
����Ŀ��������H�Ǻϳ�������Ѫ�ܼ���ҩ����м��壬��ͨ����ͼ��ʾ;���ϳɣ�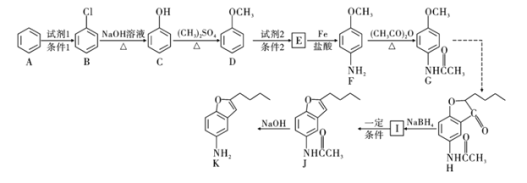
��֪��![]()
![]() ��
��![]() �����ױ�����
�����ױ�����![]() ��
��
![]()
![]() ��
��
�ش��������⣺
��1��д��������D������Ϊ______________��F�еķǺ�������������Ϊ______________
��2��д��![]() �ķ�Ӧ���ͣ�______________��������G�ķ���ʽΪ______________��
�ķ�Ӧ���ͣ�______________��������G�ķ���ʽΪ______________��
��3��д��![]() ��Ӧ�Ļ�ѧ����ʽ��__________________________________________��
��Ӧ�Ļ�ѧ����ʽ��__________________________________________��
��4��������L��Eͬ���칹�壬д��ͬʱ������������L������һ�ֽṹ��ʽ��____________________________��
������![]() ��Һ��Ӧ����ɫ
��Һ��Ӧ����ɫ
�ڱ�����������ȡ����������һ��Ϊ![]()
�۱����ϵ�һ��ȡ��������ͬ���칹��
��5���ϳ�;���У�Fת��ΪG��Ŀ����___________________________________��
��6�����������ϳ�·�ߣ��Ա���![]() Ϊԭ��
Ϊԭ��![]() ���Լ���ѡ
���Լ���ѡ![]() ������Ʊ�
������Ʊ�![]() �ĺϳ�·�ߡ�________________
�ĺϳ�·�ߡ�________________
���𰸡�������![]() ������������������ѡ��������ѡ��ױ���
������������������ѡ��������ѡ��ױ���![]() �Ѽ� ˮ�ⷴӦ
�Ѽ� ˮ�ⷴӦ![]() ��ȡ����Ӧ
��ȡ����Ӧ![]()
![]()
 ��
��
 ��
�� ������������ֹ�ϳɹ����б�����
������������ֹ�ϳɹ����б����� ![]() ��
��
��������
��1��D��![]() ������������
������Ϊ������![]() ������������������ѡ��������ѡ��ױ���
������������������ѡ��������ѡ��ױ���![]() ��FΪ
��FΪ![]() ����Ǻ�������Ϊ������
����Ǻ�������Ϊ������
��2����ͼ ֪��
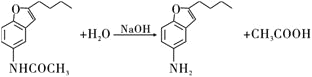
֪��![]() �ķ�Ӧ����Ϊˮ�ⷴӦ
�ķ�Ӧ����Ϊˮ�ⷴӦ![]() ��ȡ����Ӧ
��ȡ����Ӧ![]() ��GΪ
��GΪ �������ʽΪ
�������ʽΪ![]() ��
��
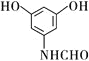
��3����֪J��������������NaOHˮ��Һ�лᷢ��ˮ�ⷴӦ���䷴Ӧ����ʽΪ�� ��
�� ��
��
��4��L��E��![]() ��ͬ���칹�壬����
��ͬ���칹�壬����![]() ��Һ��Ӧ����ɫ��˵��һ���з��ǻ���������������ȡ����������һ��Ϊ
��Һ��Ӧ����ɫ��˵��һ���з��ǻ���������������ȡ����������һ��Ϊ![]() ��������һ��ȡ��������ͬ���칹�壬�ۺϷ�����������L������
��������һ��ȡ��������ͬ���칹�壬�ۺϷ�����������L������ ��
�� ��
��
��5���ϳ�;���У�Fת��ΪGʱ![]() ������Ӧ����
������Ӧ����![]() ������ת��Ϊ������Ŀ���DZ�����������ֹ���ںϳɹ����б�������
������ת��Ϊ������Ŀ���DZ�����������ֹ���ںϳɹ����б�������
��6������![]() Ϊԭ���Ʊ�
Ϊԭ���Ʊ�![]() �������ɱ�����������Ӧ�������������������������ٷ�����ԭ��Ӧ���ɰ��������������������ȡ����Ӧ��������
�������ɱ�����������Ӧ�������������������������ٷ�����ԭ��Ӧ���ɰ��������������������ȡ����Ӧ��������![]() ����Ӧ������Ϊ��
����Ӧ������Ϊ��![]() ��
��


 Сѧ���AB��ϵ�д�
