
����Ŀ������������ա�
��.�����£�ijˮ��ҺM�д��ڵ�������Na����A2����HA����H����OH�������ڵķ�����H2O��H2A����������ش��������⣺
(1)д����H2A�ĵ��뷽��ʽ��
________________________________________________________________________��
(2)ʵ����NaHA��Һ��pH>7�������NaHA��Һ�Լ��Ե�ԭ��
________________________________________________________________________��
��д��NaHA��Һ������Ũ���ɴ�С��˳��
________________________________________________________________________��
��.(1)��ͼ�ױ�ʾ����ͬŨ�ȵ�NaOH��Һ�ֱ�ζ�Ũ����ͬ������һԪ�ᣬ��ͼ��ȷ��������ǿ����________(�����١����ڡ������ۡ�)����ͼ�ұ�ʾ����ͬŨ�ȵ�AgNO3����Һ�ֱ�ζ�Ũ����ͬ�ĺ�Cl����Br����I���Ļ����Һ����ͼ��ȷ�����ȳ�����������________��

(2)25 ��ʱ��Ksp(AgCl)��1.8��10��10����1 L 0.1 mol��L��1 NaCl��Һ�м���1 L 0.2 mol��L��1AgNO3��Һ����ַ�Ӧ����Һ��c(Cl��)��________ mol��L��1(�����Ϻ���Һ������仯���Բ���)��
���𰸡�H2A![]() H����HA����HA��
H����HA����HA��![]() H����A2�� HA����ˮ��̶ȴ��������̶� c(Na+)>c(HA-)>c(OH-)>c(H+)>c(A-) �� I�� 3.6��10��9
H����A2�� HA����ˮ��̶ȴ��������̶� c(Na+)>c(HA-)>c(OH-)>c(H+)>c(A-) �� I�� 3.6��10��9
��������
��.��1��������Һ�д��������жϣ���Ԫ����ֲ����룻
��2��0.1mol/LNaHA��Һ��HA-�ĵ���̶ȴ���ˮ��̶ȣ���Һ�����ԣ�H2A��Һ�е�һ����������������������˵ڶ������룬��NaHA��HA-�ĵ���̶ȴ���H2A��HA-�ĵ���̶ȣ�
��.(1) Ũ����ͬ��3��һԪ�ᣬ�۵�pH��С��˵��������ǿ��Ksp(AgI)��С����I�����ȳ�����
(2) NaCl��Һ�м���AgNO3��Һ������ӦNaCl+AgNO3=AgCl��+NaNO3������Ksp(AgCl)��c(Ag+) ��c(Cl��)���㡣
��.��1����Һ�д���H2A��A2-��HA-����H2A��Һ�д��ڵ���ƽ�⣬˵��H2AΪ���ᣬ����뷽��ʽΪ��H2A![]() H����HA����HA��
H����HA����HA��![]() H����A2����
H����A2����
(2) NaHA��Һ�д���HA���ĵ���ƽ�⣨HA��![]() H����A2������ˮ��ƽ��(HA��+H2O
H����A2������ˮ��ƽ��(HA��+H2O![]() OH-��H2A)��ʵ����NaHA��Һ��pH>7��˵��HA����ˮ��̶ȴ��������̶ȣ�
OH-��H2A)��ʵ����NaHA��Һ��pH>7��˵��HA����ˮ��̶ȴ��������̶ȣ�
NaHA��Һ��HA-�ĵ���̶ȴ���ˮ��̶ȣ���Һ�����ԣ�����Һ������Ũ�ȴ�С��ϵΪ��c(Na+)>c(HA-)>c(OH-)>c(H+)>c(A-)��
��.(1) Ũ����ͬ��3��һԪ�ᣬ�۵�pH��С��˵��������ǿ��������ȫ��I��Ũ����С��˵��Ksp(AgI)��С����I�����ȳ�����
(2) NaCl��Һ�м���AgNO3��Һ������ӦNaCl+AgNO3=AgCl��+NaNO3��AgNO3��ʣ�࣬���Һ��c(AgCl)��![]() (0.2mol/L-0.1mol/L)=0.05mol/L��AgCl(s)��ˮ�д����ܽ�ƽ�⣺AgCl(s)
(0.2mol/L-0.1mol/L)=0.05mol/L��AgCl(s)��ˮ�д����ܽ�ƽ�⣺AgCl(s) ![]() Ag+(aq)+Cl-(aq)������Ksp(AgCl)��c(Ag+) ��c(Cl��)����ôc(Cl��)��
Ag+(aq)+Cl-(aq)������Ksp(AgCl)��c(Ag+) ��c(Cl��)����ôc(Cl��)��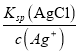 =
=![]() =3.6��10��9mol��L��1��
=3.6��10��9mol��L��1��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��ʵ������ȡ��ϩ�������¶ȹ��߶�ʹ�Ҵ���ŨH2SO4��Ӧ���������Ķ������������������ʵ����ȷ�������������������ϩ�Ͷ���������װ������ͼ��ʾ���Իش��������⡣

