
ЁОЬтФПЁПЪЕбщЪвжЦБИ1ЃЌ2ЁЊЖўфхввЭщЃЌПЩгУзуСПЕФввДМЯШжЦБИввЯЉЃЌдйгУввЯЉКЭЩйСПЕФфхжЦБИ1ЃЌ2ЁЊЖўфхввЭщЃЌзАжУШчЭМЫљЪОЁЃгаЙиЪ§ОнСаБэШчБэЫљЪОЁЃЛиД№ЯТСаЮЪЬтЃК
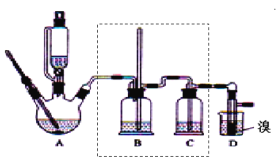
ввДМ | 1ЃЌ2ЁЊЖўфхввЭщ | ввУб | |
зДЬЌ | ЮоЩЋвКЬх | ЮоЩЋвКЬх | ЮоЩЋвКЬх |
УмЖШ/gcm-3 | 0.79 | 2.2 | 0.71 |
ЗаЕу/Ёц | 78.5 | 132 | 34.6 |
ШлЕу/Ёц | -130 | 9 | -116 |
ЃЈ1ЃЉдкДЫжЦБИЪЕбщжаЃЌвЊОЁПЩФмбИЫйЕиАбЗДгІЮТЖШЬсИпЕН170ЁцзѓгвЃЌЦфзюжївЊФПЕФЪЧ__ЁЃ
a.в§ЗЂЗДгІ b.МгПьЗДгІЫйЖШ
c.ЗРжЙввДМЛгЗЂ d.МѕЩйИБВњЮяввУбЩњГЩ
ЃЈ2ЃЉдкзАжУAжаГ§СЫХЈСђЫсКЭввДМЭтЃЌЛЙгІМгШы__ЃЌЦфФПЕФЪЧ__ЁЃзАжУAжаЩњГЩИБВњЮяввУбЕФЛЏбЇЗДгІЗНГЬЪНЮЊ__ЁЃ
ЃЈ3ЃЉЪЕбщЪвжЦШЁввЯЉЃЌГЃвђЮТЖШЙ§ИпЖјЪЙввДМКЭХЈH2SO4ЗДгІЩњГЩЩйСПЕФSO2ЃЌЮЊСЫбщжЄSO2ЕФДцдкВЂГ§ШЅSO2ЖдКѓајЗДгІЕФИЩШХЃЌФГЭЌбЇдкAКЭDжЎМфМгШыСЫBЁЂCСНИізАжУЃЌЦфжаBКЭCжаПЩЗжБ№ЪЂЗХ___ЁЃ
a.ЫсадKMnO4КЭЫЎ b.ЦЗКьКЭNaOHШмвК
c.ЫсадKMnO4КЭNaOHШмвК d.ЦЗКьКЭЫсадKMnO4
ЃЈ4ЃЉМзЁЂввСНзАжУОљПЩгУзїЪЕбщЪвгЩЮоЫЎввДМжЦШЁввЯЉЃЌввЭМВЩгУИЪгЭдЁМгШШ(ИЪгЭЗаЕу290ЁцЃЌШлЕу18.17Ёц)ЃЌЕБИЪгЭЮТЖШДяЕНЗДгІЮТЖШЪБЃЌНЋЪЂгаЮоЫЎввДМКЭХЈСђЫсЛьКЯвКЕФЩеЦПЗХШыИЪгЭжаЃЌКмПьДяЕНЗДгІЮТЖШЁЃМзЁЂввСНзАжУЯрБШНЯЃЌввзАжУгаФФаЉгХЕу__ЃЌаДГіИУЪЕбщжагЩЮоЫЎввДМжЦШЁввЯЉЕФЛЏбЇЗНГЬЪН___ЁЃ
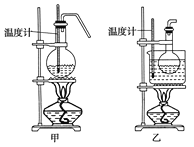
ЃЈ5ЃЉНЋ1ЃЌ2ЁЊЖўфхввЭщДжВњЦЗжУгкЗжвКТЉЖЗжаМгЫЎЃЌеёЕДКѓОВжУКѓЃЌВњЮягІдк__ВуЃЛШєВњЮяжагаЩйСПИБВњЮяввУбЁЃПЩгУ__ЕФЗНЗЈГ§ШЅЁЃ

| ФъМЖ | ИпжаПЮГЬ | ФъМЖ | ГѕжаПЮГЬ |
