
ijУ��ѧʵ����ȤС��Ϊ��̽����ʵ�����Ʊ�Cl2�Ĺ�������ˮ������HCl�ӷ�������ͬʱ֤��������ijЩ���ʣ���ͬѧ���������ͼ��ʾ��ʵ��װ�ã�֧���õ�����̨ʡ�ԣ�����Ҫ��ش����⡣
��1�����з�����,���Ƶ���������ȷ����� ��
��MnO2��Ũ�����Ϲ���
��KMnO4��Ũ������ �������ƺ�Ũ������ ��K2Cr2O7��Ũ������
| A���٢ڢ� | B���٢ڢ� | C���٢� | D��ȫ������ |
��1��D ��2��Ũ�������ŷ�Ӧ�Ľ���Ũ�����ͣ����ձ�Ϊϡ���ᣬ��Ӧ���ٲ�������
��3����֤����ˮ�������� ��ɫ�������ɫ
��������Ư���ԣ�������ˮ��Ӧ�����Ĵ�������Ư����
����������
��1��Ca(ClO)2+CO2+H2O=CaCO3��+2HClO
��2��Cl2+2NaClO2=2ClO2+2NaCl
���������������1��MnO2��KMnO4��NaClO3��K2Cr2O7������ǿ�����ԣ���Ũ���ᷴӦ��������Cl2����D����ȷ��
��2��Ũ�������ŷ�Ӧ�Ľ���Ũ�����ͣ����ձ�Ϊϡ���ᣬ��Ӧ���ٲ���������HClΪ0��2mol���������ɵ�Cl2С��0��05mol��С��1��12L��
��3����װ��B��ʢ����ˮ����ͭ�����������Ϊ֤��ˮ�����Ĵ��ڣ�����Ϊ����ɫ�������ɫ��
��װ��D��E���������ڲ����Ƿ�������ʪ��IJ����У�������ˮ��Ӧ���ɴ����ᣬ���������װ��˵���ˡ�������Ư���ԣ���������Ư���ԡ���
��Ҫ������HCl�ӷ���������Ҫ��ȥCl2������װ��E�������ǣ�����������
Ư�۳��ڷ����ڿ����У�Ca(ClO)2��CO2��H2O��Ӧ����HClO��HClO�ֽ�ʹƯ��ʧЧ��
Cl2��NaClO2�е�+3��Cl����ΪClO2����ƽ�ɵû�ѧ����ʽ��
���㣺���⿼����������ȡ�����ʡ���ѧ����ʽ����д��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ij��ѧ����С����ʵ�����������ͼ��ʾ��ʵ��װ�ã����С����Ĵ�������ʵ�顣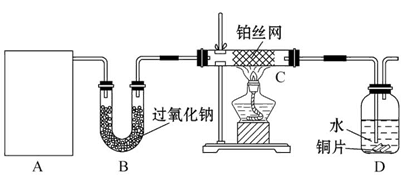
��1��A�������巢��װ�ã�A�����õ��Լ�ֻ�ܴ�����������ѡȡ��
������泥���̼��泥���̼����泥����Ȼ�泥�����ʯ�ң����������ơ�
��A����ȡ����ʱֻ����һ��ҩƷ�����ҩƷ������_________����ѡ���ţ�����ֻ��һ��ҩƷ��ȡ����ʱ��ͼ�пհ״���������ӦΪ_________ ��ѡ������������ţ��̶�װ��ʡ�ԣ���
��2����װ�ò�����������Ȼ����һ����ȱ�ݣ��ԴӰ�ȫ�뻷���ĽǶ������ǣ��Ը�װ�ý��иĽ���
��_______________________________________________________________;
��_______________________________________________________________��
��3�����ոĽ����װ�ý���ʵ�飬������������⣺
��װ��B��������__________________________________________________��
��д��C�з�����Ӧ�Ļ�ѧ����ʽ�� ________________________________��
����A��B���Լ���������װ��D�п��Թ۲쵽��ʵ��������_________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ij��ȤС����Ʋ�����������ʵ����̽��Cl2��Ư�۵��Ʊ����й����ʡ�
��1��ʵ������������װ���Ʊ����﴿�����������밴������������������ķ��������������ӣ�
H�������������������������������������������������������ƿ���е��Լ�Ϊ��������������������������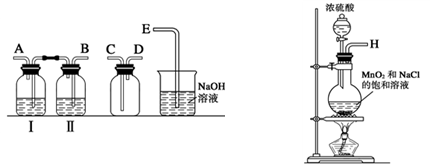
��2��д����ҵ����������ʯ������ȡƯ�۵Ļ�ѧ��Ӧ����ʽ��
������������������������������������������������������������������������������������������
ijѧ���������ʵ���һ��̽��SO2��Ư�۾��ķ�Ӧ��
| ���� | ���� |
| ȡ4 gƯ�۾����壬����100 mLˮ | ���ֹ����ܽ⣬��Һ������ɫ |
| ���ˣ���Ư�۾���Һ��pH | pH��ֽ�ȱ�����ԼΪ12��������ɫ |
 A | ��.Һ���Ϸ�������״�� ��.�Ժ��ֻ��ǣ���Һ��Ϊ����ɫ ��.�Ժ���������ɫ����������ɫ��ȥ |
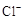 �ķ����ǣ��� ����������������������������������������
�ķ����ǣ��� �����������������������������������������鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
�������ƣ�NaNO2����¶�ڿ����л���������Ӧ���������ƣ�����ά��֯Ʒ��Ⱦɫ��Ư�ס����ࡢ��������ҩ�������й㷺Ӧ�ã�Ҳ���������ࡢ�����ʳƷ��Ⱦɫ�ͷ������������ж���������ʳƷ��ҵ�������ϸ����ơ�������ͼ��ʾ�������г�װ����ʡ�ԣ���ҩƷ��̽���������������ᷴӦ���������ɷ֡�
��֪����NO+NO2+2OH��==2NO2��+H2O ������Һ�����¶ȣ�NO2��21�棬NO��-152��
��1��Ϊ�˼���װ��A�����ɵ�����������������˳����������ӣ���A��C�� �� �� ��
��2����ӦǰӦ���ɼУ���ͨ��һ��ʱ�䵪�����ų�װ���еĿ�����Ŀ���� ��
��3���ڹرյ��ɼУ���Һ©������������70%�����A�в�������ɫ���塣
��ȷ��A�в��������庬��NO�����ݵ������� ��
��װ��E�������� ��
��4�������D��ͨ�����O2����װ��B�з�����Ӧ�Ļ�ѧ����ʽΪ �����û��װ��C����ʵ�������ɵ�Ӱ���� ��
��5��ͨ������ʵ��̽�����̣��ɵó�װ��A�з�Ӧ�Ļ�ѧ����ʽ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ij��ȤС���������ͼʵ��װ�ý���ʵ�顣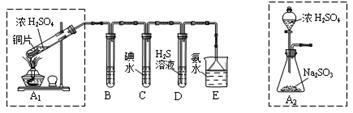
��̽��������Ⱦ��SO2������
��1��Ϊ��ʵ����ɫ������Ŀ�꣬�ܷ�����ͼA2����A1װ�� ����ܡ�����
��2��B��C��D�ֱ����ڼ���SO2��Ư���ԡ���ԭ�Ժ������ԣ���B����ʢ�Լ�Ϊ ��C�з�Ӧ�����ӷ���ʽΪ ��D�з�Ӧ�Ļ�ѧ����ʽΪ ��
��̽��ͭƬ��ŨH2SO4��Ӧ�IJ���
ʵ�������������ͭƬ���渽�ź�ɫ���塣�������ϵ�֪���˺�ɫ������ܺ���CuO��CuS��Cu2S��������CuS��Cu2S��������ϡ���ᣬ�ڿ��������ն�ת��ΪCu2O��SO2����С��ͬѧ�ռ�һ������ɫ���壬������ʵ�鷽��̽����ɷ֣�
��3��������м�������ϴ�Ӹɾ���ʵ�鷽����_____________________________��
��4����ɫ����ijɷ���________________��
�������
�ð�ˮ����β���е�SO2��������Һ���п��ܺ���OH-��SO32-��SO42-��HSO3-�������ӡ�
��5����ˮ���չ���SO2�ķ�Ӧ�����ӷ���ʽΪ ��
��6����֪����������һ��������ˮ��SO2Ҳ������ˮ�������������Լ�Ϊ��С�ձ����Թܡ�����������ͷ�ιܡ�����װ�ú���ֽ��2 mol/L���ᡢ2 mol/LHNO3��1 mol/LBaCl2��Һ��l mol/LBa(OH)2��Һ��Ʒ����Һ������ˮ�������ʵ��֤��������Һ���д���SO32-��HSO3-������±���ʵ�������Ԥ������ͽ��ۣ�
| ʵ����� | Ԥ����������� |
| ����1��ȡ����������Һ����С�ձ��У��ý�ͷ�ι�ȡl mol/L BaCl2��Һ��С�ձ��μ�ֱ�������� | �����ְ�ɫ���ǣ� ������Һ���д���SO32-�� SO42-�� |
| ����2����С�ձ��е���Һ���ˡ�ϴ�ӣ���������ˮ�Ѹ�����ֽ�ϵĹ��������һС�ձ��У�����µĹ��� �� | �� ������Һ���д��� SO32-�� |
| ����3�� �� | �� ������Һ���д��� HSO3-�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ijѧУ��ȤС���������װ�ý���ʵ��̽����a��bΪ���ɼУ����ȼ��̶�װ������ȥ����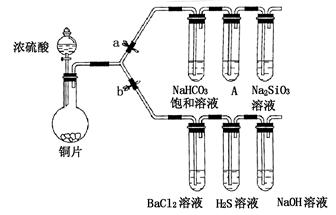
��1����֤̼����ǽ����Ե����ǿ��
������������ ����ҩƷ��a�ر�b��Ȼ�����Ũ���ᣬ���ȣ�
��װ��A�е��Լ��� ��
����˵��̼�ķǽ����Աȹ�ǿ��ʵ�������� ��
��2����֤SO2�������ԡ���ԭ�Ժ������������ͨ��
�ٴ�b���ر�a������֤SO2���������ԵĻ�ѧ����ʽ�� ��
����������SO2ͨ��NaOH��Һ�У������ӷ���ʽ�� ��
��BaCl2��Һ������������ֳ����ݣ��ֱ�μ�������Һ���������ij����Ļ�ѧʽ�����±���Ӧλ�á�
| �μӵ���Һ | ��ˮ | ��ˮ |
| �����Ļ�ѧʽ | | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
I���������ᴿ����ʯ(��Ҫ������������)������ͼ��
��Ҫ��ش��������⣺
��1����������Ҫ��ʱ�ⶨpH����ʵ��������pH��ֽ�ⶨ��ҺpH�IJ�����______________________________��
��2������Fe3+����ȫ����Fe(OH)3�����ķ�����________________________________��
��3������ҺB�õ�����B������Ҫ�ɷ���__________��
II��ij��ѧС����ʵ������Ũ�����ͭ��Ӧ��ȡ����������̽�����������Ƿ���֧��ľ̿��ȼ�գ���ʵ��װ��ͼ���£�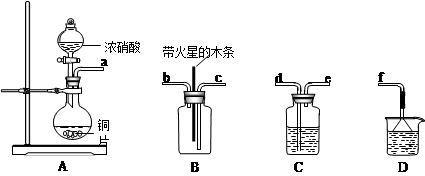