(1)ͼ�Т١��ڡ��ۡ���װ�ÿ�ʢ�ŵ��Լ���
��________����________����________����________��(�������й��Լ����������ո���)
A��Ʒ����Һ B��NaOH��Һ
C��ŨH2SO4 D������KMnO4��Һ
(2)��˵����������������ڵ�������_____________��
(3)ʹ��װ�âڵ�Ŀ����___________��ʹ��װ�â۵�Ŀ����____________��
(4)��˵��������ϩ��������____________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ������25��ʱ0.1 mol/L�İ�ˮ����ش��������⣺
��1������ˮ�м���ϡ���ᣬʹ��ǡ����ȫ�кͣ�д����Ӧ�����ӷ���ʽ��________________________��������Һ��pH________7(����>������<����������)�������ӷ���ʽ��ʾ��ԭ��__________________������������Һ�������غ��ϵ�������е�ʽ���������� c(SO42��)=________________________��
��2������ˮ�м���ϡ��������Һ��pH��7����ʱc(NH4+)��a mol/L����c(SO42��)��________��
��3������ˮ�м���pH��1�����ᣬ�Ұ�ˮ������������Ϊ1��1����������Һ�и����ӵ����ʵ���Ũ���ɴ�С��˳����______________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�������£�pH��Ϊ2������һԪ��HA��HB��1mL���ֱ��ˮϡ�ͣ�pH����Һ����ı仯��������ͼ��ʾ������˵����ȷ����( )

A��HA�����Ա�HB��������
B��a����Һ�ĵ����Ա�c����Һ�ĵ�������
C��������Һ����ϡ�ͣ������ǵ�c(H+)���
D����a��b������Һͬʱ�����¶ȣ���![]() ����
����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��X��Һֻ��![]() ��
��![]() ��
��![]() ��
��![]() ��
��![]() ��
��![]() ��
��![]() ��
��![]() �еļ��֣��ֽ�������ʵ�飺��������ʵ�飬��X��Һ���ж���ȷ����
�еļ��֣��ֽ�������ʵ�飺��������ʵ�飬��X��Һ���ж���ȷ����![]()
![]()

