
����Ŀ����֪���ᡢ��ˮ���ܶ�������ˮ���Ĺ�ϵ��ͼ��ʾ�����������백ˮ��һ�ݣ�����ݱ�����Ϣ���ش��������⣺
���ʵ����ʵ���Ũ��/molL-1 | ���ʵ��������� | ��Һ���ܶ�/gcm-3 | |
���� | c1 | w1 | ��1 |
��ˮ | c2 | w2 | ��2 |

��1�������������������w1Ϊ_____(��д��λ���ú�c1����1�Ĵ���ʽ��ʾ)��
��2�����ʵ���Ũ��Ϊc1mol��L-1��������Ϊw1��������ˮ��������(��Ϻ���Һ����仯���Բ���)��������Һ�����ʵ���Ũ��Ϊ_______mol��L-1����������_______w1/2(����ڡ�����С�ڡ����ڡ�����ͬ)��
��3����������Ϊw2�İ�ˮ��w2/5�İ�ˮ��������ϣ�������Һ���ܶ�_____��2 gcm-3��
��4�������700����İ����ܽ���1���ˮ���γɰ�ˮ������Һ����Һ���ܶ�Ϊd g/cm3�������Һ�����ʵ���Ũ��Ϊ________���ú���d�ı���ʽ��ʾ����
���𰸡�(9.8c1/��1)%��98c1/1000��1 c1/2 ���� ���� 1000d/49 mol��L��1
��������
��1������c��1000�Ѧ�/M���й�ʽ���μ��㣻
��2������ϡ�Ͷ��ɣ�ϡ��ǰ��������������ʵ������������䣬�ݴ˼���ϡ�ͺ���Һ��Ũ�Ⱥ�����������
��3����������Ϊw2�İ�ˮ��w2/5�İ�ˮ��������Ϻ���Һ��Ũ��С��w2����ͼ��֪����ˮ��Ũ��Խ���ܶ�ԽС���ݴ��жϻ�Ϻ���Һ���ܶ�����2 gcm-3��ϵ��
��4������n��V/Vm��c��n/V�����Һ���������ܶȼ��㰱ˮ��Ũ�ȡ�
��1������c��1000�Ѧ�/M��֪���������������w1��98c1/1000��1��
��2����������ˮ�����ΪVL�����Ϻ���Һ�������Ϊ2VL������ϡ�Ͷ��ɣ�ϡ��ǰ��������������ʵ������䣬ϡ�ͺ�������Һ��Ũ��Ϊ��VL��c1mol��L��1/2VL��0.5c1mol/L����������Ϊw1��������ˮ�������ϣ�ˮ������С��������Һ����������������С��ԭ������2�������������������w1/2��
��3����������Ϊw2�İ�ˮ��w2/5�İ�ˮ��������ϣ���Ϻ���Һ����������С��w2����ͼ��֪����ˮ��Ũ��Խ���ܶ�ԽС���ʻ�Ϻ���Һ���ܶȴ�����2 gcm-3��
��4��������700L�İ����ܽ���1Lˮ���γɰ�ˮ������Һ�����ʵ����ʵ�����![]() ��������
��������![]() ����Һ��������1000g+
����Һ��������1000g+![]() ����Һ���ܶ�Ϊd g/cm3�������Һ�������
����Һ���ܶ�Ϊd g/cm3�������Һ�������![]() ��������ʵ���Ũ��Ϊ
��������ʵ���Ũ��Ϊ ��
��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�����������£�CO2(g)+3H2(g)![]() CH3OH (g)+H2O(g) ��H=��57.3 kJ/mol���� 2L �����ܱ������г��� 1 mol CO2��3 mol H2���ڲ�ͬ���������·�����Ӧ�١���Ӧ���뷴Ӧ�ۣ���ͬʱ����CO2��ת�������¶ȱ仯����ͼ��ʾ��b�㷴Ӧ�ﵽƽ��״̬������˵����ȷ����
CH3OH (g)+H2O(g) ��H=��57.3 kJ/mol���� 2L �����ܱ������г��� 1 mol CO2��3 mol H2���ڲ�ͬ���������·�����Ӧ�١���Ӧ���뷴Ӧ�ۣ���ͬʱ����CO2��ת�������¶ȱ仯����ͼ��ʾ��b�㷴Ӧ�ﵽƽ��״̬������˵����ȷ����
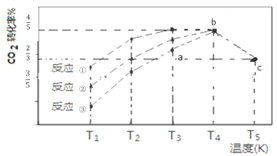
A. a �� v(��)>v(�棩
B. b�㷴Ӧ����53.7 kJ
C. ����Ч����ѵķ�Ӧ�Ǣ�
D. c��ʱ�÷�Ӧ��ƽ�ⳣ��K=4/3(mol-2![]() L-2)
L-2)
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����֪������(H2SeO3)Ϊ��Ԫ���ᣬ�����£���ijŨ�ȵ���������Һ����μ���һ��Ũ�ȵ�NaOH��Һ��������Һ��H2SeO3��HSeO3-��SeO32-�����������ʵ�����������ҺpH �Ĺ�ϵ��ͼ��ʾ������˵������ȷ���ǣ� ��
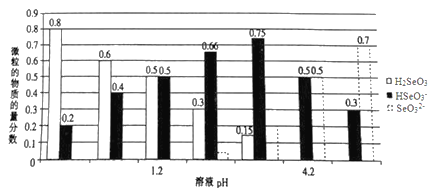
A. ����ͬ���ʵ���NaHSeO3��Na2SeO3 ������ȫ����ˮ�����pHΪ4.2�Ļ��Һ
B. pH=l.2����Һ�У�c(Na+)+c(H+)=c(OH-)+c(H2SeO3)
C. �����£�������ĵ���ƽ�ⳣ��K2=10-4.2
D. ��pH=1.2����Һ�еμ�NaOH��Һ��pH=4.2�Ĺ�����ˮ�ĵ���̶�һֱ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��[��ѧ����ѡ��5���л���ѧ����]ʯ���ѽ�����;�㷺�������ںϳɸ�����ҽҩ�м��塣����ʯ���ѽ����ϳ�CR��ҽҩ�м���K����·���£�
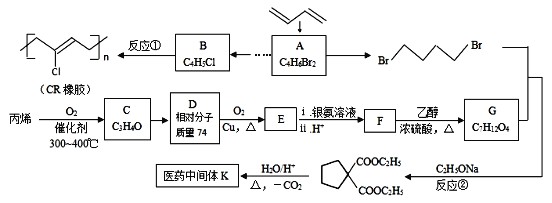
��֪��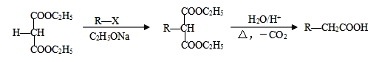
��1��A�ķ�ʽ�칹��Ľṹ��ʽΪ_________________��
��2����ϵͳ��������B��������������_________________��
��3��C��D��E��Ϊ��״�ṹ���Ҿ���������������ͭ����Һ��������ש��ɫ��������C�в������Ĺ�����������_________________�� д����ӦD��E�Ļ�ѧ����ʽ_________________��
��4��K�Ľṹ��ʽΪ_________________��
��5��д��F���Ҷ��������ۺϷ�Ӧ�Ļ�ѧ����ʽ______________________________��
��6��д��ͬʱ��������������ҽҩ�м���K������ͬ���칹��Ľṹ��ʽ___________��
a����E��Ϊͬϵ�� b���˴Ź���������3�ַ�
��7����֪˫���ϵ���ԭ�Ӻ��ѷ���ȡ����Ӧ����AΪ��ʼԭ�ϣ�ѡ�ñ�Ҫ�����Լ��ϳ�B��д���ϳ�·�ߣ�______________________________________________________________���ýṹ��ʽ��ʾ�л���ü�ͷ��ʾת����ϵ����ͷ��ע���Լ��ͷ�Ӧ��������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ������ʵ��������ʵ����ۻ�ԭ����һ�µ���
ѡ�� | ʵ������ | ʵ����ۻ�ԭ�� |
A | ����������ĭ�������� | 3HCO3-+Al3+===Al(OH)3��+3CO2�� |
B | ��AgCl����Һ�е�������KI��Һ���л�ɫ�������� | ˵��KSP��AgCl����KSP��AgI�� |
C | ��NaHS��Һ�е����̪����Һ���ɫ | HS-ˮ��̶ȴ��ڵ���̶� |
D | Na2CO3��Һ�еμӷ�̪�ʺ�ɫ | CO32-+2H2O |
A. A B. B C. C D. D
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��������������Ϊ98%���ܶ�Ϊ1.84g��cm-3��Ũ����������500mL 0.2mol/L��ϡ���ᡣ�ɹ�ѡ����������ٲ�����������ƿ�����ձ����ܽ�ͷ�ιܣ�����Ͳ��������ƿ����������ƽ����ҩ�ס�
��ش�����������
(1)���������У�������ϡ����ʱ�ò�����������______(�����)��
(2)������Һʱ��һ����Է�Ϊ���¼���������
����ȡ�ڼ����ϡ�͢�ҡ�Ȣ���Һ��ϴ�Ӣ߶��ݢ���ȴ�������װƿ��ǩ
����ȷ�IJ���˳��Ϊ�ڢ٢�______��_____�ܢ�(�����)��
(3)�����㣬��Ũ��������Ϊ______mL�����Т�10mL����50mL����100mL���ֹ�����Ͳ��Ӧѡ�õ���Ͳ��_______(�����)��
(4)�����ƹ����У�����������ȷ�����в����д�������ʹ������ҺŨ��ƫ�ߵ���____ (�����)
��ϴ����ȡŨ��������Ͳ������ϴ��Һת������ƿ��
��δ��ϡ�ͺ��������Һ��ȴ�����¾�ת������ƿ��
�۽�Ũ����ֱ�ӵ����ձ��������ձ���ע������ˮ��ϡ��Ũ����
�ܶ���ʱ��������ˮ�����̶��ߣ����ý�ͷ�ι�����
��ת��ǰ������ƿ�к�����������ˮ
����ҡ�Ⱥ���Һ����ڿ̶��ߣ����ý�ͷ�ιܼ�����ˮ���̶���
�߶���ʱ���ӿ̶���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��2017������й�����֪ʶ��Ȩ��ȫ������ú���Ҵ���ҵ����ĿͶ���ɹ���ij��ú���Ҵ��Ĺ��̱�ʾ���¡�
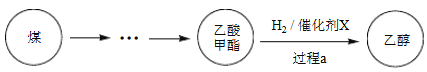
��1��Cu(NO3)2���Ʊ�������X������Ҫ�Լ���

�� ����A��_______��
�� ʵ������Cu(NO3)2����������Һ������������ϡHNO3�����û�ѧƽ��ԭ������HNO3������_______��
�� NaClO��Һ��������A�����ӷ���ʽ��_______��
��2������a��������3����Ҫ��Ӧ��
��CH3COOCH3(g)��2H2(g)![]() C2H5OH(g)��CH3OH(g) ��H1
C2H5OH(g)��CH3OH(g) ��H1
��CH3COOCH3(g)��C2H5OH(g)![]() CH3COOC2H5 (g)��CH3OH(g) ��H2
CH3COOC2H5 (g)��CH3OH(g) ��H2
��CH3COOCH3(g)��H2(g)![]() CH3CHO(g)��CH3OH(g) ��H3
CH3CHO(g)��CH3OH(g) ��H3
��ͬʱ���ڣ����CH3COOCH3ת���ʡ��Ҵ�������������ѡ���ԣ����Ҵ�ѡ����= ![]() ������ͼ��ʾ��
������ͼ��ʾ��
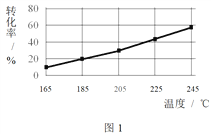
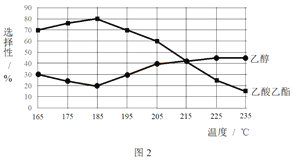
�� ��֪����H1 < 0�����¶Ƚ��ͣ���Ӧ��ѧƽ�ⳣ���ı仯������_______��
�� ����˵������������________��
A���¶ȿ�Ӱ�췴Ӧ��ѡ����
B��225�桫235�棬��Ӧ����ƽ��״̬
C������H2��Ũ�ȣ��������CH3COOCH3��ת����
�� Ϊ��ֹ����Ӧ��������Ӧ�¶�Ӧ���Ƶķ�Χ��_______��
�� ��185���£�CH3COOCH3��ʼ���ʵ���Ϊ5 mol�������Ҵ������ʵ�����____��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��ijС����Ʋ�ͬʵ�鷽���Ƚ�Cu2+��Ag+ �������ԡ�
��1������1��ͨ���û���Ӧ�Ƚ�
���ữ��AgNO3��Һ����ͭ˿��������ɫ���壬��Һ��������Ӧ�����ӷ���ʽ��_______��˵��������Ag+��Cu2+��
��2������2��ͨ��Cu2+��Ag+ �ֱ���ͬһ���ʷ�Ӧ���бȽ�
ʵ�� | �Լ� | ��ż����� | |
�Թ� | �ι� | ||
| 1.0 mol/L KI��Һ | 1.0 mol/L AgNO3��Һ | ������ɫ��������Һ��ɫ |
1.0 mol/L CuSO4��Һ | ������ɫ����A����Һ��� | ||
�� �����飬������Һ����I2����ɫ������________��
�� �����飬������Һ��I2���Ʋ�Cu2+������������ɫ����A��CuI��ȷ��A��ʵ�����£�
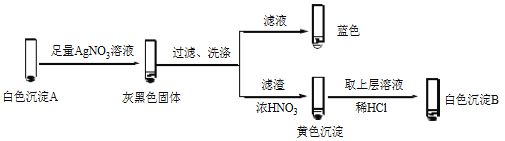
a��������Һ��I2����Һ����ɫ˵����Һ����________�������ӷ��ţ���
b����ɫ����B��________��
c����ɫ����A��AgNO3��Һ��Ӧ�����ӷ���ʽ��____��˵��������Ag+��Cu2+��
��3����������2��Ag+ δ������I- ����Cu2+������I-��ԭ�����ʵ�����£�
��� | ʵ��1 | ʵ��2 | ʵ��3 |
ʵ�� |
|
|
|
���� | �����Ա仯 | a����Һ�Ͽ���ػ�ɫ,b�е缫 ����������������ָ��ƫת | c����Һ������dz��ɫ�� ������ָ��ƫת |
���缫��Ϊʯī����ҺŨ�Ⱦ�Ϊ 1 mol/L��b��d����ҺpH��4��
�� a����Һ���ػ�ɫ��ԭ����_______���õ缫��Ӧʽ��ʾ����
�� ��ʵ��3������˵��Cu2+������I-�������ǿ����е�����Ҳ���������ã����ʵ��֤ʵ�˸����ݣ�ʵ�鷽����������_______��
�� ����2�У�Cu2+������I-,��Ag+δ������I-��ԭ��_______��
�����ϣ�Ag+ + I- = AgI�� K1 =1.2��1016��2Ag+ + 2I- = 2Ag��+ I2 K2 = 8.7��108��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com