
����25 ��ʱ0.1 mol��L��1�İ�ˮ����ش��������⣺
��1������ˮ�м�����������粒��壬һˮ�ϰ��ĵ���ƽ��________(����������ҡ�����)�ƶ�����ʱ��Һ��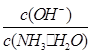 ________(���������С�����䡱)��
________(���������С�����䡱)��
��2������ˮ�м����Ũ��ϡ���ᣬʹ��ǡ���кͣ�д����Ӧ�����ӷ���ʽ��_________________��������Һ��pH________7(���������<������)��
��3������ˮ�м���ϡ��������Һ��pH��7����ʱ[NH4+]��a mol��L��1����c(SO42-)��________��
��4������ˮ�м���pH��1�����ᣬ�Ұ�ˮ������������Ϊ1��1����������Һ�и����ӵ����ʵ���Ũ���ɴ�С��˳����________________________________________��
��1������ ��С ��2��NH3��H2O��CH3COOH��CH3COO����NH4����H2O �� ��3��

��4��c(NH4��)��c(SO42��)��c(H��)��c(OH��)
���������������1����ˮ�м�����������粒��壬����淋��������笠����ӻ�����һˮ�ϰ��ĵ��룬ƽ�������ƶ�����ʱ��Һ�� ��С ��2��NH3��H2O��CH3COOH��CH3COO����NH4����H2O ����Ϊǡ���кͣ���Һ�����ԣ�������Һ��pH��7 ��3�����ݵ���غ��У�c(NH4��)��c(H��)��2c(SO42��)��c(OH��)������Ϊ��Һ��pH��7������У�c(SO42��)��
��С ��2��NH3��H2O��CH3COOH��CH3COO����NH4����H2O ����Ϊǡ���кͣ���Һ�����ԣ�������Һ��pH��7 ��3�����ݵ���غ��У�c(NH4��)��c(H��)��2c(SO42��)��c(OH��)������Ϊ��Һ��pH��7������У�c(SO42��)�� c(NH4��)��
c(NH4��)�� ����4������ˮ�м���pH��1�����ᣬ�Ұ�ˮ������������Ϊ1��1����õ���������淋���Һ����Ϊ�������ӵ�ˮ�⣬ʹ����Һ�����ԣ���������¹�ϵ��c(NH4��)��c(SO42��)��c(H��)��c(OH��)
����4������ˮ�м���pH��1�����ᣬ�Ұ�ˮ������������Ϊ1��1����õ���������淋���Һ����Ϊ�������ӵ�ˮ�⣬ʹ����Һ�����ԣ���������¹�ϵ��c(NH4��)��c(SO42��)��c(H��)��c(OH��)
���㣺����������ʵķ�Ӧ��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
������ǿ�ᣬ��ѧ�ν�������ˮ��Һ�п�����ȫ���롣����ʵ�ǣ�������ˮ�еĵ�һ����������ȫ�ģ��ڶ������벢����ȫ����������ΪH2SO4=H����HSO4-��HSO4- H����SO42-��
H����SO42-��
��ش������й����⣺
��1��Na2SO4��Һ��________(������ԡ��������ԡ��������ԡ�)����������__________________________________________��(�����ӷ���ʽ��ʾ)��
��2��H2SO4��Һ��BaCl2��Һ��Ӧ�����ӷ���ʽΪ_________________________��
��3����0.10 mol��L��1��Na2SO4��Һ�У���������Ũ�ȵĹ�ϵ��ȷ����________(��д���)��
| A��c(Na��)��c(SO42-)��c(HSO4-)��c(H2SO4) |
| B��c(OH��)��c(HSO4-)��c(H��) |
| C��c(Na��)��c(H��)��c(OH��)��c(HSO4-)��2c(SO42-) |
