
����Ŀ����֪X��һ�־��й���ζ�ĺϳ����ϣ���ͼΪ�ϳ�X��ij�����̣�

��ʾ����![]() �������ձ�����Ϊ��COOH��
�������ձ�����Ϊ��COOH��
��D�IJ���������������һ�����ҵ�ʯ�ͻ���ˮƽ��
�����������Ϣ���ش��������⣺
(1)C�����й����ŵ�������__________��E�Ľṹ��ʽ��________��
(2)D+E��X�Ļ�ѧ��Ӧ����Ϊ________��Ӧ��
(3)����A��B��C��D��E��X���������У���Ϊͬϵ�����____________________��
(4)C��һ��ͬ���칹��F���Է���ˮ�ⷴӦ����F�Ľṹ��ʽΪ________�� ________��
(5)��ӦC��E��X�Ļ�ѧ����ʽΪ______________��
���𰸡� �Ȼ� CH3CH2OH ������Ӧ/ȡ����Ӧ A��E HCOOCH2CH3 CH3COOCH3 CH3CH2COOH + CH3CH2OH ![]() CH3CH2COOCH2CH3 + H2O
CH3CH2COOCH2CH3 + H2O
��������D�IJ���������������һ�����ҵ�ʯ�ͻ���ˮƽ��DΪ��ϩ����ϩ��ˮ�����ӳɱ�Ϊ�Ҵ���E��������X�Ǻ���5��̼�ı���һԪ���������л���CΪ����3��C�ı���һԪ���ᣬ��������ԣ��л���AΪ1-�������л���BΪ��ȩ��
(1)����������CΪ���ᣬ�����ŵ������Ȼ���EΪ�Ҵ����ṹ��ʽCH3CH2OH����ȷ�𰸣��Ȼ���CH3CH2OH��
(2)�Ҵ��ͱ��ᷢ��������Ӧ��������ˮ����ȷ�𰸣�������Ӧ/ȡ����Ӧ��
(3)1-��������ȩ�����ᡢ��ϩ�������������Ҵ����������У���Ϊͬϵ�����1-�������Ҵ�����ȷ�𰸣�A��E��
(4) FΪ�����ͬ���칹�壬�ܹ�����ˮ�ⷴӦ���ṹ�к���������F�Ľṹ��ʽΪ�ֱ�Ϊ��HCOOCH2CH3 ��CH3COOCH3����ȷ�𰸣�HCOOCH2CH3 ��CH3COOCH3��
(5) ������Ҵ���Ũ������������·���������Ӧ����ѧ����ʽΪCH3CH2COOH + CH3CH2OH ![]() CH3CH2COOCH2CH3 + H2O����ȷ�𰸣�CH3CH2COOH + CH3CH2OH
CH3CH2COOCH2CH3 + H2O����ȷ�𰸣�CH3CH2COOH + CH3CH2OH ![]() CH3CH2COOCH2CH3 + H2O��
CH3CH2COOCH2CH3 + H2O��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�����й��ڱ�ϩ��![]() ���Ľṹ�����ʵ���������ȷ���ǣ� ��
���Ľṹ�����ʵ���������ȷ���ǣ� ��
A. �����������ֻ�ѧ������ͬ����
B. �ڹ�������Cl2��Ӧ���ɵ�һ��ȡ��������Ҫ�ǣ� ![]()
C. ��һ����������HCl��Ӧ�ļӳɲ������������
D. �����ʽΪC6H12���л��ﻥΪͬϵ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����һ��NaCl��Na2CO3��10H2O��NaHCO3�Ļ���ijͬѧ�������ʵ�飬ͨ��������Ӧǰ��C��Dװ�������ı仯���ⶨ�û�����и���ֵ�����������
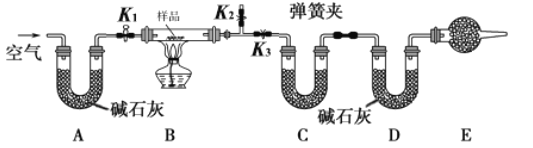
��1���뽫ʵ�鲽�貹������
�ٰ�ͼ���г�����δ��������װ��ʵ��װ�ú����Ƚ��еIJ����� _____________________��
�ڳ�ȡ��Ʒ�����������Ӳ�ʲ������У�������C��Dװ�õ�������
�۴���K1��K2���ر�K3������������������ӣ���Ŀ����____________________��
�ܹرջ���K1��K2����K3����ȼ�ƾ��Ƽ��������ٲ������塣
�ݴ���K1������������������ӣ�Ȼ��ж��װ�ã��ٴγ���C��Dװ�õ�������
��2�����ڸ�ʵ�鷽������ش��������⣺
�������ȷ�Ӧ�����������NaCl�ⶨ�����Ӱ����___________����ƫ��������ƫ����������Ӱ������
��E���������ʢ�ŵ�ҩƷ��_______________����������_____________________________�����ʵ����û�и�װ�ã���ᵼ�²������NaHCO3����������______________����ƫ��������ƫ����������Ӱ������
������Ʒ����Ϊwg����Ӧ��C��Dװ�����ӵ������ֱ�Ϊm1g��m2g���ɴ˿�֪�������Na2CO3��10H2O����������Ϊ______���ú�w��m1��m2�Ĵ���ʽ��ʾ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����֪As2O3��Zn���Է������·�Ӧ��As2O3+6Zn+6H2SO4![]() 2AsH3��+6ZnSO4+3H2O��
2AsH3��+6ZnSO4+3H2O��
(1)����˫���ŷ��������ת�Ƶķ������Ŀ____________________________________��
(2)As2O3��������Ӧ����ʾ������������____________(�����)��
A�������� B����ԭ�� C������ D������
(3)�÷�Ӧ������������__________����ԭ������________��
(4)������0.2 mol AsH3����ת�Ƶĵ�����Ϊ___________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��(1)2017���п�Ժij�о��Ŷ�ͨ�����һ������Na-Fe3O4/HZSM-5��ܸ��ϴ������ɹ�ʵ����CO2ֱ�Ӽ�����ȡ����ֵ���ͣ����о��ɹ�������Ϊ��CO2��ת�������ͻ���Խ�չ����
��֪��H2(g)+1/2O2(g)=H2O(l) ��H1 = ��aKJ/mol
C8H18(1)+25/2O2(g)=8CO2(g)+9H2O(1) ��H2= ��bKJ/mol
��д��25����101kPa�����£�CO2��H2��Ӧ��������(��C8H18��ʾ)���Ȼ�ѧ����ʽ_________________________________��
(2)����CO2��H2Ϊԭ�ϣ��ں��ʵĴ���(��Cu/ZnO����)�����£�Ҳ�ɺϳ�CH3OH���漰�ķ�Ӧ�У�
�ף�CO2(g)+3H2(g) ![]() CH3OH(g)+H2O(g) ��H= �� 53.7kJ��mol-1 ƽ�ⳣ��K1
CH3OH(g)+H2O(g) ��H= �� 53.7kJ��mol-1 ƽ�ⳣ��K1
�ң�CO2(g)+H2(g) ![]() CO(g)+H2O(g) ��H= + 41.2kJ��mol-1 ƽ�ⳣ��K2
CO(g)+H2O(g) ��H= + 41.2kJ��mol-1 ƽ�ⳣ��K2
��CO(g)+2H2(g) ![]() CH3OH(g)��ƽ�ⳣ��K=______(�ú�K1��K2�ı���ʽ��ʾ)���÷�Ӧ��H_____0(��������������С����)��
CH3OH(g)��ƽ�ⳣ��K=______(�ú�K1��K2�ı���ʽ��ʾ)���÷�Ӧ��H_____0(��������������С����)��
�����CO2ת��ΪCH3OHƽ��ת���ʵĴ�ʩ��___________(��д����)��
�۴����ͷ�Ӧ��ϵ�Ĺ�ϵ��������Կ�Ĺ�ϵһ�������и߶ȵ�ѡ���ԡ���������ʵ�飬����CO2��H2��ʼͶ�ϱȾ�Ϊ1��2.2��������ͬ��Ӧʱ��(t1min)��
�¶�(K) | ���� | CO2ת����(%) | �״�ѡ����(%) | �ۺ�ѡ�� |
543 | Cu/ZnO���װ����� | 12.3 | 42.3 | A |
543 | Cu/ZnO����Ƭ���� | 11.9 | 72.7 | B |
553 | Cu/ZnO���װ����� | 15.3 | 39.1 | C |
553 | Cu/ZnO����Ƭ���� | 12.0 | 70.6 | D |
�ɱ����е����ݿ�֪����ͬ�¶��²�ͬ�Ĵ�����CO2��ת��ΪCH3OH��ѡ����������Ӱ�죬�����ϱ��������ݽ�Ϸ�Ӧԭ������������ѡ��Ϊ___________(����ĸ����)��
(3)��CO��H2Ϊԭ�Ϻϳ��״��ķ�ӦΪ��CO(g)+2H2(g)![]() CH3OH(g)���������Ϊ2L�����������ܱ����������������У��ֱ���1molCO��2molH2�����������ķ�Ӧ�¶ȷֱ�ΪT1��T2��T3�Һ㶨���䡣��ͼΪ���������еķ�Ӧ�����е�5minʱH2���������ʾ��ͼ��������һ��������Ӧһ���ﵽƽ��״̬��
CH3OH(g)���������Ϊ2L�����������ܱ����������������У��ֱ���1molCO��2molH2�����������ķ�Ӧ�¶ȷֱ�ΪT1��T2��T3�Һ㶨���䡣��ͼΪ���������еķ�Ӧ�����е�5minʱH2���������ʾ��ͼ��������һ��������Ӧһ���ﵽƽ��״̬��
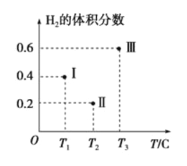
��0��5minʱ��������������CH3OH��ʾ�Ļ�ѧ��Ӧ����Ϊ_________________��
������������һ���ﵽƽ��״̬��������________(��д��������)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��������NH4I�����ܱ������У���һ���¶��·������з�Ӧ����NH4I(s)![]() NH3(g)+HI(g) ��2HI(g)
NH3(g)+HI(g) ��2HI(g)![]() H2(g)+I2(g) �ﵽƽ��ʱ��c(H2)��0.5 mol��L-1��c(HI)��4 mol��L-1��������¶��·�Ӧ����ƽ�ⳣ��Ϊ
H2(g)+I2(g) �ﵽƽ��ʱ��c(H2)��0.5 mol��L-1��c(HI)��4 mol��L-1��������¶��·�Ӧ����ƽ�ⳣ��Ϊ
A. 9 B. 16 C. 20 D. 25
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��ʵ����������װ����ȡ������������������ʵ�顣�ش��������⣺

