
Ϊ�˲ⶨ�������ƺ�̼���ƹ���������̼���Ƶ������������ס�����λͬѧ�ֱ���������µ�ʵ�鷽����
(I)��ͬѧ�ķ����ǣ���mg��Ʒ�ܽ⣬�ӹ����Ȼ�����Һ�����ˡ�ϴ�ӡ���ɣ��Ƶù���ng��
��1���������̼���Ƶ���������Ϊ(��m��n��ʾ) ����ͬѧϴ�ӳ����ľ�������� ��
��2��Ca2+��Ba2+������ʹCO32-������ȫ����ʹ���Ȼ�����Һ���Ȼ�����Һ���õĽ�����и��ߵľ�ȷ�ȣ�ԭ���ǣ��� ;
�� ��
(��)��ͬѧ�ķ�����ͼ��ʾ��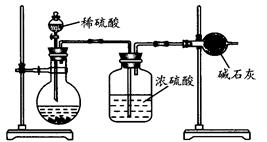
��1��������ͬѧ��ʵ��װ��ͼ��������ÿ��ʵ���У���ɲⶨ��������Ҫ���� �γ������������ո�ͬѧ�ķ������вⶨ�������ʵ��������ڽϴ��������Ϊ��Ҫԭ�������(��д����)��
�� ��
�� ��
��2����ͬѧ�ķ�����װ���д���һ����ȱ�ݣ���������Ľ��ķ���(���������������ػ�ͼ)�� ��
��1��106n/197m ������м�������ˮ���պ�û�����������ã����2��3��
��2��������������ˮ������Ӱ�������������̼�ᱵ��Ħ��������̼��ƵĴ����ij��������������С
(��)��1��4 ��2�֣���װ����ԭ�е�CO2Ҳ����ʯ�����գ��ڷ�Ӧ������CO2δ����ȫ���գ��ۿ����е�H2O��CO2Ҳ����ʯ�����գ��ܼ��������������㣬δ�ܽ�̼������ȫ��Ӧ����װ�õ������Բ��á�����ѡ���㣬��������Ҳ�ɣ���2�֣�
��2��������ķ���װ��ǰ���װͨ������������Һ��װ�ã��ڼ�ʯ��װ�ú�������һ����ʯ��װ�ã��Է����տ����е�ˮ��CO2��
�����������������1����̼���Ƶ�����Ϊx
��ͬѧϴ�ӳ����ľ�������ǣ�������м�������ˮ��Ȼ����ˣ��ظ�2-3��
��2�������Ȼ��ƵĻ�����Ӧ���ɵ�������������ˮ����ʹ������������ʴ�Ϊ����Ca��OH��2w����ˮ������Ӱ�������������BaCl2��CaCl2����Է������������ij��������������С
����1��ʵ��ʱҪ�������²�����������ʯ�ҵ�����������װ�á���һ��������Ʒ�м���������ϡ���ᡢ��Ӧ�����������ʯ�ҵ��������ʴ�Ϊ��4 ��װ����ԭ�еĿ����еĶ�����̼Ҳ����ʯ�����գ��ڷ�Ӧ�����Ķ�����̼δ����ȫ���գ��ۿ����е�ˮ�Ͷ�����̼Ҳ����ʯ�����բܼ��������������㣬δ�ܽ�̼������ȫ��Ӧ��װ�õ������Բ���
��2���÷�����û�г�ȥװ���п����е�ˮ�����Ͷ�����̼����ʯ��Ҳ��������������е�ˮ�Ͷ�����̼���ʴ�Ϊ��������ķ���װ��ǰ���װͨ������������Һ��װ�ã��ڼ�ʯ��װ�ú�������һ����ʯ��װ�ã��Է����տ����е�ˮ�Ͷ�����̼��
���㣺����ʵ��̽��������ɳɷֺͺ����Լ�ʵ�����ݴ����������������֪ʶ��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
����ȥNaHCO3��Һ��������Na2CO3����ѡ�õ��Լ���
| A������ʯ��ˮ | B��NaOH��Һ | C������ | D��CO2 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
����˵������ȷ���ǣ� ��
| A����������������ø�ƿ���� |
| B���ƿ��Ա�����CCl4Һ���У���֪���ܶ�CCl4 > Na�� |
| C��Na2O2����ΪDZˮͧ�Ĺ����� |