��1���������������Ӹ������ӿڣ�˳��Ϊa��______��______��______��______��f��װ��C������________________��װ��D���Լ���__________________��
��2����֪����������̼�ڵ�ȼ�����·�Ӧ�����������ʵ�����Ϊ2��1�������壬��÷�Ӧ�Ļ�ѧ����ʽΪ��___________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
Ϊ��֤��ͭ��ϡ���ᷴӦ����NO��ijУѧ��ʵ��С�������һ��ʵ�飬��װ����ͼ��ʾ������װ�ú̶�װ�þ�����ȥ����BΪһ���ý���˿�̶��ĸ���ܣ���װ��״̼��ƹ��壻EΪһ���յ�������ƿ��F�����ڹ��������˫��������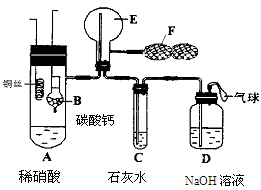
��1�� ʵ��ʱ���Ƚ�Bװ�����ƣ�ʹ̼�����ϡ����Ӵ��������壬��C��������ɫ����ʱ�����̽�Bװ�����ᣬʹ֮��ϡ������롣����ѧ����ƴ˲�������Ŀ��Ϊ ��
��2����A��ͭ˿����ϡ�����У���װ��A���ȣ���װ��A�в�����ɫ���壬�䷴Ӧ�Ļ�ѧ����ʽΪ ��
��3��װ��E�п�ʼʱ����dz����ɫ���壬��F��E�й�������ɹ۲쵽��ƿE��������ɫ����������������ԭ���� ��
��4��һ��ʱ���C�а�ɫ�����ܽ⣬��ԭ����________________________ __��
��5��װ��D��������_________ ___________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
Ϊ̽���������Ƶ����ȶ��ԣ�ij�о���ѧϰС�齫��ˮ�������Ƹ����������ȣ����������Ⱥ�Ĺ�����������ͼ��ʾ��ʵ��װ�ý���ʵ�顣��ش������й����⣺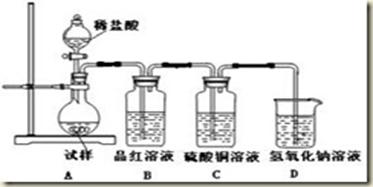
��1���������ϣ���ˮ�������Ƹ����������ȵ�600��ſ�ʼ�ֽ⣬�ҷֽ����ֻ�����ƺ�����һ�ֹ��塣��������¶ȵ���600�棬��������ȴ����������л����μ�ϡ�������������ڵμ�ϡ���������������HSO3-�����ʵ���Ũ�ȱ仯����Ϊ________________��
��2����������¶�Ϊ700�棬��������ȴ����������л����μ�ϡ�������������۲쵽��ƿ�г��ֵ���ɫ���������д������ݲ�������Ӧ���ɵ���ɫ���������ӷ���ʽΪ__________����ʱ��B��C��װ���п��ܹ۲쵽������Ϊ___________��___________��
��3���ڣ�2���еμ������������ƿ�ڳ�Cl-�⣬��������һ��Ũ�Ƚϴ�������ӣ�X��Ϊ___________������������ӣ�X������ȡ������������ˮ�����Һ��Ϊ�˼��ٹ����������ܽ⣬�ɲ�ȡ____________��ʩ�������Ǽ��������ӣ�X��������ʵ�鷽��������Ϊ�����ķ�����______����ס����ҡ�����
�����ף�ȡ����������Һ���Թ��У��ȼ�ϡHNO3���ټ�BaCl2��Һ���а�ɫ�������ɣ�֤�������Ӵ��ڡ�
�����ң�ȡ����������Һ���Թ��У��ȼ�ϡHCl���ټ�BaCl2��Һ���а�ɫ�������ɣ�֤�������Ӵ��ڡ�
��4��д��Na2SO3������ȵ�600�����Ϸֽ�Ļ�ѧ����ʽ____________��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com