Сѧ���AB��ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��ijѧ������ͼ��ʾװ����ȡ���������ռ���������ʵ�顣
������������⣺
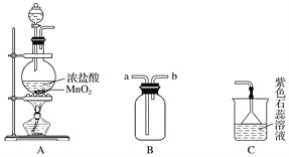
(1)װ��A�з�����Ӧ�����ӷ���ʽΪ______________________________________��
(2)װ��B��ͼ����ʾ���ţ�������A�Ľӿ���________(�����)��
(3)ʵ���пɹ۲쵽װ��C����ɫʯ����Һ��������__________________����ԭ��______________________________________________________________________��
(4)ȡ��B�еļ���ƿ����������ͭ�ķ�Ӧʵ�飬Ӧ�ý��еIJ�����______________��Ȼ�����̷�����������ļ���ƿ�С�ʵ������ͬѧ���Ӧ���ڼ���ƿ���ȷ�������ˮ��ϸɳ������Ϊ__________(������Ҫ����������Ҫ��)�ţ�������____________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����25�桢101kPa�����£�C��s����H2��g����CH3COOH��l����ȼ���ȷֱ�Ϊ393.5kJ/mol��285.8kJ/mol��870.3kJ/mol����2C(s)+2H2(g)+O2(g)=CH3COOH(l)�ķ�Ӧ��Ϊ
A. ![]() B.
B. ![]() C.
C. ![]() D.
D. ![]()
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����![]() Ϊ�����ӵ�������ֵ������˵����ȷ���ǣ� ��
Ϊ�����ӵ�������ֵ������˵����ȷ���ǣ� ��
A.��״���£�![]() ���к���
�����![]() ��������
��������
B.��״���£�![]() �ֱ�������������ͭ��ַ�Ӧ��ת�Ƶĵ�������֮��Ϊ
�ֱ�������������ͭ��ַ�Ӧ��ת�Ƶĵ�������֮��Ϊ![]()
C.![]() ��Һ�У������Ӻ������ӵ���Ŀ֮��Ϊ
��Һ�У������Ӻ������ӵ���Ŀ֮��Ϊ![]()
D.![]() �к��еĹ��ۼ���Ϊ
�к��еĹ��ۼ���Ϊ![]()
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��Ϊ�˺������û�ѧ�ܣ�ȷ����ȫ���������������Ҫ��ֿ��ǻ�ѧ��Ӧ�ķ�Ӧ�ȣ�����ȡ��Ӧ��ʩ����ѧ��Ӧ�ķ�Ӧ��ͨ����ʵ����вⶨ��Ҳ�ɽ����������㡣
��1��ʵ���ã�5g�״���CH3OH��Һ���������г��ȼ�����ɶ�����̼�����Һ̬ˮʱ�ͷų�113.5kJ�����������ʾ�״���ȼ���ȵ��Ȼ�ѧ����ʽΪ�� ��
��2���������������Ȼ�ѧ����ʽ����a b����������������������������
H2(g)+ 1/2O2(g)��H2O(g) ��H1��a kJ��mol-1
H2(g)+ 1/2O2(g)��H2O(l) ��H2��b kJ��mol-1
��3����1mol��̬������ij�ֹ��ۼ���Ҫ���յ������м��ܡ��ӻ�ѧ���ĽǶȷ�������ѧ��Ӧ�Ĺ��̾��Ƿ�Ӧ��Ļ�ѧ�����ƻ���������Ļ�ѧ�����γɹ��̡��ڻ�ѧ��Ӧ�����У���ѧ����Ҫ�����������γɻ�ѧ���ֻ��ͷ�������
��ѧ�� | H��H | N��H | N��N |
����/kJ��mol��1 | 436 | 391 | 945 |
��֪��ӦN2(g)��3H2(g)![]() 2NH3(g) ��H��a kJ��mol��1���Ը��ݱ������м������ݹ���a��ֵ��_______________(ע����+����������)��
2NH3(g) ��H��a kJ��mol��1���Ը��ݱ������м������ݹ���a��ֵ��_______________(ע����+����������)��
��4�����ݸ�˹���ɿ��Զ�ijЩ����ͨ��ʵ��ֱ�Ӳⶨ�Ļ�ѧ��Ӧ�ķ�Ӧ�Ƚ������㡣����ˮú���ϳɶ����ѵ�������Ӧ���£�
�� 2H2(g) + CO(g)![]() CH3OH(g)����H ����90.8 kJ��mol��1
CH3OH(g)����H ����90.8 kJ��mol��1
�� 2CH3OH(g)![]() CH3OCH3(g) + H2O(g)����H����23.5 kJ��mol��1
