
(1)�й��Ŵ��Ĵ���֮һ�����ڻ�ҩ�����ı�ը��ӦΪ
2KNO3
��3C��S��A��N2��3CO2��(����ƽ)�ٳ�S�⣬����Ԫ�صĵ縺�ԴӴ�С����Ϊ________��
�����������У�A�Ļ�ѧ������Ϊ________�������Թ��ۼ��ķ��ӵ�����ԭ�ӹ���ӻ�����Ϊ________����________���ӣ�
����֪CN����N2�ṹ���ƣ�����HCN�����ЦҼ���м���Ŀ֮��Ϊ________��
(2)ԭ������С��36��Ԫ��Q��T�������ڱ��мȴ���ͬһ������λ��ͬһ�壬��ԭ������T��Q��2��T�Ļ�̬ԭ����Χ����(�۵���)�Ų�Ϊ________��Q2+��δ�ɶԵ�������________��
(3)��CrCl3��ˮ��Һ�У�һ�������´������Ϊ[CrCln(H2O)6��n]x+(n��x��Ϊ������)�������ӣ�����ͨ�������ӽ�����֬(R��H)���ɷ������ӽ�����Ӧ��
[CrCln(H2O)6��n]x+��xR��H![]() Rx[CrCln(H2O)6��n]x+��xH+
Rx[CrCln(H2O)6��n]x+��xH+
����������H+���к͵ζ����������x��n��ȷ�������ӵ���ɣ�����0.0015 mol��[CrCln(H2O)6��n]x+����Һ����R��H��ȫ�������к����ɵ�H+��Ũ��Ϊ0.1200 mol��L��1��NaOH��Һ25.00 ml���������ӵĻ�ѧʽΪ________��
(4)�������������Ļ����15 g�м���ϡ����150 mL����״���·ų�����1.68 L��ͬʱ��������������ʣ�࣮����Һ�е���KSCNδ����ɫ�仯��Ϊ���к��������ᣬ��ʹFe2+��ȫת��Ϊ������������������3 mol/L������������Һ200 mL����ԭ��������ʵ���Ũ��Ϊ________mol/L��

| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ��2012-2013ѧ���Ĵ�ʡ�ɶ����и߶�10�½��Կ��Ի�ѧ�Ծ����������� ���ͣ������
(7��)(1)�й��Ŵ��Ĵ���֮һ�����ڻ�ҩ�����ı�ը��ӦΪ��
2KNO3+ 3C+S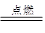 A+N2��+3CO2��������ƽ��
A+N2��+3CO2��������ƽ��
�ٳ� S �⣬����Ԫ�صĵ縺�ԴӴ�С����Ϊ_________________________;
���������У�A �ĵ���ʽΪ__________________�������Թ��ۼ��ķ��ӵ�����ԭ�ӹ���ӻ�����____________;
����֪CN����N2�ṹ���ƣ�����HCN�����ЦҼ���м���Ŀ֮��Ϊ____________��
(2)ԭ������С��36��Ԫ��Q��T�������ڱ��мȴ���ͬһ������λ��ͬһ�壬��ԭ������T��Q��2��T�Ļ�̬ԭ����Χ����(�۵���)�Ų�ʽΪ_______________________________��Q2����δ�ɶԵ�������________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012-2013ѧ���Ĵ�ʡ�Ѿ���ѧ�߶�10���¿���ѧ�Ծ����������� ���ͣ������
(1)�й��Ŵ��Ĵ���֮һ�����ڻ�ҩ�����ı�ը��ӦΪ��
2KNO3+ 3C+S A+N2��+3CO2��������ƽ��
A+N2��+3CO2��������ƽ��
�� �� S �⣬����Ԫ�صĵ縺�ԴӴ�С����Ϊ_________________________;
���������У�A �ĵ���ʽΪ__________________ �������Թ��ۼ��ķ��ӵ�����ԭ�ӹ���ӻ�����____________;
����֪CN����N2�ṹ���ƣ�����HCN�����ЦҼ���м���Ŀ֮��Ϊ____________��
(2)ԭ������С��36��Ԫ��Q��T�������ڱ��мȴ���ͬһ������λ��ͬһ�壬��ԭ������T��Q��2��T�Ļ�̬ԭ����Χ����(�۵���)�Ų�ʽΪ_______________________________��Q2����δ�ɶԵ�������________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2014���Ĵ�ʡ�߶�10���¿���ѧ�Ծ��������棩 ���ͣ������
(1)�й��Ŵ��Ĵ���֮һ�����ڻ�ҩ�����ı�ը��ӦΪ��
2KNO3+ 3C+S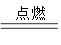 A+N2��+3CO2��������ƽ��
A+N2��+3CO2��������ƽ��
�� �� S �⣬����Ԫ�صĵ縺�ԴӴ�С����Ϊ_________________________;
���������У�A �ĵ���ʽΪ__________________ �������Թ��ۼ��ķ��ӵ�����ԭ�ӹ���ӻ�����____________;
����֪CN����N2�ṹ���ƣ�����HCN�����ЦҼ���м���Ŀ֮��Ϊ____________��
(2)ԭ������С��36��Ԫ��Q��T�������ڱ��мȴ���ͬһ������λ��ͬһ�壬��ԭ������T��Q��2��T�Ļ�̬ԭ����Χ����(�۵���)�Ų�ʽΪ_______________________________��Q2����δ�ɶԵ�������________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2014���Ĵ�ʡ�߶�10�½��Կ��Ի�ѧ�Ծ��������棩 ���ͣ������
(7��)(1)�й��Ŵ��Ĵ���֮һ�����ڻ�ҩ�����ı�ը��ӦΪ��
2KNO3+ 3C+S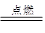 A+N2��+3CO2��������ƽ��
A+N2��+3CO2��������ƽ��
�ٳ� S �⣬����Ԫ�صĵ縺�ԴӴ�С����Ϊ_________________________;
���������У�A �ĵ���ʽΪ__________________�������Թ��ۼ��ķ��ӵ�����ԭ�ӹ���ӻ�����____________;
����֪CN����N2�ṹ���ƣ�����HCN�����ЦҼ���м���Ŀ֮��Ϊ____________��
(2)ԭ������С��36��Ԫ��Q��T�������ڱ��мȴ���ͬһ������λ��ͬһ�壬��ԭ������T��Q��2��T�Ļ�̬ԭ����Χ����(�۵���)�Ų�ʽΪ_______________________________��Q2����δ�ɶԵ�������________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��ͬ���� ���ͣ������
 A+ N2�� +3CO2��
A+ N2�� +3CO2�� �鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com