| D��ʵ���ʣ��������ɷŻ�ԭ�Լ�ƿ�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
��agþ���Ͻ�Ͷ�뵽x mL 2mol/L�������У�������ȫ�ܽ���ټ���y mL 1mol��L������������Һ���õ��ij������������Ϊ��a+1.7��g������˵������ȷ����
| A��þ���Ͻ������ᷴӦת�Ƶ�������Ϊ0.lNA | B��x��2y |
| C��������Mg(OH)2��Al(OH)3�Ļ���� | D��2x��y |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
���������ǡ�DZͧ�ϵĺ˷�Ӧ����ʹ����Һ�����ƺϽ�(�����ƺ͵������ۺ϶���)�����Ƚ��ʣ��й�˵������ȷ����
| A���Ͻ���۵�һ�����ֽ����� |
| B�����ƺϽ���Ͷ��һ������ˮ�пɵ���ɫ��Һ����n(Al)��n(Na) |
| C�����ƺϽ�Ͷ�뵽�����Ȼ�ͭ��Һ�У�����������ͭ����Ҳ������ͭ���� |
| D����m g��ͬ��ɵ����ƺϽ�Ͷ�����������У��ų���H2Խ�࣬��������������ԽС |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��16�֣�������ҵ��ϴ�ֲ�ʱ�����ķ�Һ��Ҫ�ɷ�ΪFe2+��H+��Cl-�������������������÷�Һ������������Ʊ�������Ϳ�ϡ� 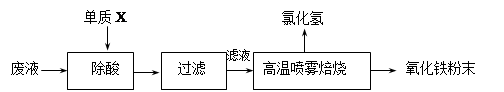
��1������X�Ļ�ѧʽ�� ��
��2���Ȼ�������Һ��������������ת��ΪHCl�������������ĩ���йصĻ�ѧ����ʽ����Ϊ�� ��
��3��ij����Ϳ���г�����Fe2O3�⣬������������CuO��FeO�е�һ��,�����ʵ�鷽����̽��������Ϳ����������ijɷ֡�
�� �����������
����1��������CuO
����2��������FeO
�� ����Ʒ�������֤�������裬д��ʵ�鲽�衢Ԥ������ͽ��ۡ�
��ѡ�Լ������ۡ�3mol?L-1H2SO4��0.1 mol?L-1����KMnO4��Һ��10%NaOH��Һ��10%H2O2��KSCN��Һ
| �������� | Ԥ������ͽ��� |
| ����1��ȡ������Ʒ���Թ��У� ____________________________________________ | ��Ʒȫ���ܽ⣬�õ��������Һ�� |
| ����2�� ����3�� | ___________________________ ___________________________ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
Cu(NO3)2����Ҫ�Ļ���ԭ�ϡ��ش����й�ҵ�����ͻ�ѧʵ���еļ������⡣
��1��ʵ�������Ʊ�0.3 mol Cu (NO3)2���壬�ס�����ͬѧ�ֱ����ʵ�鷽�����£�
�ף�Cu 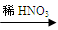 Cu(NO3)2 �ң�Cu
Cu(NO3)2 �ң�Cu 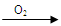 CuO
CuO 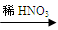 Cu(NO3)2
Cu(NO3)2
�ٴ���ɫ��ѧ�ĽǶȷ����� ͬѧ�ķ�������������ͬѧ�ɽ�Լ2mol��L��ϡ���� mL��
����ͬѧʵ��ʱͨ��O2�����ʲ��˹���Ϊ���ڹ۲�Ϳ��Ʋ���O2�����ʣ���ѡ��
����������ĸ��ţ���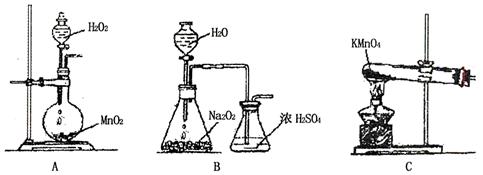
��������װ�ã�����ͼ������װ�õ�·��ͨ��2mol����ʱ��Cu�缫�����ţ� ��
��a������64g ��b������64g ��c����������64g��Ҳ���ܼ���64g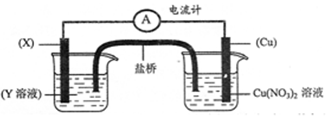
��2����ijУ��ѧ��ȤС���ͬѧ������ͭ�����ȷֽ����̽�����������������ͼװ�ü���Cu(NO3)2���壨���ȼ��г�װ��δ�����������ȹ��̷��֣�װ�â��еĹ�����Ϊ��ɫ��װ�â��������ݲ��������������Ĺ�������ʧ��ʯ����Һ��Ϊ��ɫ��Һ�弸������ѹ��װ�â��С���д��Cu(NO3)2���ȷֽ�Ļ�ѧ����ʽ�� ��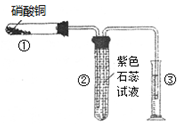
����װ���Т��Թܵ���Ƥ�������������ס��ԭ���� ��
��3��NO2��O2������NaNO3������ȼ�ϵ�أ���ԭ������ͼ���õ����ʹ�ù�����ʯīI�缫������������Y����缫��ӦʽΪ ��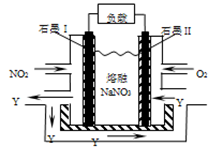
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��16�֣�ijУ����ͬѧ�߿�ǰ�������������α�ʵ�飬����ش�ʵ������е�������⣺
��1�����ȷ�Ӧʵ�飺ȡ�����������۰��α��е�ʵ��װ�ã���ͼ���������ȷ�Ӧ������Ӧ
�����á����顱�������ᣬ��Ӧ�����Һ�еμ�KSCN��Һ��������Һ��Ѫ��ɫ��
�ٳ������������ԭ���������顱�п��ܻ���û��Ӧ���
�����������⣬����һ�ֿ���ԭ������ ��
�� ��Ҫ֤����Ӧ���á����顱�к��н����������� ���ѧʽ����Һ����������Ӧ�����ӷ���ʽΪ�� ��
��2����ѧ��Ӧ����Ӱ�����ص�̽��ʵ�飺
KI�ھ��ữ����Һ�б����������ķ�ӦʽΪ��4H+ + 4I-+ O2 = 2I2 + 2H2O���÷�Ӧ���������¶ȡ���ȡ��ܼ����Լ�Ũ�ȵ�Ӱ�죬���õ���������ɫ��Ӧ���۲�÷�Ӧ�����ʡ���֪������������ɫ��Ӧ���¶�����ʱ�����Ȼή�ͣ�����75��������ɫ������Ũ��Խ����ɫԽ��������ɫҲԽ�
ʵ��С������0.8 mol��L-1 KI��Һ��0.1 mol��L-1H2SO4��Һ��������Һ����̽���¶ȡ���ȶ�������Ӧ���ʵ�Ӱ�죬��������A��C����ʵ�飬����ʵ���������±���
| ��� | �¶�/�� | H2SO4���/mL | KI��Һ���/mL | H2O���/mL | ������Һ / mL | ������ɫʱ�� /s |
| A | 39 | 10 | 5 | 5 | 1 | 5 |
| B | TB | 10 | 5 | 5 | 1 | û������ɫ |
| C | 5 | 10 | 5 | 5 | 1 | 39 |
| D | | | | | | t |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��֪AΪ����ɫ���壬T��RΪ���ֳ�������;�ܹ�Ľ������ʣ�D�Ǿ��д��Եĺ�ɫ���壬C����ɫ��ζ�����壬H�ǰ�ɫ�������ƶ�A��W���ʸ���ʲô���ش��������⡣
��1��д���������ʵĻ�ѧʽ��D�� N�� ��
��2��A��ˮ�ķ�Ӧ�������к��ַ�Ӧ���� ������ţ���
A�����Ϸ�Ӧ B��������ԭ��Ӧ C�����ӷ�Ӧ D���û���Ӧ
��3����Ҫȷ��E��Һ���Ƿ���W���ʣ���ѡ�Լ�Ϊ ������ţ���
A����ˮ��KSCN��Һ B��KSCN��Һ
C��Ũ��ˮ D�����Ը��������Һ
��4��B��E��͵õ�H���ڳ�ʪ�����б��M�Ĺ����У����ܹ۲쵽������ ��
��5��B��R��Ӧ����N�����ӷ���ʽ�� ��
M��W�����ӷ���ʽ�� ��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com