
X��Y��Z��W��Ԫ�����ڱ���ԭ������������������ֶ�����Ԫ�أ��������Ϣ���±���

��1��Wλ��Ԫ�����ڱ��� ���ڵ� �壻W��ԭ�Ӱ뾶��X�� �����С������
��2��Z�ĵ�һ�����ܱ�W�� �����С������  ��̬��Ϊ��̬����˷��������������� ����Ԫ�ء�
��̬��Ϊ��̬����˷��������������� ����Ԫ�ء� ��
��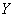 ��ԭ�ӿɹ�ͬ�γɶ��ַ��ӣ�д������һ�����γ�ͬ�ַ��Ӽ�������������� ��
��ԭ�ӿɹ�ͬ�γɶ��ַ��ӣ�д������һ�����γ�ͬ�ַ��Ӽ�������������� ��
��3�����£���Z���������ᷴӦ�����ɫ��Һ�еμ�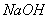 ��Һֱ���������ܹ۲쵽�������ǣ�
��Һֱ���������ܹ۲쵽�������ǣ� �ĵ���������ᷴӦ����������ɫ���壬�÷�Ӧ�Ļ�ѧ����ʽ�� ��
�ĵ���������ᷴӦ����������ɫ���壬�÷�Ӧ�Ļ�ѧ����ʽ�� ��
��4����25�㡢101 �£���֪13.5g��
�£���֪13.5g��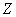 ���嵥����
���嵥���� ��������ȫȼ�պ�ָ���ԭ״̬������419
��������ȫȼ�պ�ָ���ԭ״̬������419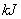 ,�÷�Ӧ���Ȼ�ѧ����ʽ�� ��
,�÷�Ӧ���Ȼ�ѧ����ʽ�� ��
25
 ���㼤�������100�ִ��Ծ�ϵ�д�
���㼤�������100�ִ��Ծ�ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�������ȼҵ�еķ�������Ҫ�ɷ���þ�Ĺ����κ�̼����(���������������Ƶ���)��ʵ����������Ϊԭ����ȡMgSO4��7H2O��ʵ��������£�

 ��֪����������Ksp[Mg(OH)2]��6.0��10��12��������Һ�У�Fe2����Fe3����Al3���ӿ�ʼ������������ȫ��pH��Χ����Ϊ7.1��9.6��2.0��3.7��3.1��4.7�������ֻ�������ܽ��(S)���¶ȱ仯��������ͼ��ʾ��
��֪����������Ksp[Mg(OH)2]��6.0��10��12��������Һ�У�Fe2����Fe3����Al3���ӿ�ʼ������������ȫ��pH��Χ����Ϊ7.1��9.6��2.0��3.7��3.1��4.7�������ֻ�������ܽ��(S)���¶ȱ仯��������ͼ��ʾ��
(1)�������м���ϡ�����pHΪ1��2�Լ���е�Ŀ����______________________��
(2)�������µ���Һ��Mg2����Ũ��Ϊ6.0 mol��L��1������ҺpH��________�ſ��ܲ���Mg(OH)2������
(3)����Һ����Һ�����ȼ���NaClO����ҺpHԼΪ5���ٳ��ȹ��ˣ�����ȹ��˵�Ŀ����__________________����������Ҫ�ɷ���______________________��
(4)����Һ���л��MgSO4��7H2O�����ʵ�鲽������Ϊ������Һ���м���______________���ڹ��ˣ��ó�������________________��������Ũ�������½ᾧ���ݹ��ˡ�ϴ�ӵò�Ʒ��
(5)����õ�MgSO4��7H2O������Ϊ24.6 g�����������þ[��Mg(OH)2��]�İٷֺ���ԼΪ________(MgSO4��7H2O����Է�������Ϊ246)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
Ǧ���仯������������ء������豸��X���߷������ϵȡ��ش��������⣺
(1)Ǧ��̼��ͬ��Ԫ�أ���̼��4�����Ӳ㡣Ǧ��Ԫ�����ڱ���λ��Ϊ��________���ڡ���________�壻PbO2�����Ա�CO2������________(�ǿ��������)��
(2)PbO2��Ũ���Ṳ�����ɻ���ɫ���壬��Ӧ�Ļ�ѧ����ʽΪ_______________________��
(3)PbO2����PbO�����������Һ��Ӧ�Ƶã���Ӧ�����ӷ���ʽΪ___________________��PbO2Ҳ����ͨ��ʯīΪ�缫��Pb(NO3)2��Cu(NO3)2�Ļ����ҺΪ���Һ�����ȡ�����������ĵ缫��ӦʽΪ____________________�������Ϲ۲쵽��������____________________�������Һ�в�����Cu(NO3)2�����������ĵ缫��ӦʽΪ______________________________������������Ҫȱ����____________________��
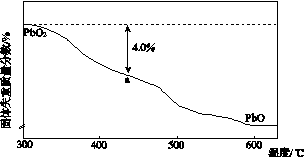
(4)PbO2�ڼ��ȹ��̷����ֽ��ʧ����������ͼ��ʾ����֪ʧ�������ϵ�a��Ϊ��Ʒʧ��4.0%(��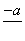 ��100%)�IJ������塣��a�������ɱ�ʾΪPbOx��mPbO2��nPbO����ʽ����xֵ��m��nֵ_______________________________________
��100%)�IJ������塣��a�������ɱ�ʾΪPbOx��mPbO2��nPbO����ʽ����xֵ��m��nֵ_______________________________________
________________________________________________________________________
________________________________________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�����й�Ԫ�ص����ʼ���ױ߹�����ȷ����
A��IA����VIIA��Ԫ�ؼ���γɹ��ۻ���������ӻ�����
B����ߵڶ�����Ԫ�ش����ң����۴�+1������+7
C��ͬ�� ��Ԫ�صļ������ӻ�ԭ��Խǿ��ˮ��̶�Խ��
��Ԫ�صļ������ӻ�ԭ��Խǿ��ˮ��̶�Խ��
D��ͬ���ڽ���Ԫ�صĻ��ϼ�Խ�ߣ���ԭ��ʧ��������Խǿ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
������Ԫ��X��Y��Z��W��Q��Ԫ�����ڱ���λ�������ʾ������XԪ�ص�ԭ���ڲ��������������������һ�룬������˵����ȷ����
| X | Y | ||
| Z | W | Q |