A.һ������![]() ��
��![]() ��
��![]() ��
��![]() ��һ������
��һ������![]() ��
��![]() ��
��![]() ��
��![]()
B.һ������![]() ��
��![]() ��
��![]() �����ܺ���
�����ܺ���![]()
C.һ������![]() ��
��![]() ��
��![]() �����ܺ���
�����ܺ���![]() ��
��![]()
D.һ������![]() ��
��![]() ��
��![]() ��
��![]()
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��Ϊ�ⶨ��ʽ̼����[COx(OH)y(CO3)z]�Ļ�ѧ��ɣ��о���ѧϰС���ͬѧ�������ͼ��ʾ��װ�ý���ʵ��̽������֪����ʽ̼��������ʱ�ɷֽ��������������
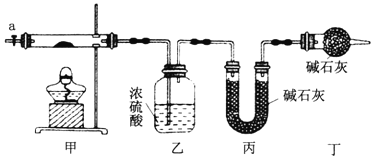
��1������ͼ��ʾװ����װ����������___________����ȡ3.65g��Ʒ����Ӳ�ʲ������ڣ����ȣ�����װ����_______(��ʵ������)��ֹͣ���ȣ�����a������ͨ����������ӣ�ͨ�������Ŀ����__________��
��2��ijͬѧ��Ϊ����ʵ���д���һ��ȱ�ݣ���ȱ����_________��
��3��ͨ����ȷʵ�����ҡ���װ�����طֱ�Ϊ0.36g��0.88g����ü�ʽ̼���ܵĻ�ѧʽΪ__________��
��4��CoCl2��6H2O���������ˮ������Ӽ����Ժ��ܷ���(������Fe��Al������)��ȡCOCl2��6H2O��һ�ֹ������£�
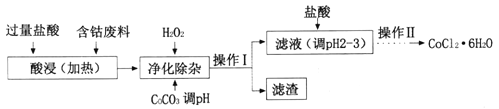
��֪��25��ʱ
������ | Fe(OH)3 | Fe(OH)2 | CO(OH)2 | Al(OH)3 |
��ʼ������pH�� | 2.3 | 7.5 | 7.6 | 3.4 |
��ȫ������pH�� | 4.1 | 9.7 | 9.2 | 5.2 |
�پ�������ʱ������H2O2������Ӧ�����ӷ���ʽΪ______________��
�ڼ���CoCO3��pHΪ5.2��7.6�������I��õ������ɷ�Ϊ_________��
�ۼ��������pHΪ2~3��Ŀ��Ϊ___________��
�ܲ��������Ϊ����Ũ������ȴ�ᾧ�����ˡ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��ʵ�����Ʊ���ˮ����ͭ[Cu(HCOO)24H2O]����ʵ�鲽�����¡�
(1)��ʽ̼��ͭ���Ʊ���
a.����i�ǽ�һ����������̼�����ƹ���һ��ŵ��в�����ĥ����Ŀ����______��
b.����ii���ڽ����½���������ֶ�λ���������ˮ�У���Ӧ�¶ȿ�����70�桪80�棬�������_____(дʵ������)��˵���¶ȹ��ߡ�
c.��صĻ�ѧ����ʽ______��
(2)��ˮ����ͭ������Ʊ�������ʽ̼��ͭ��������ձ��У�����һ�����ȵ�����ˮ������μ����������ʽ̼��ͭǡ��ȫ���ܽ⣬���ȹ��˳�ȥ�������������ʣ�Ȼ��������ȴ���ˣ�����������ˮ�Ҵ�ϴ�Ӿ���2��3�����ɣ��õ���Ʒ��
a.��صĻ�ѧ����ʽ______��
b.���ȹ����У�������ȵ�ԭ����______��
c.���Ҵ�ϴ�Ӿ����Ŀ��______��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��25��ʱ��10 mL 0.1mol��L��1Na2CO3��Һ����μ���0.1 mol��L��1HCl��Һ20 mL����Һ�в��ֺ�̼�������ʵ�������ҺpH�ı仯��ͼ��ʾ������˵������ȷ����

A��HCl��Һ�μ�һ��ʱ����ҺpH>7
B������Һ��pH=8ʱ����Һ��NaHCO3�����ʵ���Ũ��Ϊ0.1mol��L��1
C��0.1 mol��L��1Na2CO3��Һ��c(Na+)+c(H+)=c(OH��)+2c(CO32��)+c(HCO3��)
D����M����c(Na+)>c(CO32��)=c(HCO3��)>c(OH��)>c(H+)
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com