| ИпвЛ | ИпвЛУтЗбПЮГЬЭЦМіЃЁ | ГѕвЛ | ГѕвЛУтЗбПЮГЬЭЦМіЃЁ |
| ИпЖў | ИпЖўУтЗбПЮГЬЭЦМіЃЁ | ГѕЖў | ГѕЖўУтЗбПЮГЬЭЦМіЃЁ |
| ИпШ§ | ИпШ§УтЗбПЮГЬЭЦМіЃЁ | ГѕШ§ | ГѕШ§УтЗбПЮГЬЭЦМіЃЁ |
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁПЪЕбщЪвгУШМЩеЗЈВтЖЈФГжжАБЛљЫс![]() ЕФЗжзгзщГЩЁЃШЁWgИУжжАБЛљЫсЗХдкДПбѕЦјжаШМЩеЃЌШМЩеКѓЩњГЩЕФ
ЕФЗжзгзщГЩЁЃШЁWgИУжжАБЛљЫсЗХдкДПбѕЦјжаШМЩеЃЌШМЩеКѓЩњГЩЕФ![]() ЕФЬхЛ§гУFзАжУНјааВтСПЃЌЫљашзАжУШчЯТЭМ
ЕФЬхЛ§гУFзАжУНјааВтСПЃЌЫљашзАжУШчЯТЭМ![]() МаГжвЧЦїМАВПЗжМгШШзАжУвбТдШЅ
МаГжвЧЦїМАВПЗжМгШШзАжУвбТдШЅ![]() ЃК
ЃК

(1)ИУЪЕбщзАжУЕФКЯРэСЌНгЫГађЮЊЃКAЁЂCЁЂ____ЁЂFЁЃ(зАжУПЩвджиИДбЁгУ)
(2)ЪЕбщПЊЪМЪБЃЌЪзЯШДђПЊжЙЫЎМаaЃЌЙиБежЙЫЎМаbЃЌЭЈвЛЖЮЪБМфЕФДПбѕЃЌетбљзіЕФФПЕФЪЧ_____ЁЃ
(3)ШМЩеЙмDжаЗХШыCuOЕФзїгУЪЧ___________________ЃЌзАжУBЕФзїгУЪЧ____________________ ЁЃ
(4)ЮЊСЫШЗЖЈДЫАБЛљЫсЕФЗжзгЪНЃЌГ§СЫзМШЗВтСПN2ЕФЬхЛ§ЭтЃЌЛЙашЕУЕНЕФЪ§Онга______________(ЬюзжФИ)ЁЃ
A.ИУАБЛљЫсЕФФІЖћжЪСП B.ЩњГЩЖўбѕЛЏЬМЦјЬхЕФжЪСП
C.ЩњГЩЫЎЕФжЪСП D.ЭЈШыO2ЕФЬхЛ§
(5)дкЖСШЁFзАжУжаЫљХХЫЎЕФЬхЛ§ЪБЃЌвКУцзѓЕЭгвИпЃЌдђЫљВтЦјЬхЕФЬхЛ§________(ЬюЁАЦЋДѓЁБЁЂЁАЦЋаЁЁБЛђЁАЮогАЯьЁБ)ЁЃ
(6)вбжЊЗжзгЪНЮЊ![]() ЕФгаЛњЮявВКЌгаАБЛљЫсжаЕФФГИіЙйФмЭХЃЌЧыЩшМЦЪЕбщжЄУїИУЙйФмЭХ
ЕФгаЛњЮявВКЌгаАБЛљЫсжаЕФФГИіЙйФмЭХЃЌЧыЩшМЦЪЕбщжЄУїИУЙйФмЭХ![]() ЪдМСШЮбЁ
ЪдМСШЮбЁ![]() _____ЁЃ
_____ЁЃ
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁПЯТСаРызгЗНГЬЪНе§ШЗЕФЪЧ( )
