
����Ŀ��ˮ��������(DO)�Ǻ���ˮ���Ծ�������һ��ָ�꣬ͨ����ÿ��ˮ���ܽ������ӵ�������ʾ����λmg/L���ҹ����ر�ˮ�������������涨����������ˮԴ��DO���ܵ���5mg/L��ij��ѧС��ͬѧ���������װ��(�г�װ����)���ⶨij��ˮ��DO��
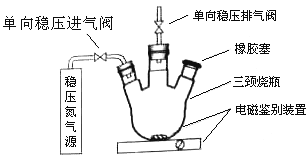
1���ⶨԭ����
��������£�O2��Mn2+����ΪMnO(OH)2:��2Mn2++O2+4OH-=2 MnO(OH)2�������������£�MnO(OH)2��I-����ΪI2:��MnO(OH)2+I-+H+��Mn2++I2+H2O(δ��ƽ)����Na2S2O3����Һ�ζ����ɵ�I2����2S2O32-+I2=S4O62-+2I-
2���ⶨ����
a����װװ�ã����������ԣ���N2�ž�������ֹͣ��N2��
b������ƿ�м���200mlˮ��
c������ƿ������Ѹ�ټ���1mlMnSO4������Һ(����)2ml����KI������Һ(����)������������������Ӧ����ȫ��
d���貢����ƿ�м���2ml����������Һ����Ӧ����ȫ����ҺΪ���Ի������ԡ�
e������ƿ��ȡ��40��00ml��Һ���Ե�����ָʾ������0��001000mol/L Na2S2O3��Һ���еζ�����¼���ݡ�
f������
g����������(����������ˮ���е�������ͼ����Լ���ˮ������ı仯)��
�ش��������⣺
��1����������������Һʱ����ȥ�����ܼ�ˮ�����ļ���Ϊ__________��
��2��������������ˮ�����й��Լ�Ӧѡ�������Ϊ__________��
���ζ�����ע��������Ͳ
��3�������������__________��
��4����ƽ��Ӧ���ķ���ʽ���仯ѧ����������Ϊ__________��
��5������fΪ__________��
��6������e�дﵽ�ζ��յ�ı�־Ϊ__________����ij�εζ�����Na2S2O3��Һ4.50ml��ˮ����DO=__________mg/L(����һλС��)����Ϊ����ˮԴ���˴β��DO�Ƿ��꣺__________(���ǻ��)
��7������d�м���������Һ��Ӧ������ҺpH���ͣ��ζ�ʱ��������Ե���д������������ԭ��(�����ӷ���ʽ��ʾ������д��2��)__________��
���𰸡�
��1�����ܼ�ˮ��к���ȴ
��2����
��3��ʹ��Һ��Ͼ��ȣ�������ɷ�Ӧ
��4��1��2��4��1��1��3
��5���ظ�����e�IJ���2-3��
��6����Һ��ɫ��ȥ(������ڲ���ɫ) 9.0 ��
��7��2H++S2O32-=S��+SO2��+H2O��SO2+I2+2H2O=4H++SO42-+2I-��4H++4I-+O2=2I2+2H2O(�����2��)
��������
�����������1��������ˮ�е��ܽ�������¶����߶���С�����ܼ�ˮ��п��Գ�ȥ�����ܼ�ˮ�������ʴ�Ϊ�����ܼ�ˮ��к���ȴ��
��2��������������ˮ�����й��Լ�Ӧѡ��ע��������ѡ����
��3���������ʹ��Һ��Ͼ��ȣ��ӿ췴Ӧ�����ʣ��ʴ�Ϊ��ʹ��Һ��Ͼ��ȣ�������ɷ�Ӧ��
��4�����ݻ��ϼ������غ㣬��Ӧ����ƽ��MnO(OH)2+2I-+4H+=Mn2++I2+3H2O���ʴ�Ϊ��1��2��4��1��1��3��
��5���ζ�����һ����Ҫ�ظ�����2-3�Σ��Ա��Сʵ������˲���fΪ�ظ�����e�IJ���2-3�Σ��ʴ�Ϊ���ظ�����e�IJ���2-3�Σ�
��6�������ӱ�����Ϊ�ⵥ�ʺ���Na2S2O3��Һ�ζ����ԭΪ�����ӣ���˵ζ���������Һ����ɫ��ʧ��n(Na2S2O3)= 0.001000mol/L��0.0045L=4.5��10-6mol�����ݷ�Ӧ�٢ڢ���O2~2 MnO(OH)2��~ 2I2~4S2O32-��n(O2)=![]() n(Na2S2O3)=1.125��10-6mol���ú�ˮ��DO=
n(Na2S2O3)=1.125��10-6mol���ú�ˮ��DO=![]() ��1.125��10-6��32=9��10-3g/L=9.0 mg/L��5 mg/L����꣬�ʴ�Ϊ����Һ��ɫ��ȥ(������ڲ���ɫ)��9.0���ǣ�
��1.125��10-6��32=9��10-3g/L=9.0 mg/L��5 mg/L����꣬�ʴ�Ϊ����Һ��ɫ��ȥ(������ڲ���ɫ)��9.0���ǣ�
��7����������������������·����绯��Ӧ�����ɵĶ�������Ҳ�ܹ������ɵĵ�������ͬʱ�����е�����Ҳ�ܹ�����������������Ӧ�����ӷ���ʽ�ֱ�Ϊ��2H++S2O32-=S��+SO2��+H2O��SO2+I2+2H2O=4H++SO42-+2I-��4H++4I-+O2=2I2+2H2O���ʴ�Ϊ��2H++S2O32-=S��+SO2��+H2O��SO2+I2+2H2O=4H++SO42-+2I-��4H++4I-+O2=2I2+2H2O��


 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����0.1 mol��þ�������������100 mL��2 mol��L��1��H2SO4��Һ�У�Ȼ���ٵμ�1 mol��L��1��NaOH��Һ����ش�
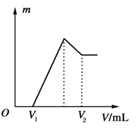
��1�����ڵμ�NaOH��Һ�Ĺ����г�������m�����NaOH��Һ�����V�仯����ͼ��ʾ����V1��160 mLʱ���������ĩ��n��Mg����________mol��V2��________ mL��
��2�����ڵμ�NaOH��Һ�����У���ʹMg2����Al3���պó�����ȫ�������NaOH��Һ�����V��NaOH����________ mL��
��3�����������Ϊ0.1 mol������Mg�۵����ʵ�������Ϊa����100 mL��2 mol��L��1��H2SO4�ܽ�˻������ټ���450 mL��1 mol��L��1��NaOH��Һ�����ó�������Al��OH��3�������������a��ȡֵ��Χ��________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�����й����л��������˵����ȷ����
A��2-������Ҳ���춡��
B�����Ǻ���ά�ض����ڸ߷��ӻ�����
C����ʹ��ˮ��ɫ����Ϊ������ȡ����Ӧ
D��C3H6O2��3��ͬ���칹��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�������л����������ȷ����
A��CH3CH2CH(CH3)CH(CH3)OH ��3-��-2-�촼
B��CH3CH(CH3)CH=CH2��3-����ϩ
C��CH3CH2C(CH3)2CH3��2��2-������
D��CH2Cl-CH2Cl����������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��һ�ֹ�ҵ�Ʊ�SrCl2��6H2O�������������£�
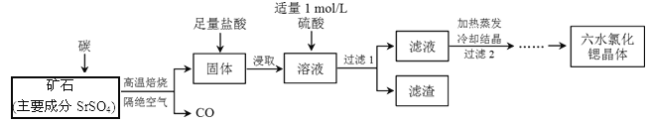
��֪����M(SrCl2��6H2O)=267 g/mol��
�� Kap(SrSO4)= 3.3��10��7��Kap(BaSO4)=1.1��10��10��
�۾������ȡ����Һ�г�����Sr2+��Cl���⣬����������Ba2+���ʡ�
��1�������������±��գ���2 mol SrSO4��ֻ��S����ԭ��ת����16mol���ӡ�д���÷�Ӧ�Ļ�ѧ����ʽ��_____________________________________________��
��2������2����������������______��_________��
��3�����������Ŀ����_________________��Ϊ�����ԭ�ϵ������ʣ���Һ��Sr2+��Ũ��Ӧ������_________ mol/L��ע����ʱ��Һ��Ba2+Ũ��Ϊ1��10��5 mol/L����
��4����Ʒ���ȼ�⣺��ȡ1.000 g��Ʒ�ܽ�������ˮ�У������м��뺬AgNO3 1.100��10��2 mol��AgNO3��Һ����Һ�г�Cl�D�⣬����������Ag+��Ӧ�����ӣ�����Cl�D��ȫ�������ú�Fe3+����Һ��ָʾ������0.2000 mol/L��NH4SCN����Һ�ζ�ʣ���AgNO3��ʹʣ���Ag+ ��AgSCN��ɫ��������ʽ������
�ٵζ���Ӧ�ﵽ�յ��������_______________________________��
�����ζ�������ȥ����Ũ�ȵ�NH4SCN��Һ20.00 mL�����Ʒ��SrCl2��6H2O�������ٷֺ���Ϊ____________������4λ��Ч���֣���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����֪2A2(g)+B2(g) ![]() 2C3(g)����H=��Q1 kJ/mol(Q1��0)�� ��һ���д����Ĺ̶��ݻ��������м���2 molA2��1 molB2����500��ʱ��ַ�Ӧ����ƽ���C3��Ũ��Ϊw mol��L��1���ų�����ΪQ2 kJ��
2C3(g)����H=��Q1 kJ/mol(Q1��0)�� ��һ���д����Ĺ̶��ݻ��������м���2 molA2��1 molB2����500��ʱ��ַ�Ӧ����ƽ���C3��Ũ��Ϊw mol��L��1���ų�����ΪQ2 kJ��
��1���ﵽƽ��ʱ�� A2��ת����Ϊ________��
��2���ﵽƽ�������ԭ������ͨ�������������A2��ת���ʽ�_____�����������С�����䡱��
��3������ԭ���������У�ֻ����2mol C3��500��ʱ��ַ�Ӧ��ƽ�����������Q3 kJ��C3Ũ�� (������=����)w mol��L��1��Q1��Q2��Q3 ֮��������ֹ�ϵ
��4����˵���÷�Ӧ�Ѿ��ﵽƽ��״̬���� ��
a��v(C3)=2 v (B2)��
b��������ѹǿ���ֲ���
c��2v��(A2)=v��(B2)
d�������ڵ��ܶȱ��ֲ���
��5���ı�ijһ�������õ���ͼ�ı仯����
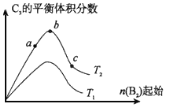
��ͼ��T��ʾ�¶ȣ�n��ʾ���ʵ��������ɵó��Ľ�����ȷ���� ��
a����Ӧ����c��b��a
b���ﵽƽ��ʱA2��ת���ʴ�СΪ��b��a��c
c��T2��T1
d��b��A2��B2�����ʵ���֮��Ϊ2��1
��6����������������Ϊ��ѹ����(��Ӧǰ�����ͬ)����ʼʱ����2 molA2��l molB2��500��ʱ��ַ�Ӧ��ƽ��ų�����Q4 kJ����Q2 Q4 �����������������=������
��7�����д�ʩ����ͬʱ��߷�Ӧ���ʺ�B2��ת�����ǣ���ѡ����ţ� ��
a��ѡ���ʵ��Ĵ��� b������ѹǿ
c����ʱ�������ɵ�C3 d�������¶�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����ͼ��ʾΪһ����AlCl3��Һ�м���NaOH��Һ����Al(OH)3��ɫ������������NaOH�����ʵ���֮��Ĺ�ϵ���ߡ�

��ش��������⣺
��1��A��ʱ�Ѳμӷ�Ӧ��AlCl3��NaOH�����ʵ���֮��Ϊ________��
��2��AB����������ʾ�ķ�Ӧ�����ӷ���ʽΪ_________________________��
��3����B�����ɵ���Һ��ͨ�������̼���ɹ۲쵽��������_______________________��
��4������0.1 mol NH4Al(SO4)2����Һ����μ���5 mol��L��1 NaOH��Һ����ʼ������Һ�г��ְ�ɫ�����������ࣻһ��ʱ����д̼�����ζ�������ݳ�������ɫ�������ٲ�������ʧ��������ͼ�л������ɳ��������ʵ��������NaOH��Һ����Ĺ�ϵʾ��ͼ��

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����FeCl3��Һʴ��ͭ�������·��Ĺ����У���Һ������Fe2+��Fe3+��Cu2+����������Դ���պ������壬�����ǻ��ս���ͭ�Ϳ�ʴҺ����������ͼ���ش��������⣺
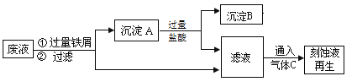
��1������A�к��еĵ����� ��
��2��ͨ�������C�Ļ�ѧʽ�� ��
��3����Һ������C��Ӧ���ɿ�ʴҺ�����ӷ���ʽ�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ���Ҷ����������D������ʯ�����ѽ�õ���ϩ���ϳɡ��ش��������⣺
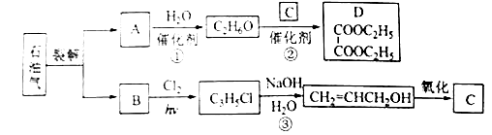
��1��B��AΪͬϵ�B�Ľṹ��ʽΪ_______��
��2����Ӧ���Ļ�ѧ����ʽΪ___________���䷴Ӧ����Ϊ__________��
��3����Ӧ���ķ�Ӧ����Ϊ____________��
��4��C�Ľṹ��ʽΪ___________��
��5����Ӧ���Ļ�ѧ����ʽΪ______��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com