CH3OCH3(g) + H2O(g)����H����23.5 kJ��mol��1
�� CO(g) + H2O(g)![]() CO2(g) + H2(g)����H����41.3 kJ��mol��1
CO2(g) + H2(g)����H����41.3 kJ��mol��1
�ܷ�Ӧ��3H2(g) + 3CO(g)![]() CH3OCH3(g) + CO2(g)����H�� ��
CH3OCH3(g) + CO2(g)����H�� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��ij�о�С��������ͼװ��̽���¶ȶ�һ����̼��ԭ��������Ӱ�졣
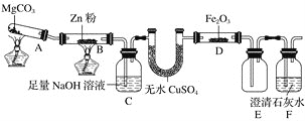
(1)MgCO3�ķֽ����Ϊ____________��
(2)װ��C��������______________������β���ķ���Ϊ______________________��
(3)���о�С���Ϊ���飬����ͼװ�ý��жԱ�ʵ�飬�����þƾ��ơ������þ�����ƶ�װ��D���ȣ���Ӧ�����Ϊ��ɫ��ĩ(������)������ֱ��ò����������ʵ�顣��֪Fe2����K3[Fe(CN)6](���軯��)��Һ��Ӧ���ɴ���������ɫ�����軯����������
������õ��ĺ�ɫ��ĩ��______________��
�ڼ��鲽��1�з�Ӧ�����ӷ���ʽΪ______________��
�����鲽��4�У���Һ����ԭ��Ϊ______________________����Һ��ɫ���ܵ�ԭ������֤����Ϊ________________________��
�ܴ�ʵ�鰲ȫ���ǣ���ͼװ�û��ɲ�ȡ�ĸĽ���ʩ��__________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�����������Ҫ�ɷ���������(Al2O3)���ڹ�ҵ���Dz��õ���������ķ����ó���������
(1)������������Ԫ������Ԫ�ص�����֮����__________��
(2)�����ַ���������170 t��������80%�������������Ͽ�ұ����������__________�֣�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����ͼ����ѧ��ѧ�г����ڻ����ķ�����ᴿ��װ�ã������װ�ûش����⣺
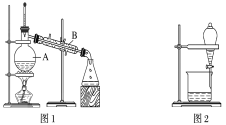
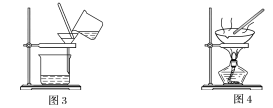
(1)װ��ͼ1��B��������________________��ͼ2��©����������________________��A��һ��Ҫ�������Ƭ����������__________________________�����й�������ʵ�����˵��һ����ȷ����_��
A��ͼ1ʵ���У�����һ��ʱ�����δ�������Ƭ��Ӧ�������ӣ��Է�����Σ��
B��ͼ2ʵ���У�Ӧ���������л��ܼ����¶˵����зų�
C��ͼ3ʵ���У������ò�������©���н��裬�Լӿ�����ٶ�
D��ͼ4ʵ���У����������н϶��������ʱ����ֹͣ����
(2)����һƿA��B�Ļ��Һ����֪���ǵ��������±���
���� | �۵�/�� | �е�/�� | �ܶ�/g��cm��3 | �ܽ��� |
A | ��11.5 | 198 | 1.11 | A��B���ܣ��Ҿ�������ˮ�;ƾ� |
B | 17.9 | 290 | 1.26 |
�ݴ˷�������A��B������ѡ����ͼ�е�ͼ________________��ʾ������
(3)��ͼ2��ʾʵ���У����÷ֲ�������֪����һ��Һ������ˮ�����������һ�ּ����жϷ�����____��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��NAΪ�����ӵ�������ֵ������˵����ȷ����
A. 1.4gC2H4��C3H6 �Ļ��������������̼ԭ������Ϊ 0. lNA
B. 1.0 mol CH4 �� Cl2 �ڹ����·�Ӧ���ɵ� CH3Cl ������Ϊ 1.0NA
C. ��״���£�22. 4LC2H6 ���������Թ��ۼ�����ĿΪ 7NA
D. 50mL l2mol��L ��Ũ���������� MnO2 ��Ӧ��ת�Ƶ�������Ϊ 0. 3NA
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com