
ȡA��B�������ʵ���Ũ����ȵ�NaOH��Һ�������Ϊ50 mL���ֱ�������ͨ��һ������CO2���ٷֱ�ϡ��Ϊ100 mL��
(1)��NaOH��Һ��ͨ��һ������CO2����Һ�е����ʵ���ɿ����ǣ�
�� ���� ���� ���� ��
(2)��ϡ�ͺ����Һ�зֱ���μ���0.1 mol��L��1�����ᣬ������CO2�����(��״��)����������������ϵ��ͼ��ʾ��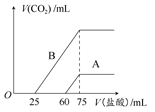
�ٷֱ�����������������Һ�е������� ��ԭNaOH��Һ�����ʵ���Ũ���� ��
��A���߱�����ͨ��CO2����Һ�е������� �������ᷴӦ����CO2���������� mL(��״��)��
��B���߱�����ԭNaOH��Һͨ��CO2���������ʵĻ�ѧʽΪ �������ʵ���֮��Ϊ ��
 ��У����ϵ�д�
��У����ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�Ȼ���������ض��dz�����ˮ����������ͼΪ�Ʊ��Ȼ�������һ�������Ʊ�������صĹ������̡�
��ش��������⣺
��1���Ȼ����ж�����;���������ӷ���ʽ��ʾ������;��ԭ����
���Ȼ�������ˮ��______________________��
����FeCl3��Һ��32%��35%����ʴͭӡˢ��·��____________________________��
��2�����ռ�X�Ļ�ѧʽΪ ���� ��������Y�Ļ�ѧʽΪ________________��
��3�����������·�Ӧ�ٵ����ӷ���ʽΪ____________________________________��
��4�����̢ڽ������Һ�����Сʱ�����ã����˻�ôֲ�Ʒ���÷�Ӧ�Ļ�ѧ����ʽΪ
2KOH��Na2FeO4��K2FeO4��2NaOH������ݸ��ֽⷴӦԭ��������Ӧ������ԭ��_________��
��5��K2FeO4��ˮ��Һ��������Ӧ��4FeO42-+10H2O 4Fe(OH)3+8OH-+3O2�������ᴿK2FeO4ʱ�����ؽᾧ��ϴ�ӡ����º�ɵķ�������ϴ�Ӽ����ѡ�� ������ţ���
4Fe(OH)3+8OH-+3O2�������ᴿK2FeO4ʱ�����ؽᾧ��ϴ�ӡ����º�ɵķ�������ϴ�Ӽ����ѡ�� ������ţ���
| A��H2O | B��ϡKOH��Һ������� | C��NH4Cl��Һ������� | D��Fe(NO3)3��Һ������� |
 CrO42-��Fe(OH)3����OH-
CrO42-��Fe(OH)3����OH- Cr2O72-��H2O
Cr2O72-��H2O 2Cr3����6Fe3����7H2O
2Cr3����6Fe3����7H2O�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�����仯����Ӧ�ù㷺��
��1�����Ȼ�����һ��ˮ����������ҵ�Ʊ���ˮ���Ȼ�������IJ�����������ͼ��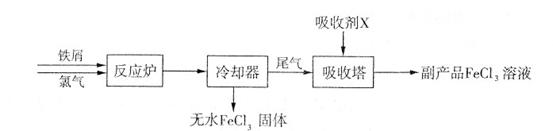
�ټ��鸱��Ʒ�к���Xʱ��ѡ�õ��Լ��� (�����и��������)��
a��NaOH��Һ b��KSCN��Һ c������KMnO4��Һ d������
�����������У����ɸ���ƷFeCl�������ӷ���ʽΪ
��2���������(K2FeO4)Ҳ��һ��������ˮ����������ҵ�ϣ������������������KOH��Һ�Ʊ�������ء��������У������ĵ缫��ӦʽΪ �����һ��ʱ�����������������28 g�����ڴ˹����У����������������ڱ�״���µ����Ϊ L��
��3�������������ڹ�ҵ��ˮ�Ĵ�����
������Ϊ���ܷ���������������Cd2+�Ĺ�ҵ��ˮ? (��ܡ���)������ݳ����ܽ�ƽ���ԭ��������Ĺ۵�(�ñ�Ҫ�����ֺ����ӷ���ʽ˵��)�� (��֪��25��ʱ���ܶȻ�����Ksp(FeS)=6��310-18��Ksp(CdS)=3��610-29)
�ڹ�ҵ�ϴ�����Cd2+��ˮ�����Բ��ü�̼���Ƶķ�������Ӧ���£�2Cd2++2CO32-+H2O=Cd2(OH)2CO3 +A����A�Ļ�ѧʽΪ ��
+A����A�Ļ�ѧʽΪ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��������������һ�ֳ��õĽ������洦������������ʹ���ı�������һ�����ܵ�����Ĥ��������Ĥ������ϡ���ᡣ
ij��ѧ�о�С����ʵ�����а����в���ģ�������桰�ۻ����IJ������̡�
(1)����ʵ���õ���Һ��Ҫ����200 mL�ܶ�Ϊ1.2 g/cm3��������������Ϊ16%��NaOH��Һ����Ҫ��ȡ________g NaOH���塣
(2)����Ƭ�����ȵ�16%��NaOH��Һ��Լ���������ϴȥ���ۣ���ȥ���������Ĥ��ȡ����ˮ��ϴ��д����ȥ����Ĥ�����ӷ���ʽ��________��
(3)����ͼ��װ����������ͨ����K��ͨ��Լ25 min�������������������������������塣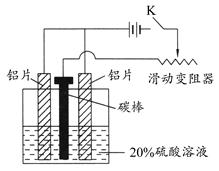
д���ù����еĵ缫��Ӧʽ��
������_________________________________________��
������_________________________________________��
(4)�Ͽ���·��ȡ����Ƭ������������Ϊ1%��ϡ��ˮ�кͱ������Һ������ˮ��ϴ�ɾ���д���ù��̷�����Ӧ�����ӷ���ʽ��______________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�����Ƽ�IJ�������ͼ��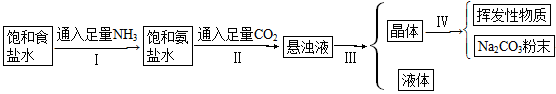
�Իش��������⣺
(1)������������Һ��NaHCO3��NH4Cl�Ļ��Һ�����͢���ܷ�ӦΪ
_______________________________________________
(2)����һ��Ӧ��֪NaCl��NaHCO3�ܽ��______________________��
(3)��IJ�����____________��
(4)���IJ�����____________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
������Ԫ��A��B��C��Dλ��ͬ������ԭ����������������֪A��C��D����Ԫ��ԭ�ӵ�����㹲��10�����ӣ���������Ԫ�ص��������������Ӧ��ˮ����֮���������ܷ�Ӧ���������κ�ˮ����������и�С�⣺
(1)A��B��C���������ӵİ뾶�ɴ�С��˳���ǣ�____>_____>____(��д���ӷ���)��
(2)ָ��Ԫ��D��Ԫ�����ڱ��е�λ�ã���________���ڵ�________�塣
(3)Ԫ��B������������________������(����ӡ����ۡ�)�������ʽΪ________________��
(4)A��CԪ�ص��������������Ӧ��ˮ����֮�䷴Ӧ�����ӷ���ʽΪ________________��
(5)����C�����ӵ��γ���������ˮ����ԭ��Ϊ______________(�����ӷ���ʽ��ʾ���ʵ�������˵��)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
����ʯ��������õ�����[KAl(SO4)2��12H2O]���������Ʊ�Al��K2SO4����H2SO4�Ĺ��չ���������ʾ��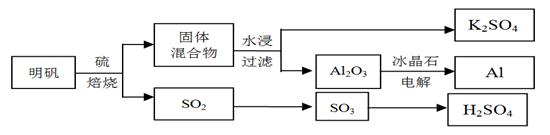
���������Ļ�ѧ����ʽΪ��4KAl(SO4)2��12H2O + 3S = 2K2SO4 + 2Al2O3 + 9SO2 + 48H2O
��ش��������⣺
��1���ڱ��������ķ�Ӧ�У���ԭ���� ��
��2����ˮ�������Һ�еõ�K2SO4����ķ����� ��
��3��Al2O3��һ�������¿��Ƶ�AlN���侧��ṹ��ͼ��ʾ���þ�����Al����λ���� ��
��4����Al��NiO(OH)Ϊ�缫��NaOH��ҺΪ���Һ���һ�����͵�أ��ŵ�ʱNiO(OH)ת��ΪNi(OH)2���õ�ط�Ӧ�Ļ�ѧ����ʽ�� ��
��5�����ղ�����SO2�����������ᡣ��֪25�桢101kPaʱ��
2SO2��g��+ O2��g�� 2SO3��g�� ��H1 = ��197 kJ /mol��
2SO3��g�� ��H1 = ��197 kJ /mol��
H2O��g�� H2O��l�� ��H2 = ��44 kJ/mol��
H2O��l�� ��H2 = ��44 kJ/mol��
2SO2��g��+ O2��g��+ 2H2O��g��=2H2SO4��aq�� ��H3 = ��545 kJ/mol��
��SO3��g����H2O��l����Ӧ���Ȼ�ѧ����ʽ�� ��
����948 t������M =" 474" g/mol������SO2��������Ϊ96%���ɲ�����������Ϊ98%������ t��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ʵ�����������᳧����(��Ҫ�ɷ�Ϊ���������P����FeS��SiO2��)�Ʊ�����(��ʽ�������ľۺ���)���̷�(FeSO4��7H2O)���������£�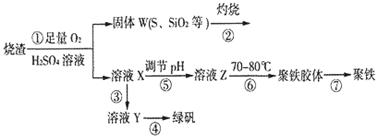
��1�������̢��еIJ���������ͨ��������Һ�У���Һ����ɫ����__________��
A��Ʒ����Һ B����ɫʯ����Һ C������KMnO4��Һ D����ˮ
��2�����̢��У�FeS��O2��H2SO4��Ӧ�Ļ�ѧ����ʽΪ��___________________________________��
��3�����̢��У�������������___________________________��
��4�����̢��У������ᾧ��Ҫʹ�þƾ��ơ����Ǽܡ������ǣ�����Ҫ��������_______________��
��5�����̢ݵ���pH��ѡ�������Լ��е�___________ (��ѡ�����)��
A��ϡ���� B��CaCO3 C��NaOH��Һ
��6�����̢��У�����ҺZ���ȵ�70һ80�棬Ŀ����_____________________��
��7��ʵ����Ϊ�������õ��ľ�����Ʒ����Ԫ�ص�������������������ʵ�顣���÷�����ƽ��ȡ2.70g��Ʒ���ڽ���Ʒ�����������������������Ȼ�����Һ���۹��ˡ�ϴ�ӡ�����������ù�������Ϊ3.495g�����þ�����Ҫ�ɷ�Ϊ[(Fe(OH)(SO4)]n����þ�����Ʒ����Ԫ�ص���������Ϊ___________��(���������в�����Ԫ�غ���Ԫ��)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ҵ����������Ϊԭ���Ʊ��������ѵĹ�����������ͼ��ʾ�����������Ҫ�ɷ�Ϊ��������(FeTiO3)������һ������Ԫ���ڷ绯�����л�ת��Ϊ��3�ۡ�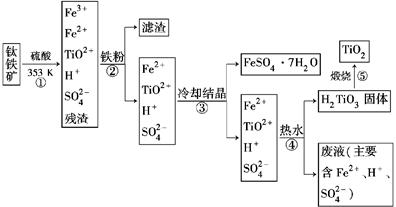
��֪��TiOSO4��ˮ��ˮ�⡣
(1)������У������۽�Fe3��ת��ΪFe2�������ӷ���ʽΪ_______________________
(2)������У�ʵ�ֻ����ķ������������ʵ�________(����ĸ���)��
a���ۡ��е���졡 b���ܽ��Բ��졡 c�������ԡ���ԭ�Բ���
(3)����ڡ��ۡ����У�����Ҫ���еIJ�����________(���������)��
(4)���ϻ�ѧ�����û�ѧƽ�����۽��Ͳ�����н�TiO2��ת��ΪH2TiO3��ԭ����
____________________________________________________________��
(5)�������������еķ�Һ�����̿�(��Ҫ�ɷ�ΪMnO2)��Ӧ������������(MnSO4��������ˮ)���÷�Ӧ�����ӷ���ʽΪ__________________________________
(6)�о����֣���ʯī������������������������CaF2��CaO������ʣ�������ͼ��ʾװ�ÿɻ�ý����ƣ������Ը�Ϊ��ԭ������ԭ���������Ʊ������ѡ�
д�������ĵ缫��Ӧʽ��_________________________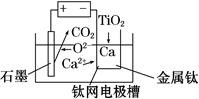
���Ʊ�������ǰ��CaO���������䣬��ԭ����______________________________________(���ϻ�ѧ�������)��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com