| D��c(Na��)��2c(SO42-)��2c(HSO4-) |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
Ϊ��֤��������������ʣ��ס��ҡ����������졢���������˷ֱ�ѡ�������Լ�����ʵ�飺
0.10 mol/L������Һ��0.10 mol/L��������Һ��pH=3�����ᡢpH=3�Ĵ��ᡢ�����ƾ��塢�Ȼ��ƾ��塢���ȡ�pH��ֽ������ˮ��
(1)����pH��ֽ���0.10 mol/L�Ĵ�����ҺpH=4�����϶�������������ʣ�����Ϊ��һ������ȷ�� ��(���ȷ������ȷ��)
(2)��ȡ��10 mL 0.10 mol/L�Ĵ�����Һ����pH��ֽ�����pH=a��Ȼ��������ˮϡ�͵�1 000 mL������pH��ֽ�ⶨ��pH=b��Ҫȷ��������������ʣ���a��bӦ������Ĺ�ϵ�� (�õ�ʽ��ʽ��ʾ)��
(3)��ȡ��10 mL 0.10 mol/L������Һ�����������Һ���Ժ�ɫ���ټ�������ƾ��壬��ɫ���ɫ������Ϊ��һ�����ܷ�֤��������������ʣ� ��(��ܡ����ܡ�)
(4)����pH��ֽ���ⶨ0.10 mol/L��������Һ��pH������0.10 mol/L��������Һ��pHΪ9�����϶�������������ʣ�����Ϊ��һ������ȷ�� ��(���ȷ������ȷ��)
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
X��Y��Z��W�ֱ���HNO3��NH4NO3��NaOH��NaNO2����ǿ������е�һ�֡��±��dz�����Ũ�Ⱦ�Ϊ0.0 1 mol��L-1��X��Y��Z��W��Һ��pH��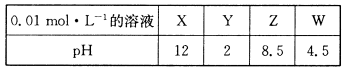
��1��X��W�Ļ�ѧʽ�ֱ�Ϊ_______��________��
��2��W�ĵ��뷽��ʽΪ______________________________��
��3��25��ʱ��Z��Һ��pH��7��ԭ����______________________________________(�����ӷ���ʽ��ʾ)��
��4����X��Y��Z��1mol��L-1ͬʱ����ˮ���Ƶû����Һ��������Һ�и����ӵ�Ũ���ɴ�С��˳��Ϊ _______________________________________________________ ��
��5��Z��Һ��W��Һ��ϼ��ȣ��ɲ���һ����ɫ��ζ�ĵ������壬�÷�Ӧ�Ļ�ѧ����ʽΪ__________________________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ijѧ��������֪���ʵ���Ũ�ȵĴ������ⶨδ֪���ʵ���Ũ�ȵ�����������Һʱ��ѡ���ʵ���ָʾ��������д���пհף�
��1���ñ�����ζ����������������Һʱ��������ѡ����ѡ����ǡ����һ��________��
| | ��ƿ����Һ | �ζ�������Һ | ѡ��ָʾ�� | ѡ�õζ��� |
| A | �� | �� | ʯ�� | ���ң� |
| B | �� | �� | ���� | ���ף� |
| C | �� | �� | ��̪ | ���ף� |
| D | �� | �� | ʯ�� | ���ң� |
| | ������������ | 0.100mol/L�������� | |
| �ζ����� | ��Һ�������ml�� | �ζ�ǰ�Ŀ̶ȣ�ml�� | �ζ���Ŀ̶ȣ�ml�� |
| ��һ�� | 25.00 | 0.00 | 24.98 |
| �ڶ��� | 25.00 | 1.56 | 27.86 |
| ������ | 25.00 | 0.22 | 25.24 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
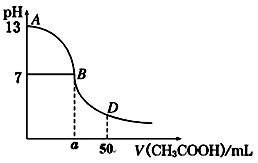
��1����֪�����£�A�����ҺpH=a��B�����ҺpH=b���ش��������⣺
����AΪ���ᣬBΪ�������ƣ���a=4��b=10�����ߵ������ϣ�����ǰc(CH3COOH) c(NaOH )���������������ͬ������Ϻ���Һ��pH 7��
����A�Ļ�ѧʽΪHR��B�Ļ�ѧʽΪMOH����a+b=14�����ߵ������Ϻ���Һ�Լ��ԣ�������Һ�бض���һ�������ܷ���ˮ�⣬��ˮ�ⷴӦ�����ӷ���ʽΪ ����2��������25��ʱ���������pH��Ba(OH)2��NaOH��NH3��H2O������Һ�������Ƿֱ���V1 L��V2 L��V3 L��Ũ�ȵ������ϣ���ǡ���кͣ���V1��V2��V3�Ĵ�С��ϵ�� ��
������25�棬����������ʵ���Ũ�ȵ�Ba(OH)2��NaOH��NH3��H2O������Һ�������Ƿֱ���V1 L��V2 L��V3 L��Ũ�ȵ������ϣ���Ϻ���Һ�������ԣ���V1��V2��V3�Ĵ�С��ϵ�� ���������£�������ͬŨ�ȵ�NaCl��NH4Cl��Һ��1L������Һ����������N(NaCl) N(NH4Cl)���������������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��1����������pH=12��NaCN��Һ�У���ˮ�����c(OH��)Ϊ mol?L��1��
��2��Ũ��Ϊ0.1mol?L��1�����и����ʵ���Һ�У�c(NH4+)�ɴ�С��˳����___������ţ���
��NH4Cl ��NH4HSO4 ��NH3?H2O ��CH3COONH4
��3��ij��Ԫ�ᣨ��ѧʽ��H2A��ʾ����ˮ�еĵ��뷽��ʽ�ǣ�
H2A=H+ +HA����HA�� H+ +A2����
H+ +A2����
����Na2A��Һ��____�ԣ�NaHA��Һ�� �ԣ�����ԡ��������ԡ����ԡ�����
������0.1mo1?L��1Na2A����Һ�����и�������Ũ���ɴ�С��˳���ǣ� ������ţ���
| A��c(Na+)>c(A2��)>c(OH��)>c(HA��)>c(H+) |
| B��c(Na+)> c(OH��)>c(HA��)> >c(A2��) > c(H+) |
| C��c(Na+)> c(H+)> c(A2��)> c(OH��)>c(HA��) |
| D��c(A2��)>c(Na+)> c(OH��) > c(H+)>c(HA��) |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
����pH��2�Ĵ�����Һ��pH��2�������ң���������в���������⣺
(1)ȡ10 mL�ļ���Һ������������ˮ������ĵ���ƽ��________(����������ҡ�����)�ƶ�����ȡ10 mL�ļ���Һ������������ˮ�����ƹ���(����������ǰ����Һ������ֲ���)���������������Һ��[H��]/[CH3COOH]�ı�ֵ��________(���������С������ȷ����)��
(2)��ͬ�����£�ȡ������ļס�������Һ����ϡ��100����ϡ�ͺ����Һ����pH��С��ϵΪ��pH(��)________pH(��)��(����ڡ�����С�ڡ����ڡ�����ͬ)
(3)��ȡ25 mL�ļס�������Һ���ֱ��õ�Ũ�ȵ�NaOHϡ��Һ�к���pH��7�������ĵ�NaOH��Һ�������С��ϵΪ��V(��)________V(��)��
(4)ȡ25 mL�ļ���Һ����������pH��12��NaOH��Һ����Ӧ����Һ��[Na��]��[CH3COO��]�Ĵ�С��ϵΪ��[Na��]________[CH3COO��]��
(5)ȡ25 mL������Һ����������pH��12�İ�ˮ����Ӧ����Һ�е�����Ϊ_________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ijѧϰС����̽��CaSO4����ת��ΪCaCO3�������Ӷ������ȥ�Ŀ����ԣ�����������ϣ�(�������ݺ�ʵ���ָ��25���²ⶨ)
| ���ܵ���� | CaCO3 | CaSO4 |
| Ksp(mol2��L��2) | 3��10��9 | 9��10��6 |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com