
��ҵ�����ý�̿��ʯ��Ҥ��ȼ�շ��ȣ�ʹʯ��ʯ�ֽ�����CO2����Ҫ��Ӧ���£�
C+O2��CO2 �٣� CaCO3��CO2��+CaO ��
��1����̼���95%��ʯ��ʯ2.0 t������ȫ�ֽ⣨�����ʲ��ֽ⣩���ɵñ�״����CO2�����Ϊ_________________m3��
��2��������CaCO3�ͽ�̿���٢���ȫ��Ӧ����Ҥ�������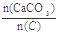 =2.2ʱ��Ҥ����CO2������������Ϊ���٣��������ֻ��N2��O2���������Ϊ4��1����ͬ��
=2.2ʱ��Ҥ����CO2������������Ϊ���٣��������ֻ��N2��O2���������Ϊ4��1����ͬ��
��3��ij��Ҥ���ɷ����£�O2 0.2%��CO 0.2%��CO2 41.6%������ΪN2����˴�Ҥ������� Ϊ��ֵ��
Ϊ��ֵ��
��1��425.6 ��2��44.4%��3��1.90
���������������1�����ݷ�Ӧ�ڵĻ�ѧ����ʽ��֪CO2�����ʵ�������CaCO3�����ʵ��������Ա�״����CO2�����Ϊ��2.0��95%��106g��100g/mol��22.4L/mol=425600L����425.6m3��
��2����n��C��=1mol����n��CaCO3��=2.2mol���������е�O2��C��ȫ��Ӧʱ������Ҥ����CO2����������1molCȼ������1mol CO2��2.2mol CaCO3�ֽ�����2.2mol CO2������CO2���������=��1+2.2���£�1+2.2+4����100%=44.4%
��3����Ҥ��Ϊ100mol����n��N2��=100mol-41.6mol-0.2mol-0.2mol=58mol�����Է�ӦǰO2������Ϊ��58mol��4=14.5mol���μӷ�Ӧ��O2Ϊ��14.5mol-0.2mol=14.3mol��2C+O2=2CO������0.2mol CO��O2 0.1mol��C+O2=CO2����C��Ӧ����CO2��O2Ϊ��14.3mol-0.1mol=14.2mol�����ɵ�CO2Ϊ14.2mol��CaCO3�ֽ����ɵ�CO2Ϊ��41.6mol-14.2mol=27.4mol����CaCO3�����ʵ���Ϊ27.4mol������Ҥ������� Ϊ��27.4mol����0.2mol+14.2mol��=1.90
��27.4mol����0.2mol+14.2mol��=1.90
���㣺���⿼������ļ��㡣



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ҵ���ú�п����(��FeO��CuO������)���Ƶû���ZnO���������£�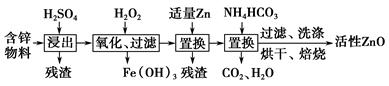
(1)���������У������õ���60%H2SO4(1.5 g��cm��3)����������H2SO4 100 mL��Ҫ18.4 mol��L��1��ŨH2SO4________ mL(����һλС��)��
(2)����������H2O2����Fe(OH)3�������֣�û��Cu(OH)2�������֣�����Һ��c(Fe3��)��2.6��10��18 mol��L��1������Һ��c(Cu2��)��ȡֵ��Χ��________mol��L��1��(��֪Ksp[Fe(OH)3]��2.6��10��39��
Ksp[Cu(OH)2]��2.2��10��20)
(3)����NH4HCO3�����ɵij�������̬��ΪZna(OH)b(CO3)c(a��b��cΪ������)�����ּ�ʽ̼��пA��B�Ļ���A��a��5��b��6�������ɼ�ʽ̼��пA�Ļ�ѧ����ʽΪ__________________________________________________��
(4)ȡϴ�ӡ���ɺ�ļ�ʽ̼��пA��B�Ļ����49.70 g�������ʵ���Ϊ0.10 mol�����±�����ȫ�ֽ�õ�37.26 g ZnO��3.584 L CO2(��״����)��ˮ��ͨ�����������ʽ̼��пB�Ļ�ѧʽ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
����ʯ����Ҫ�ɷ�ΪK2SO4��Al2��SO4��3��2Al2O3��6H2O�������������Fe2O3���ʡ�ijУ�о�С����������ʯ�Ʊ������������������£�
��1�������ա������з�Ӧ�Ļ�ѧ����ʽΪ Al2��SO4��3��
Al2��SO4��3�� S
S
 Al2O3��
Al2O3�� ______����
______����
��2��������Һ�ͷ����IJ�����________�����ܽ⡱ʱ��Ӧ�����ӷ���ʽΪ_________________________________________________________________��
��3��������pH������ˡ�ϴ��Al��OH��3������֤��������ϴ�Ӹɾ���ʵ�������������________��
��4����ĸҺ���пɻ��յ�������________��
��5���������ա�������ǡ������48 g��ƣ������������տɵõ�________ g����������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
����98%��ŨH2SO4��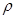 =1��84g/cm3�����Ƴ�Ũ��Ϊ0��5mol/L��ϡ����500mL��
=1��84g/cm3�����Ƴ�Ũ��Ϊ0��5mol/L��ϡ����500mL��
��1���뽫���в�������ȷ��������ں����ϣ�
| A������Ͳ��ȡŨH2SO4 |
| B�������ߵ�ҡ�� |
| C���ý�ͷ�ιܼ�ˮ���̶� |
| D��ϴ���ձ��ڱںͲ�����������ϴҺת������ƿ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�Իش��������⣺
��1����֪24��A��40��Bǡ����ȫ��Ӧ����0��8molC��32��D����C��Ħ������Ϊ ��
��2����ͼΪʵ����ijŨ�����Լ�ƿ�ı�ǩ�ϵ��й����ݣ��Ը��ݱ�ǩ�ϵ��й����ݻش��������⣺
�ٸ�Ũ������HCl�����ʵ���Ũ��Ϊ mol/L��
����ʵ��������450mL2��38 mol/L��ϡ���ᣬ���ø�Ũ����________ mL,����ͼ��ʾ��������������Һ�϶�����Ҫ����________(�����)������������Һ�����õ��IJ���������______________________(����������)��
��ʵ����������������ȷ��������ʱ���ӿ̶��ߣ���������ҺŨ��________2��38mol/L (����ڡ������ڡ���С�ڡ�����ͬ)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ʵ������Na2CO3·10H2O��������0.02 mol/L��Na2CO3��Һ480 mL��
��1���� ʵ�����õ��IJ�����������Ͳ�����������ձ�����ȱ�� ��
�� ʵ���������õ��������������÷ֱ��� �� ��
��2) Ӧ��������ƽ��ȡNa2CO3·10H2O���������Ϊ�� g��
��3���������в�����������Һ��Ũ�ȸ���ʲôӰ�죬���������������ո�
��̼���ƾ���ʧȥ�˲��ֽᾧˮ�� ���á���������ij��������������壻
��̼���ƾ��岻�������л����Ȼ��ƣ� ������ƿ��ԭ����������ˮ��
�ݶ���ʱ����Һ�棻��ҡ�Ⱥ���Һ����ڿ̶����ټ�ˮ��
��������������ҺŨ�ȣ�a��ƫ�ߵ��� ��
b��ƫ�͵��� ��
c����Ӱ����� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
Ϊά������ѪҺ�е�Ѫ�Ǻ������ڸ�������Һʱ��ͨ����������ע��Һ����ͼ��ҽԺ������Һʹ�õ�һƿ��������Ϊ5%�������ǣ�C6H12O6��ע��Һ��ǩ��������۲��ǩ���������ݺ���㣺
��1������Һ�����ʵ���Ũ��Ϊ mol��L��1���������2λ��Ч���֣���
��2������Һ���ܶ�Ϊ g��mL��1��
��3������Ӹ�ƿ��ȡ��75g������ע��Һ�������Ϊ15%��ע��Һ����Ҫ���� g�����ǹ��壨��ȷ��0.1g����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��32.64gͭ��140mLһ��Ũ�ȵ����ᷴӦ��ͭ��ȫ�ܽ������NO��NO2��������ڱ�״���µ����Ϊ11.2L����ش�
��1��NO�����Ϊ L��NO2�����Ϊ L��
��2��������������ȫ���ͷź�����Һ����V mL amol��L��1��NaOH��Һ��ǡ��ʹ��Һ�е�Cu2��ȫ��ת���ɳ�������ԭ������Һ��Ũ��Ϊ mol/L��
��3����ʹͭ�����ᷴӦ���ɵ�������NaOH��Һ��ȫ��ת��ΪNaNO3��������Ҫ30%��˫��ˮ g��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��һ��пƬ��������CuSO4��Һ1L�У����Թ۲쵽пƬ�����л�ɫ������������������ʱпƬ����������0.1g����������Һ��仯���Բ��ƣ�����
��1����1���μӷ�Ӧ�Ļ�ԭ�������ʵ����� ��
��2����ԭ��������ʵ����� ��
��3������ZnSO4�����ʵ���Ũ���� ��
��4����Һ������ ��������ӡ����١���
��5��д���÷�Ӧ�����ӷ���ʽ ��
�鿴�𰸺ͽ���>>
����ѧУ��ѡ - ��ϰ���б� - �����б�
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com