
����һ�ֻ��ý����������ܶ�С���۵�ߡ�������ǿ����еǿ�ȸߵ����ܡ���ҵ�ϳ�������ֽ����ѿ�ʯ���Ʊ��������ѣ�����ұ���ѣ���Ҫ�����������Ӧ��
��FeTiO3��2H2SO4=TiOSO4��FeSO4��2H2O
��TiOSO4��2H2O=H2TiO3����H2SO4
��H2TiO3 TiO2��H2O
TiO2��H2O
��TiO2��2C��2Cl2 TiCl4����2CO��
TiCl4����2CO��
��TiCl4��2Mg 2MgCl2��Ti
2MgCl2��Ti
(1)������������Ӧ�����������������________��
A����Ӧ���Ƿ�������ԭ��Ӧ
B����Ӧ��������������
C����Ӧ���е�TiO2��������
D����Ӧ�ݱ����˽���þ�Ƚ����ѵĻ�ԭ��ǿ
(2)�Ѿ��к�ǿ����ʴ�ԣ����¶���ԭ��ķ�����ȷ����________��
A��������𡢲�һ���IJ����ý���
B�������ѵı������γ����ܵ�����Ĥ
C��������������ȸ�ʴ������Ӧ
D�����кܸߵ�ǿ��
 ���ѵ����Ԫ��ĩ���100��ϵ�д�
���ѵ����Ԫ��ĩ���100��ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
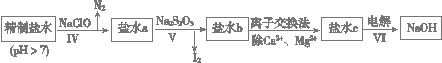
(14��)���ռ�������ˮ�����ξ��ơ���һ�ξ�����Ҫ���ó�������ȥ����ˮ��Ca2����Mg2����SO42�������ӣ��������£�
��. �����ˮ�м������BaCl2��Һ�����ˣ�
��. ��������Һ�м������Na2CO3��Һ�����ˣ�
��. ��Һ���������pH����õ�һ�ξ�����ˮ��
��1�����̢��ȥ��������______��
��2�����̢����ɵIJ��ֳ��������ܽ��(20 ��/g)���±��������ݱ������ݽ����������⣺
| CaSO4 | Mg2(OH)2CO3 | CaCO3 | BaSO4 | BaCO3 |
| 2.6��10��2 | 2.5��10��4 | 7.8��10��4 | 2.4��10��4 | 1.7��10��3 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
���������Ǵ�����Ⱦ��֮һ��������������ķ����ж��֡�
��1�����ü������ԭ���������֪��
��CH4 (g)��4NO2(g)��4NO(g)��CO2(g)��2H2O(g�� ��H ����574 kJ/mol
��CH4(g)��4NO(g���� 2N2(g)��CO2(g)��2H2O(g�� ��H ����1160 kJ/mol
��CH4 ��NO2 ��ԭΪN2 ���Ȼ�ѧ����ʽΪ�� ��
��2������NH3����ԭ�������SCR����)���ü�����ĿǰӦ����㷺���������������ѳ������� ��Ӧ�Ļ�ѧ����ʽΪ: Ϊ��ߵ��������ת���ʿɲ�ȡ�Ĵ�ʩ�� ��д��1�����ɣ���
Ϊ��ߵ��������ת���ʿɲ�ȡ�Ĵ�ʩ�� ��д��1�����ɣ���
��3������ClO2�������������ת����������: NO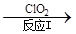 NO2
NO2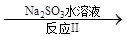 N2����֪��Ӧ��Ļ�ѧ����ʽΪ2NO+ ClO2 + H2O ��NO2 + HNO3 + HCl����Ӧ��Ļ�ѧ����ʽ�� ��������11.2 L N2����״������������ClO2 g ��
N2����֪��Ӧ��Ļ�ѧ����ʽΪ2NO+ ClO2 + H2O ��NO2 + HNO3 + HCl����Ӧ��Ļ�ѧ����ʽ�� ��������11.2 L N2����״������������ClO2 g ��
��4���û���̿��ԭ��������������йط�ӦΪ��C��s��+2NO��g�� N2 ��g��+CO2 ��g����H��ij�о�С����ij�ܱ���������һ�����Ļ���̿��NO�����£�T1�棩�����·�Ӧ����Ӧ���е���ͬʱ���ø����ʵ�Ũ�����£�
N2 ��g��+CO2 ��g����H��ij�о�С����ij�ܱ���������һ�����Ļ���̿��NO�����£�T1�棩�����·�Ӧ����Ӧ���е���ͬʱ���ø����ʵ�Ũ�����£�
| Ũ��/mol?L-1/ ʱ��/min | NO | N2 | CO2 |
| 0 | 0.100 | 0 | 0 |
| 10 | 0.058 | 0.021 | 0.021 |
| 20 | 0.040 | 0.030 | 0.030 |
| 30 | 0.040 | 0.030 | 0.030 |
| 40 | 0.032 | 0.034 | 0.017 |
| 50 | 0.032 | 0.034 | 0.017 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
(1)��ⱥ��ʳ��ˮ֮ǰʳ��ˮ��Ҫ���ƣ�Ŀ���dz�ȥ�����е�Ca2����Mg2����SO42�����������ӣ�ʹ�õ��Լ��Ͳ�����a.Na2CO3��Һ��b.Ba(OH)2��Һ��c.ϡ���ᣬd.���ˣ�������ļ���˳��Ϊ________(����ĸ����)��
(2)��ⱥ��ʳ��ˮʱ���ӽ���Ĥ��������_____________________________
(3)��ⱥ��ʳ��ˮʱ��������ݻ�Ϊ10 L�����ӽ���Ĥ�����У�1 min�������ɲ���11.2 L(��״��)Cl2����ʱ��Һ��pH��(�����ά�ֲ���)________��
(4)Cl2����������ˮ��ɱ������������һ������������ClO2����������ɱ�������л�ԭ�����ΪCl������������������ˮ������Cl2��ClO2�����ʵ���֮��Ϊ________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
������ˮ��Һ���в�ͬ����Ϊ����Ҫ��ش��������⣺
��1��Na2SO3��Һ�Լ��ԣ���ԭ����______________________________(�����ӷ���ʽ��ʾ)������Һ�и�����Ũ���ɴ�С��˳��Ϊ______________________��
��2����������10mL��ˮ��Һ�м�ˮϡ�ͺ��������������__________(���ţ���ͬ)����С����___________���������____________��
a����Һ������Ũ�� b����ˮ�ĵ���̶�
c��ˮ�����ӻ����� d��c(H+)/ c(NH3��H2O)
��3���������ƣ�NaClO2����һ��ǿ������Ư�����㷺���ڷ�֯��ӡȾ��ʳƷ��ҵ��NaClO2���ʿɷֽ�ΪNaClO3��NaCl��ȡ�������ѱ��ʺ�δ���ʵ�NaClO2�����������Һ���ֱ�������FeSO4��Һ��Ӧʱ������Fe2+�����ʵ��� �����ͬ������ͬ��������ԭ���� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��100 mL FeI2��Һ����ͨ��Cl2������������I2��Fe3����IO3-������Fe3����I2�����ʵ�����n(Cl2)�ı仯��ͼ��ʾ����ش��������⣺
��1����ͼ��֪��I����Fe2����I2�������ӵĻ�ԭ����ǿ������˳��Ϊ________��________��________��
��2����n(Cl2)��0.12 molʱ����Һ�е�������ҪΪ________________________________��
�ӿ�ʼͨ��Cl2��n(Cl2)��0.12 molʱ���ܷ�Ӧ�Ļ�ѧ����ʽΪ______________________��
��3������Һ��n(Cl��)��n(IO3-)��8��1ʱ��ͨ���Cl2�ڱ�״���µ����Ϊ________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�������(K2FeO4)��һ�����͡���Ч�������ɫˮ����������Cl2��O2��ClO2��KMnO4�����Ը�ǿ��������Ⱦ����ҵ�������Ƶø������ƣ�Ȼ���ڵ����£������������Һ�м���KOH�����ͣ�ʹ�������������
(1)�ɷ��Ʊ�������ص���Ҫ��ӦΪ��2FeSO4�� 6Na2O2=2Na2FeO4�� 2Na2O �� 2Na2SO4�� O2��
�ٸ÷�Ӧ�е���������________����ԭ����________��ÿ����1 mol Na2FeO4ת��________mol���ӡ�
�ڼ�Ҫ˵��K2FeO4��Ϊˮ������ʱ���������__________________________________
(2)ʪ���Ʊ��������(K2FeO4)�ķ�Ӧ��ϵ�����������ӣ�Fe(OH)3��ClO����OH����FeO42����Cl����H2O��
��д������ƽʪ���Ƹ�����ط�Ӧ�����ӷ���ʽ��____________________________
________________________________________________________________________��
��ÿ����1 mol FeO42��ת��________mol���ӣ�����Ӧ������ת����0.3 mol���ӣ���ԭ��������ʵ���Ϊ________mol��
�۵����£��ڸ���������Һ�м���KOH�����Ϳ������������(K2FeO4)��˵��ʲô����_______________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
���������������������ˮ��ҵ��ˮ����������������������Ͽ졣ʵ���ҿ��ö�������Ϊ��Ҫԭ���Ʊ�������ء��䲿���������£�
(1)�ڢٲ��в��������������ô�������ԭ����(�û�ѧ����ʽ��ʾ)________________________________________��
(2)KOH��KClO3��MnO2���۷�Ӧ����ī��ɫK2MnO4�Ļ�ѧ����ʽΪ________________________________________��
(3)�ڢܲ�ͨ��CO2����ʹMnO42��������Ӧ������MnO4����MnO2����K2MnO4��ȫ��Ӧʱ��ת��ΪKMnO4�İٷ���ԼΪ________(��ȷ��0.1%)��
(4)�ڢݲ����ȹ��˵�Ŀ����______________________________________��
(5)�ڢ�����Ũ����Һ����ϸС��������ʱ��ֹͣ���ȣ���ȴ�ᾧ��________��ϴ�ӡ������������У��¶Ȳ��˹��ߣ���Ϊ____________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
����һδ��ƽ��������ԭ��Ӧ��
KClO3��PH3��H2SO4��K2SO4��H3PO4��H2O��X
��1���÷�Ӧ�Ļ�ԭ����_________��
��2����֪0.2 mol KClO3�ڷ�Ӧ�еõ�1 mol��������X����X�Ļ�ѧʽ��__________��
��3������������Ӧ����֪__________________����д��ţ���
| A�������ԣ�KClO3��H3PO4 | B�������ԣ�H3PO4��KClO3 |
| C����ԭ�ԣ�PH3��X | D����ԭ�ԣ�X��PH3 |
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com