A.ЬМЫсЧтФЦШмвКжаМгШыЧтбѕЛЏФЦШмвКЃКHCO3-+OH-=CO2+H2O
B.ЯђДЮТШЫсМиШмвКжаЕЮШыЩйСПFeSO4ШмвКЃЛ2Fe2++ClO-+2H+=Cl-+2Fe3++H2O
C.Na2S2O3гыЯЁH2SO4ЛьКЯЃКS2O32-+2H+=SЁ§+SO2Ёќ+H2O
D.гУЪЏФЋЕчМЋЕчНтMgCl2ШмвКЃК2Cl-+2H2O![]() 2OH-+Cl2Ёќ+H2Ёќ
2OH-+Cl2Ёќ+H2Ёќ
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁПЯѕЫсЙЄвЕЮВЦјжаЕФNOЁЂNO2НјШыДѓЦјКѓЃЌЛсаЮГЩЙтЛЏбЇбЬЮэЁЂЦЦЛЕГєбѕВуЕШЁЃПЩгУЧтбѕЛЏФЦШмвКЖдКЌЕЊбѕЛЏЮяЕФЮВЦјНјааДІРэЃЌЗДгІЕФЛЏбЇЗНГЬЪНШчЯТЃКNO2+NO+2NaOH=2NaNO2+H2OЃЛ2NO2+2NaOH=NaNO2+NaNO3+H2OЁЃАБЦјвВПЩвдгУРДДІРэЕЊбѕЛЏЮяЁЃР§ШчЃЌАБЦјгывЛбѕЛЏЕЊПЩЗЂЩњШчЯТЗДгІЃК4NH3+6NO=5N2+6H2OЁЃНЋвЛЖЈСПNOКЭNO2ЕФЛьКЯЦјЬхЭЈШы300mL5mol/LNaOHШмвКжаЃЌЧЁКУБЛЭъШЋЮќЪеЁЃЯТСаЫЕЗЈВЛе§ШЗЕФЪЧ
A.дкгУАБЦјДІРэЕЊбѕЛЏЮяЪБЃЌЕЊбѕЛЏЮяЗЂЩњЛЙдЗДгІ
B.ЫљЕУШмвКжаNaNO3КЭNaNO2ЕФЮяжЪЕФСПжЎБШПЩФмЮЊ2ЃК1
C.ШєгУАБЦјДІРэЃЌЫљашАБЦјдкБъзМзДПіЯТЕФЬхЛ§ПЩФмЮЊ39.2L
D.дЛьКЯЦјЬхжаNOдкБъзМзДПіЯТЕФЬхЛ§ПЩФмЮЊ16.8L
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁПКуЮТЯТЃЌШнЛ§ОљЮЊ2LЕФУмБеШнЦїMЁЂNжаЃЌЗжБ№гавдЯТСаСНжжЦ№ЪМЭЖСЯНЈСЂЕФПЩФцЗДгІ ![]() ЕФЛЏбЇЦНКтзДЬЌЃЌЯрЙиЪ§ОнШчЯТЃКMЃК
ЕФЛЏбЇЦНКтзДЬЌЃЌЯрЙиЪ§ОнШчЯТЃКMЃК![]() ЃЛ 2minДяЕНЦНКтЃЌЩњГЩ
ЃЛ 2minДяЕНЦНКтЃЌЩњГЩ![]() ЃЌВтЕУДгЗДгІПЊЪМЕНЦНКтCЕФЫйТЪЮЊ
ЃЌВтЕУДгЗДгІПЊЪМЕНЦНКтCЕФЫйТЪЮЊ![]() ЃЛNЃК
ЃЛNЃК![]() ЃЛДяЕНЦНКтЪБ
ЃЛДяЕНЦНКтЪБ![]() ЯТСаЭЦЖЯЕФНсТлжаВЛе§ШЗЕФЪЧЃЈ ЃЉ
ЯТСаЭЦЖЯЕФНсТлжаВЛе§ШЗЕФЪЧЃЈ ЃЉ
A.![]() B.ЦНКтЪБMжа
B.ЦНКтЪБMжа![]()
C.![]() D.