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
���й���ʵ������ȡ���������Լ�������в���������(����)
A����Cl2��MnO2��Ũ����
B����NH3��Ũ��ˮ����ʯ��
C����NO��ϡ���ᡢCu
D����CO2��ϡ���ᡢCaCO3
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
25 ��ʱ����0.01 mol��L��1��������Һ�У�ˮ�������H��Ũ���� ( )
A��5��10��13 mol��L��1 B��0.02 mol��L��1
C��1��10��7 mol��L��1 D��1��10��12 mol��L��1
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����������������ȷ���� �� ��
A��2NO2(g)(����ɫ)  N2O4(g)(��ɫ)����H<0����ƽ���ƽ����ϵ��ȡ��С�ݻ�������ѹǿ�Ĵ�ʩ����Ϊƽ��������Ӧ�����ƶ�������ϵ��ɫ��dz
N2O4(g)(��ɫ)����H<0����ƽ���ƽ����ϵ��ȡ��С�ݻ�������ѹǿ�Ĵ�ʩ����Ϊƽ��������Ӧ�����ƶ�������ϵ��ɫ��dz
B������N2(g)��3H2(g) 2NH3(g)��ƽ�����ѹǿ���䣬����Ar��ƽ������
2NH3(g)��ƽ�����ѹǿ���䣬����Ar��ƽ������
C��FeCl3��3KSCN Fe(SCN)3(��ɫ)��3KCl����ƽ�������KCl���壬��Ϊƽ�����淴Ӧ�����ƶ�������ϵ��ɫ��dz
Fe(SCN)3(��ɫ)��3KCl����ƽ�������KCl���壬��Ϊƽ�����淴Ӧ�����ƶ�������ϵ��ɫ��dz
D��H2(g)��I2(g)  2HI(g)����H<0����ƽ���ƽ����ϵ��ȡ�����ݻ�����Сѹǿ�Ĵ�ʩ����Ϊƽ�ⲻ�ƶ�������ϵ��ɫ����
2HI(g)����H<0����ƽ���ƽ����ϵ��ȡ�����ݻ�����Сѹǿ�Ĵ�ʩ����Ϊƽ�ⲻ�ƶ�������ϵ��ɫ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��1 200 ��ʱ����Ȼ���������лᷢ�����з�Ӧ
H2S(g)��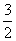 O2(g)===SO2(g)��H2O(g)����H1
O2(g)===SO2(g)��H2O(g)����H1
2H2S(g)��SO2(g)===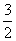 S2(g)��2H2O(g)����H2
S2(g)��2H2O(g)����H2
H2S(g)�� O2(g)===S(g)��H2O(g)����H3
O2(g)===S(g)��H2O(g)����H3
2S(g)===S2(g)����H4
��H4����ȷ����ʽΪ
A����H4�� (��H1����H2��3��H3)
(��H1����H2��3��H3)
B����H4�� (3��H3����H1����H2)
(3��H3����H1����H2)
C����H4��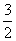 (��H1����H2��3��H3)
(��H1����H2��3��H3)
D����H4��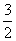 (��H1����H2��3��H3)
(��H1����H2��3��H3)
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com