��1��A��ʢ��Ũ���ᣬB��ʢ��MnO2��д����Ӧ�Ļ�ѧ����ʽ_____________��
��2��D�з���ŨH2SO4����Ŀ����_____________________________��
��3��E��Ϊ��ɫ�ɲ�����F��Ϊ��ɫʪ�������ɹ۲쵽��������___________���Ա�E��F������IJ���ɵó��Ľ��ۼ�������________________________________��
��4��G����������____________________________________��
��5������H��β������װ��ͼ��ע���Լ�____________��
��6����ͥ�г�������Һ����Ҫ�ɷ�NaClO�������飨��Ҫ�ɷ����ᣩ���������ijƷ������Һ��װ��˵������ͼ��

�١�������ͬʱʹ�á�������ж���������д����Ӧ�����ӷ���ʽ__________��
���衰�ܱձ��桱��ԭ��____________________________________________��
��7��������һ����Ϊ����ˮ�衱�IJ�ƷҲ�ܶ�����ˮ���п��ٵ�ɱ��������ҩ��ͨ�����������㡣�������Ⱦ�Cl2Na(NCO)3����ˮ��Ӧ�����ɴ�������ɱ���������ã������Ӻ��ڲ���������ƣ�Na2SO3���ܳ����ɽ�ˮ�е����ȣ�������ȣ���ȥ���������ƽ�ˮ�ж���������ȥ�����ӷ�Ӧ����ʽΪ______________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����ͼ����A��ͨ�봿����Cl2����B��ʱ��C���ĺ�ɫ�����Ͽ��������������ر�B����C���첼����ɫ����Dƿ��װ���ǣ�������

A. ŨH2SO4 B. NaOH��Һ C. Na2SO3��Һ D. ����NaCl��Һ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��ij��ѧ��ȤС��������ͼװ�ý�������ˮ������Ӧ��ʵ�飬�������������ʣ���ش��������⣺

��1��Aװ�õ�������____________����ƿ�ײ������Ƭ��������_____________��
��2��װ��B�з�����Ӧ�Ļ�ѧ����ʽ��___________���÷�Ӧ����������__________������������_____________��
��3��D��������__________��
��4��E�е�ʵ��������_______________��
��5��A��B����װ����Ӧ�ȵ�ȼ____���ľƾ�(��)�ƣ���ȼE���ƾ���֮ǰӦ���еIJ�����_______��
��6����ͬѧ�Է�Ӧ��Ӳ���Թ��й������ʵ������������¼��裺
����1��ֻ��Fe��
����2��ֻ��________��
����3������FeҲ��Fe3O4��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com