D.![]()
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁПКЃбѓжВЮяШчКЃДјЁЂКЃдхжаКЌгаЗсИЛЕФЕтдЊЫиЃЌЧвЫљКЌЕтдЊЫивдЕтРызгЕФаЮЪНДцдкЁЃЪЕбщЪвРяДгКЃдхжаЬсШЁЕтЕФСїГЬШчЭМЫљЪОЃК

(1)жИГіжЦШЁЕтЕФЙ§ГЬжагаЙиЪЕбщВйзїЕФУћГЦЃКЂй__________ЃЌЂк__________ЁЃ
(2)ВйзїЂкжаПЩЙЉбЁдёЕФгаЛњЪдМСЪЧ_________(ЬюађКХ)ЁЃ
A.МзБНЁЂОЦОЋ B.ЫФТШЛЏЬМЁЂБН C.ЦћгЭЁЂввЫс D.ЦћгЭЁЂИЪгЭ
(3)ЯжашзіЪЙКЃдхЛвжаЕФЕтРызгзЊЛЏЮЊгаЛњШмвКжаЕФЕтЕЅжЪЕФЪЕбщЃЌЪЕбщЪвРягаЩеБЁЂВЃСЇАєЁЂМЏЦјЦПЁЂОЦОЋЕЦЁЂЕМЙмЁЂдВЕзЩеЦПЁЂЪЏУоЭјвдМАБивЊЕФМаГжвЧЦїЁЂвЉЦЗЃЌЩаШБЩйЕФВЃСЇвЧЦїЪЧ__________ЁЂ___________ЁЃ
(4)вЊДгЕтЕФБНШмвКжаЬсШЁЕтКЭЛиЪеБНЃЌЛЙашвЊОЙ§еєСѓВйзїЁЃНјааеєСѓВйзїЪБЃЌашЪЙгУЫЎдЁМгШШЃЌФПЕФЪЧ__________ЃЌзюКѓОЇЬЌЕтдк________жаОлМЏЁЃ
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁПЦћГЕЮВЦјЕФжївЊГЩЗжгаCOЁЂSO2ЁЂЕЊбѕЛЏЮяЕШЃЌПЦбЇМвУЧ-жБжТСІгкЮлШОЮяЕФгааЇЯћГ§ЁЃ
(1)РћгУАБЫЎПЩвдНЋSO2ЁЂЕЊбѕЛЏЮяЮќЪеЃЌдРэШчЯТЭМЫљЪОЁЃ
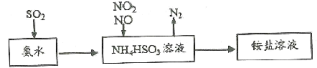
ЧыаДГіNO2КЭNOАДЬхЛ§БШ1ЃК1БЛЮќЪеЪБЗДгІЕФРызгЗНГЬЪН_________________________ЁЃ
(2)ПЦбаЙЄзїепФПЧАе§дкГЂЪдвдЖўбѕЛЏюб(TiO2)ДпЛЏЗжНтЦћГЕЮВЦјЕФбаОПЁЃ
ЂйМКжЊЃК2NO(g)+O2(g)=2NO2(g) ЁїH1=-113.0kJЁЄ mol-1
2SO2(g)+O2(g)=2SO3(l) ЁїH2=-288.4kJЁЄ mol-1
N2(g)+O2(g)![]() 2NO(g) ЁїH3=+180.5kJЁЄ mol-1
2NO(g) ЁїH3=+180.5kJЁЄ mol-1
ЧыХаЖЯЗДгІNO2(g)+SO2(g)=NO(g)+SO3(l) ЁїH4ЃЌдкЕЭЮТЯТФмЗёздЗЂНјаа_______(ЬюЁАФмЁБЛђЁАЗёЁБ)ЃЌРэгЩЪЧ__________________________ЁЃ
ЂкМКжЊTiO2ДпЛЏЮВЦјНЕНтдРэЮЊЃК
2CO(g)+O2(g)![]() 2CO2(g)ЃЛ2H2O(g)+4NO(g)+3O2(g)
