φ–K2.
ΔτΘ°¥ρΩΣΜν»ϊbΘ§ Ι‘Φ2mlΒΡ»ή“ΚΝς»κD ‘Ιή÷–Θ§Φλ―ιΤδ÷–ΒΡάκΉ”.
ΔθΘ°¥ρΩΣΒ·Μ…Φ–K3ΓΔΜν»ϊcΘ§Φ”»κ70%ΒΡΝρΥαΘ§“ΜΕΈ ±ΦδΚσΦ–ΫτΒ·Μ…Φ–K3.
Δω.Ηϋ–¬ ‘ΙήDΘ§÷ΊΗ¥Ιΐ≥ΧΔωΘ§Φλ―ιB»ή“Κ÷–ΒΡάκΉ”.
Θ®1Θ©Ιΐ≥ΧΔώΒΡΡΩΒΡ «___________________________
Θ®2Θ©ΟόΜ®÷–Ϋΰ»σΒΡ»ή“ΚΈΣ_______________________
Θ®3Θ©A÷–ΖΔ…ζΖ¥”ΠΒΡΜ·―ßΖΫ≥Χ ΫΈΣ_____________________________________________
Θ®4Θ©”Ο70%ΒΡΝρΥα÷ß»ΓSO2Θ§Ζ¥”ΠΥΌ¬ ±»”Ο98%ΒΡΝρΥαΩιΘ§‘≠“ρ «___________________
Θ®5Θ©Ιΐ≥ΧΔτ÷–Φλ―ιB»ή“Κ÷– «ΖώΚ§”–ΝρΥαΗυΒΡ≤ΌΉς «______________________________
Θ®6Θ©ΦΉΓΔ““ΓΔ±ϊ»ΐΈΜΆ§―ßΖ÷±πΆξ≥…ΝΥ…œ ω Β―ιΘ§ΥϊΟ«ΒΡΦλ≤βΫαΙϊ“ΜΕ®ΡήΙΜ÷ΛΟς―θΜ·Cl2ΘΨFe3+ΘΨSO2ΒΡ «
| | Ιΐ≥ΧΔτ B»ή“Κ÷–Κ§”–ΒΡάκΉ” | Ιΐ≥ΧΔω B»ή“Κ÷–Κ§”–ΒΡάκΉ” |
| ΦΉ | ”–Fe3+ΈόFe2+ | ”–SO42- |
| ““ | Φ»”–Fe3+”÷”–Fe2+ | ”–SO42- |
| ±ϊ | ”–Fe3+ΈόFe2+ | ”–Fe2+ |
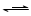 FeSO3Θ®sΘ©
FeSO3Θ®sΘ©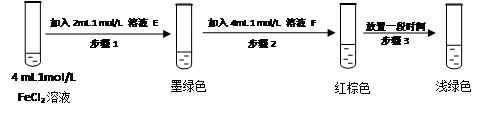
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ Χβ–ΆΘΚΒΞ―ΓΧβ
Θ®14Ζ÷Θ©ΔώΘ°Ρ≥Ά§―ßΉωΓΑ¬Ν»»Ζ¥”ΠΓ±ΒΡ Β―ιΓΘ≤ι‘ΡΓΕΜ·―ß ÷≤αΓΖ÷ΣΘ§AlΓΔAl2O3ΓΔFeΓΔFe2O3»έΒψΓΔΖ–Βψ ΐΨί»γœ¬ΘΚ
| Έο÷ | Al | Al2O3 | Fe | Fe2O3 |
| »έΒψ/Γφ | 660 | 2054 | 1535 | 1462 |
| Ζ–Βψ/Γφ | 2467 | 2980 | 2750 | ΓΣΓΣ |
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ Χβ–ΆΘΚΒΞ―ΓΧβ