2CO2(g)ЃЛ2H2O(g)+4NO(g)+3O2(g)![]() 4HNO3(g)
4HNO3(g)
iЁЂдквЛЖЈЬѕМўЯТЃЌФЃФтCOЁЂNOЕФНЕНтЃЌЕУЕННЕНтТЪ(МДзЊЛЏТЪ)ЫцЪБМфБфЛЏШчЭМЫљЪОЃЌ
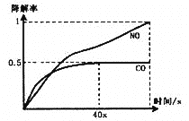
ЗДгІ40УыКѓМьВтЕНЛьКЯЦјЬхжаN2ХЈЖШЩЯЩ§ЃЌHNO3ЦјЬхХЈЖШгаЫљНЕЕЭЃЌЧыгУЛЏбЇЗНГЬЪНВЂНсКЯЛЏбЇЗДгІдРэжЊЪЖНтЪЭПЩФмЕФдвђ____________________________________________ЁЃ
iiЃЌСЄЧрЛьФ§ЭСвВПЩНЕНтCOЁЃШчЭМЮЊдкВЛЭЌПХСЃМфЯЖЕФСЄЧрЛьФ§ЭС(ІСЁЂІТаЭ)дкВЛЭЌЮТЖШЯТЃЌЗДгІЯрЭЌЪБМфЃЌВтЕУCOНЕНтТЪБфЛЏЁЃНсКЯЭМБэЛиД№ЯТСаЮЪЬтЃК
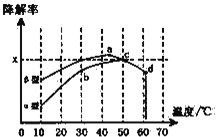
вбжЊдк50ЁцЪБдкІСаЭСЄЧрЛьФ§ЭСШнЦїжаЃЌЦНКтЪБO2ХЈЖШЮЊ0.01molЁЄL-1ЃЌЧѓДЫЮТЖШЯТCOНЕНтЗДгІЕФЦНКтГЃЪ§____________________(гУКЌxЕФДњЪ§ЪНБэЪО)ЃЛвдІТаЭСЄЧрЛьФ§ЭСПХСЃЮЊдиЬхЃЌНЋTiO2ИФЮЊДпЛЏаЇЙћИќКУЕФTiO2ФЩУзЙмЃЌдк10ЁЋ60ЁцЗЖЮЇФкНјааЪЕбщЃЌЧыдкЭМжагУЯпЖЮгывѕгАЃЌЗТееЁАЪОР§ЁБУшЛцГіCOНЕНтТЪЫцЮТЖШБфЛЏЕФЧњЯпПЩФмГіЯжЕФзюДѓЧјгђЗЖЮЇ(ЪОР§ЃК![]() )_____________________ЁЃ
)_____________________ЁЃ
(3)РћгУШчЭМЫљЪОзАжУ(ЕчМЋОљЮЊЖшадЕчМЋ)вВПЩЮќЪеSO2ЃЌВЂгУвѕМЋХХГіЕФШмвКЮќЪеNO2ЃЌbМЋЕФЕчМЋЗДгІЪНЮЊ_______________________________ЁЃ

ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁПИпЬњЫсФЦ(Na2FeO4)ЪЧвЛжжаТаЭЁЂИпаЇЕФЫЎДІРэМСЃЌгыЫЎЗДгІЕФЛЏбЇЗНГЬЪНЮЊ4Na2FeO4ЃЋ10H2O===4Fe(OH)3Ё§ЃЋ3O2ЁќЃЋ8NaOHЁЃЕчНтжЦБИNa2FeO4зАжУЪОвтЭМШчЭМЫљЪОЁЃ
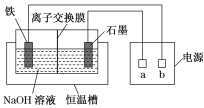
(1)aЪЧЕчдДЕФ________(ЬюЁАе§ЁБЛђЁАИКЁБ)МЋЁЃЕчНтЪБЃЌЪЏФЋЕчМЋИННќШмвКЕФМюад________(ЬюЁАдіЧПЁБЁАМѕШѕЁБЛђЁАВЛБфЁБ)ЁЃ
(2)ЬњЕчМЋЕФЗДгІЪНЮЊ_________________________________________________ЁЃ