»ή“Κ÷–Ω…ΡήΚ§”–H+ΓΔNH4+ΓΔMg2+ΓΔAl3+ΓΔFe3+ΓΔCO32?ΓΔSO42?ΓΔNO3?÷–ΒΡΦΗ÷÷ΓΘΔΌΦ”»κ¬ΝΤ§Θ§≤ζ…ζΈό…ΪΈόΈΕΒΡΤχΧεΘΜΔΎΦ”»κNaOH»ή“ΚΘ§≤ζ…ζΑΉ…Ϊ≥ΝΒμΘ§«“≤ζ…ζΒΡ≥ΝΒμΝΩ”κΦ”»κNaOHΒΡΈο÷ ΒΡΝΩ÷°ΦδΒΡΙΊœΒ»γœ¬ΆΦΥυ ΨΓΘ‘ρœ¬Ν–ΥΒΖ®’ΐ»ΖΒΡ «Θ®ΓΓΓΓΘ©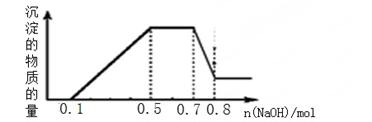
| AΘ°»ή“Κ÷–“ΜΕ®≤ΜΚ§CO32?Θ§Ω…ΡήΚ§”–SO42?ΚΆNO3? |
| BΘ°‘ΎΒΈΦ”NaOH»ή“ΚΈο÷ ΒΡΝΩΈΣ0Θ°5÷Ν0Θ°7mol ±Θ§ΖΔ…ζΒΡάκΉ”Ζ¥”ΠΈΣΘΚAl3+ΘΪ4OH-ΘΫ[Al(OH)4]- |
| CΘ°»ή“Κ÷–ΒΡ―τάκΉ”÷Μ”–H+ΓΔMg2+ΓΔAl3+ |
| DΘ°n(H+)ΓΟn(NH4+)ΓΟn(Mg2+) =2ΓΟ4ΓΟ1 |
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ Χβ–ΆΘΚΒΞ―ΓΧβ
”Οœ¬ΆΦΥυ ΨΉΑ÷Ο≤ΜΡή¥οΒΫ”–ΙΊ Β―ιΡΩΒΡ «
| AΘ°”ΟΦΉΆΦΉΑ÷Ο÷ΛΟςΟήΕ»Π―(ΟΚ”Ά)ΘΦΠ―(ΡΤ)ΘΦΠ―(Υ°)ΓΓΓΓΓΓ ΓΓ |
| BΘ°”Ο““ΆΦΉΑ÷Ο÷Τ±ΗFe(OH)2 |
| CΘ°”Ο±ϊΆΦΉΑ÷Ο÷Τ»ΓΫπ τΟΧ |
| DΘ°”ΟΕΓΆΦΉΑ÷Ο±»ΫœNaHCO3ΚΆNa2CO3ΒΡ»»Έ»Ε®–‘ |
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΩΤΡΩΘΚΗΏ÷–Μ·―ß ά¥‘¥ΘΚ Χβ–ΆΘΚΒΞ―ΓΧβ
“―÷Σ‘ΎΥα–‘ΧθΦΰœ¬ΡήΖΔ…ζœ¬Ν–Ζ¥”ΠΘΚCuΘΪΓζ CuΘΪCu2ΘΪ(Έ¥≈δΤΫ)ΓΘNH4CuSO3”κΉψΝΩΒΡ2molΓΛL-1ΝρΥα»ή“ΚΜλΚœΈΔ»»Θ§≤ζ…ζœ¬Ν–œ÷œσΘΚΔΌ”–ΉœΚλ…ΪΫπ τ…ζ≥…ΘΜΔΎ”–¥ΧΦΛ–‘ΤχΈΕΤχΧε≤ζ…ζΘΜΔέ»ή“Κ≥ άΕ…ΪΓΘΨί¥Υ≈–Εœœ¬Ν–ΥΒΖ®“ΜΕ®ΚœάμΒΡ «
| AΘ°ΗΟΖ¥”Πœ‘ ΨΝρΥαΨΏ”–Υα–‘ | BΘ°NH4CuSO3÷–Νρ‘ΣΥΊ±Μ―θΜ· |
| CΘ°¥ΧΦΛ–‘ΤχΈΕΒΡΤχΧε «Εΰ―θΜ·Νρ | DΘ°Ζ¥”Π÷–ΝρΥαΉς―θΜ·ΦΝ |
≤ιΩ¥¥πΑΗΚΆΫβΈω>>
ΙζΦ ―ß–Θ”≈―Γ - ΝΖœΑ≤αΝ–±μ - ‘ΧβΝ–±μ
Κΰ±± ΓΜΞΝΣΆχΈΞΖ®ΚΆ≤ΜΝΦ–≈œΔΨΌ±®ΤΫΧ® | Άχ…œ”–ΚΠ–≈œΔΨΌ±®Ή®«χ | Βγ–≈’©Τ≠ΨΌ±®Ή®«χ | …φάζ Ζ–ιΈό÷ς“ε”–ΚΠ–≈œΔΨΌ±®Ή®«χ | …φΤσ«÷»®ΨΌ±®Ή®«χ
ΈΞΖ®ΚΆ≤ΜΝΦ–≈œΔΨΌ±®ΒγΜΑΘΚ027-86699610 ΨΌ±®” œδΘΚ58377363@163.com