(3)ЮЌГжвЛЖЈЕФЕчСїЧПЖШКЭЕчНтЮТЖШЃЌNaOHЦ№ЪМХЈЖШЖдNa2FeO4ХЈЖШгАЯьШчЭМ(ЕчНтвКЬхЛ§ЯрЭЌЕФЧщПіЯТНјааЪЕбщ)ЁЃ
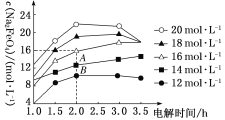
ЂйЕчНт3.0 hФкЃЌЫцNaOHЦ№ЪМХЈЖШдіДѓЃЌNa2FeO4ХЈЖШБфЛЏЧїЪЦЪЧ________(ЬюЁАдіДѓЁБЁАВЛБфЁБЛђЁАМѕаЁЁБ)ЁЃ
ЂкЕБNaOHЦ№ЪМХЈЖШЮЊ16 molЁЄLЃ1ЪБЃЌ1.0ЁЋ2.0 hФкЩњГЩNa2FeO4ЕФЫйТЪЪЧ__________molЁЄLЃ1ЁЄhЃ1ЁЃ
(4)ЬсДПЕчНтЫљЕУNa2FeO4ЃЌВЩгУжиНсОЇЁЂЙ§ТЫЁЂЯДЕгЁЂЕЭЮТКцИЩЕФЗНЗЈЃЌдђЯДЕгМСзюКУбЁгУ________(ЬюБъКХ)ШмвККЭвьБћДМЁЃ
AЃЎFe(NO3)3ЁЁ ЁЁBЃЎNH4ClЁЁ ЁЁCЃЎCH3COONa
(5)ДЮТШЫсФЦбѕЛЏЗЈвВПЩвджЦЕУNa2FeO4ЁЃ
вбжЊЃК2H2(g)ЃЋO2(g)===2H2O(l)ЁЁІЄHЃНa kJЁЄmolЃ1
NaCl(aq)ЃЋH2O(l)===NaClO(aq)ЃЋH2(g)ЁЁІЄHЃНb kJЁЄmolЃ1
4Na2FeO4(aq)ЃЋ10H2O(l)===4Fe(OH)3(s)ЃЋ3O2(g)ЃЋ8NaOH(aq)ЁЁІЄHЃНc kJЁЄmolЃ1
ЗДгІ2Fe(OH)3(s)ЃЋ3NaClO(aq)ЃЋ4NaOH(aq)===2Na2FeO4(aq)ЃЋ3NaCl(aq)ЃЋ5H2O(l)ЕФІЄHЃН_______kJЁЄmolЃ1ЁЃ
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК ЬтаЭЃК
ЁОЬтФПЁПЯТСагаЙиЮФЯзМЧдижаЩцМАЕФЛЏбЇжЊЪЖБэЪіВЛе§ШЗЕФЪЧЃЈ ЃЉ
A.ЁАвддјЧрЭПЬњЃЌЬњГрЩЋШчЭЁБЫЕУїЮвЙњЙХДњОЭеЦЮеСЫЁАЪЊЗЈвБН№ЁБММЪѕ
B.ЁАЧНЫњбЙЬЧЃЌШЅЭСЖјЬЧАзЁБжаЕФЭбЩЋЙ§ГЬЗЂЩњСЫЛЏбЇБфЛЏ
C.ЁАЕЄЩАЩежЎГЩЫЎвјЃЌЛ§БфгжЛЙГЩЕЄЩАЁБНВЕФЪЧЕЅжЪгыЛЏКЯЮяжЎМфЕФЛЅБф
D.ЁЖБОВнИйФПЁЗжаМЧдиЃКЁАЩеОЦЗЧЙХЗЈвВЃЌЁЁЃЌгУХЈОЦКЭдуШыъЕЃЌеєСюЦјЩЯЃЌгУЦїГаШЁЕЮТЖЁЃЁБЩцМАЕФВйзїЗНЗЈЪЧеєСѓ
ВщПДД№АИКЭНтЮі>>
ЙњМЪбЇаЃгХбЁ - СЗЯАВсСаБэ - ЪдЬтСаБэ
КўББЪЁЛЅСЊЭјЮЅЗЈКЭВЛСМаХЯЂОйБЈЦНЬЈ | ЭјЩЯгаКІаХЯЂОйБЈзЈЧј | ЕчаХеЉЦОйБЈзЈЧј | ЩцРњЪЗащЮожївхгаКІаХЯЂОйБЈзЈЧј | ЩцЦѓЧжШЈОйБЈзЈЧј
ЮЅЗЈКЭВЛСМаХЯЂОйБЈЕчЛАЃК027-86699610 ОйБЈгЪЯфЃК58377363@163.com